Figures & data
Table 1. Model inputs according to clinical events
Table 2. Treatment costs through social insurance
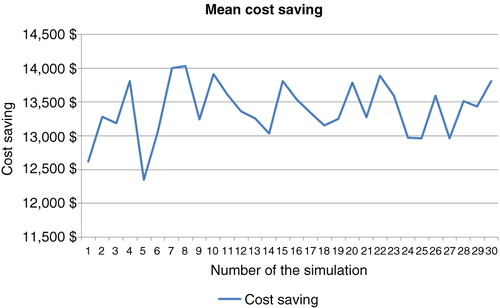
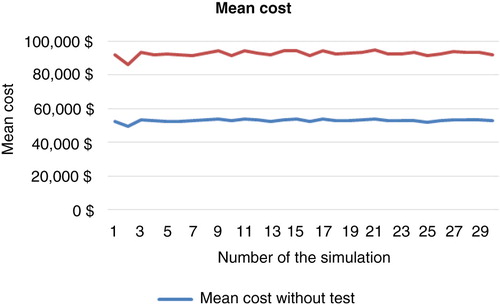
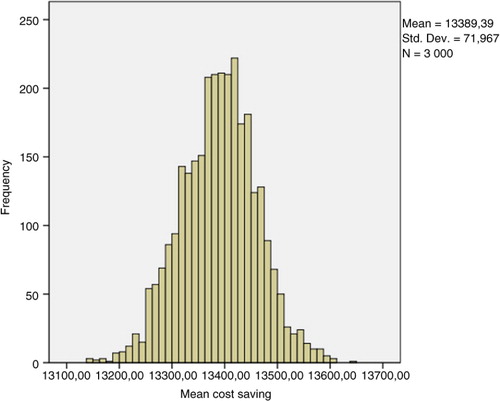
Table 3. Sample of 30 simulations using the bootstrap technique (10,000 patients, 5 years)
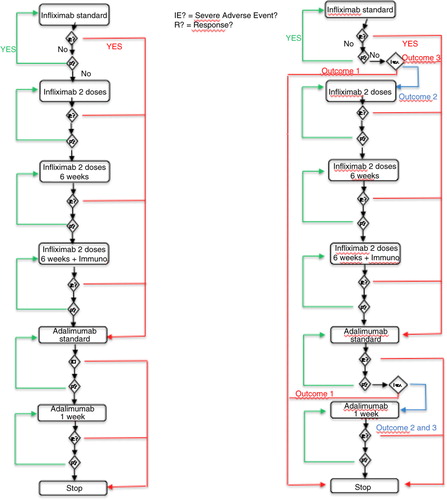
Table 4. Probabilities of treatment success following empirical drug optimization at every consultation
Table 5. Probabilities of the three outcomes of the diagnostic test, decision based on trough levels and antibodies to IFX, and efficacy of every therapeutic decision based on literature data
Table 6. Probabilities of the three outcomes of the diagnostic test, decision based on trough levels and antibodies to ADA, and efficacy of every therapeutic decision based on literature data
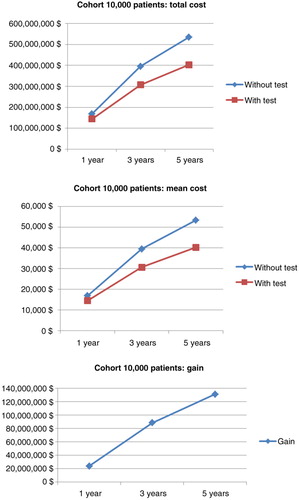
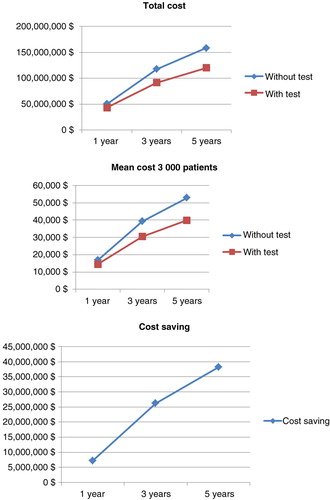
Table 7. Drug costs of anti-TNF therapy among 3,000 patients with empirical dose escalation compared with a test-based strategy