Figures & data
Table 1. Baseline Demographic Characteristics of the Study Population
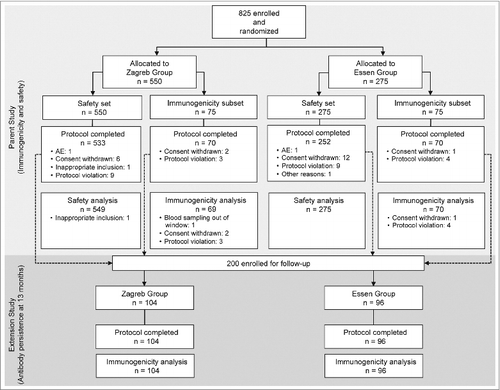
Figure 2. Rabies virus neutralizing antibody concentrations (GMC) in the Zagreb and Essen groups (PP set) on Days 7, 14, 42, and Month 13. Error bars and values in parenthesis represent 95% CI. *Dotted reference line: the WHO recommends a specific antibody level of 0.5 IU/ml as being indicative of an adequate immune response after vaccination.
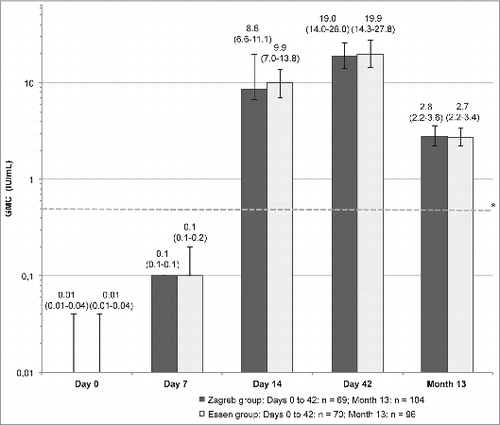
Figure 3. Rates of seroconversion (defined as RVNA ≥ 0.5 IU/mL) in the Zagreb and Essen groups (PP set) on Days 7, 14, 42, and Month 13 month. Error bars and values in parenthesis represent 95% CI.
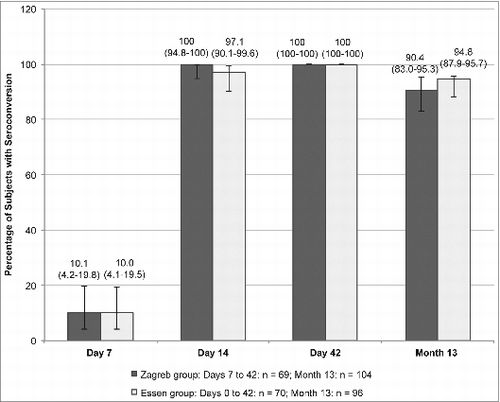
Table 2. Local and systemic reactions occurring in the Zagreb and Essen groups after the first vaccination and during the whole trial duration
Table 3. Incidence of all adverse events occurring in the Zagreb and Essen groups during the trial