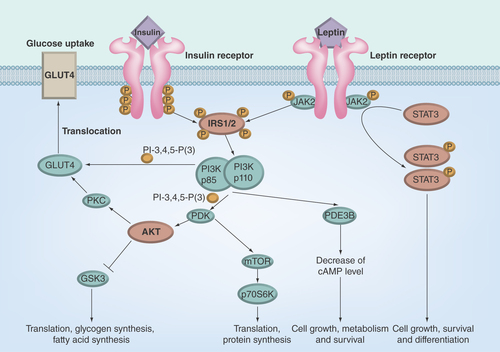
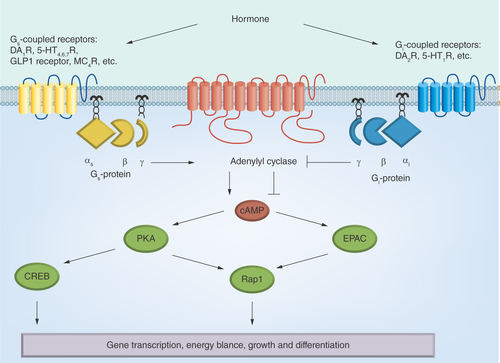
Figures & data
Table 1. The approach to the improvement of brain signaling systems regulated by hormones and neuromediators in Type 2 diabetes mellitus and metabolic syndrome.