Figures & data
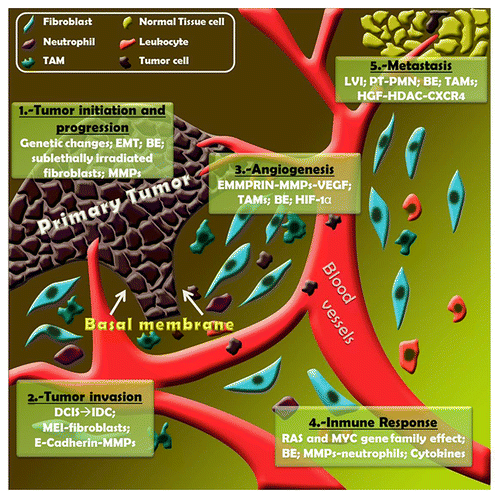
Figure 1. Microenvironmental players in breast cancer development. (1) Tumor initiation and progression: genetic changes involved (genetic profile changes, microsatellite instability, tumor suppressor gene and oncogene point mutations, LOH); epithelial to mesenchymal transition (EMT); bystander effects (BEs) are related to soluble factors (TNFα, free radicals -ROS, RNS-); sublethally-irradiated fibroblasts enhance tumor growth; metalloproteinases (MMPs) control the growth-antigrowth balance. (2) Tumor invasion: DCIS to IDC transition is associated with stromal gene expression changes; MMPs are responsible for altering extracellular matrix remodeling; mesenchymal-epithelial interactions (MEIs) are a consequence of the transcriptional response of fibroblasts to Wnt signals; E-cadherin cleavage is produced by MMPs. (3) Angiogenesis: extracellular matrix metalloproteinase inducer (EMMPRIN) appears to modulate VEGF secretion in an MMP-dependent manner; hypoxic conditions stimulate HIF-1α production by TAMs. (4) Immune response: leukocyte recruitment and tumor microenvironment (TME) remodeling is performed by some members of RAS and MYC gene families; Some MMPs modulate the bioactivity of CXCL-1/KC and CXCL-8/IL-8 implicated in chemotactic activity on neutrophils; cytokines control the immune and inflammatory milieu. (5) Metastasis: lymphovascular invasion (LVI) is related to the presence of lymph node metastasis and is a marker of poor prognosis. Mobilization of bone marrow-derived cells induced by primary tumor (PT) generates pre-metastatic niche (PMN); epigenetics regulation; HGF-HDAC-CXCR-4 interactions related to invasive phenotype.