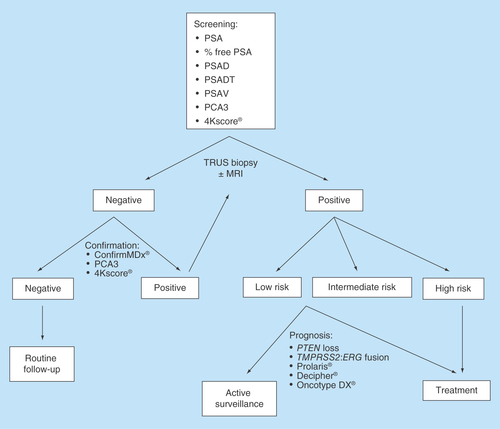
Figures & data
Table 1. Description of prostate-specific antigen-based markers utilized in clinical practice for prostate cancer diagnosis.
Table 2. Commercially available biomarkers utilized in clinical practice for prostate cancer diagnosis.
Table 3. Commercially available RNA-based gene panels utilized in clinical practice for prostate cancer prognosis.