Figures & data
Figure 1 Biodistributions of the 188Re-cMORF effector from plotted individually in %ID/g (parts A to I) and, in the case of tumor only, also in %ID (part J)
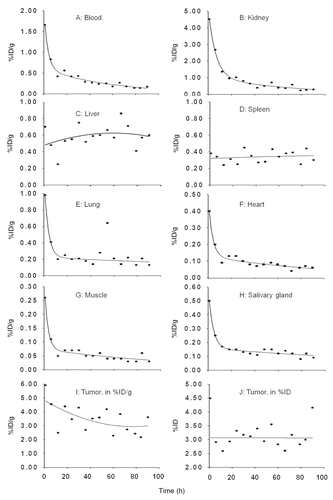
Figure 2 (A) The tumor to blood ratios with time since radioactivity administration. The solid line represents the linear best fit. (B) Histograms showing tumor to normal tissue AUC ratios for listed organs.
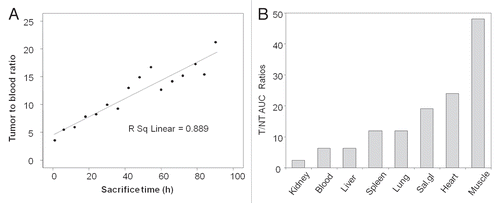
Figure 3 Anterior, left lateral and posterior projections for each SPECT/CT acquisition of one pretargeted mouse imaged at 3.5 h (A) and 10 h (B) after injection of 188Re-cMORF. Except for the intense urine radioactivity at 3.5 h, the radioactivity is essentially restricted to tumor at both time points.
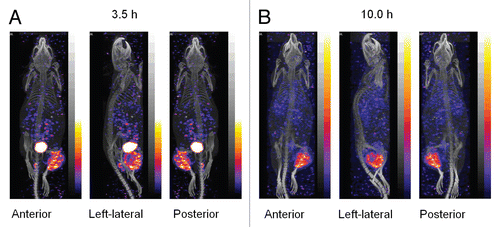
Figure 4 Histograms presenting the increase in tumor weight at day 5 (A) and day 11 (B) after 188Re administration. The estimated tumor dimensions over time for groups 0 (0 mCi) and 4 (1.40 mCi) are also presented (C). Average, N = 5. Error bars signify one standard deviation.
Figure 5 Representative H&E stained tissue sections of tumor and several normal tissues from untreated mice and mice receiving the highest 188Re dose.
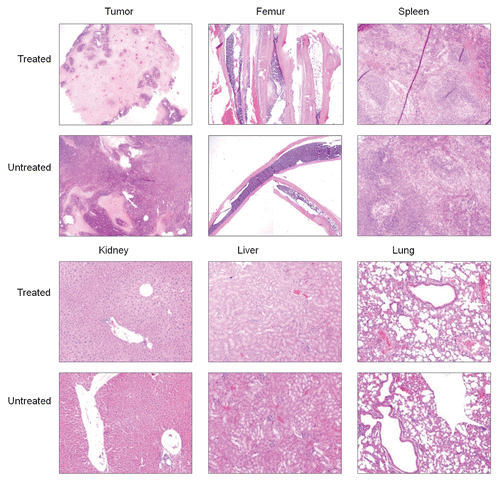
Table 1 Individual biodistributions in %ID/g and, for stomach and intestines, %ID/organ from 1–90 h post IV injection of 188Re-cMORF to tumored mice pretargeted 48 h earlier with MORF-CC49
Table 2 The AUCs and absorbed radiation doses for tumor and organs
Table 3 Tumor dimension (width × thickness of the tumor thigh, cm2) of mice receiving only 30 µg of the MORF-CC49 compared to untreated mice