Figures & data
TABLE I. Common pharmacokinetic gene variants included in clinical testing panels for antipsychotic therapy. *Designation for allele; CYP, cytochrome P450; EM, extensive metabolizer; NM, normal metabolizer; IM, intermediate metabolizer; PM, poor metabolizer; UM, ultrarapid metabolizer
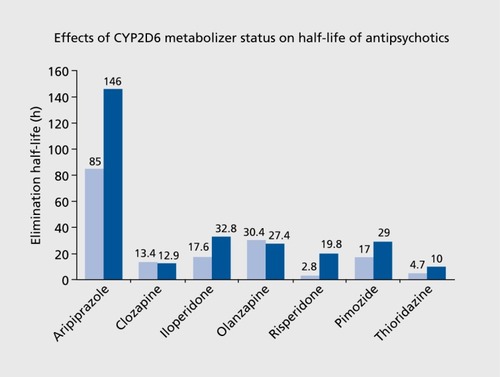
TABLE II. Drug metabolism pathways, labeling, and guideline information, and drug exposure (AUC) implications of pharmacogenetic information in antipsychotic medications. Bold font, major metabolism pathway; *Recommendation is based on haloperidol plasma levels studies.Citation4,Citation17Percent Change in AUC: AUC in PM - AUC in EM/AUC in EM*100 AUC, area under the concentration-time curve; CPIC, Clinical Pharmacogenetics Implementation Consortium; DPWG, Royal Dutch Association for the Advancement of Pharmacy-Pharmacogenetics Working Group; EM, extensive metabolizer; FDA, US Food and Drug Administration; N/A, not available; NS, nonsignificant; PGx, pharmacogenetic; PK, pharmacokinetic; PM, poor metabolizer