Figures & data
Figure 1. Zinc concentration in liver tissue from HCC and normal subjects. “NORMAL” is liver tissue from non-cancerous patients. “Non-Cirr” is normal tissue adjacent to the HCC malignant tissue. “Cirr” is cirrhotic tissue adjacent to the HCC malignant tissue. “Malig” is the HCC tumor tissue region. *P < 0.001 for “Malig” zinc levels compared with the other groups. (Taken and modified from ref. Citation7.)
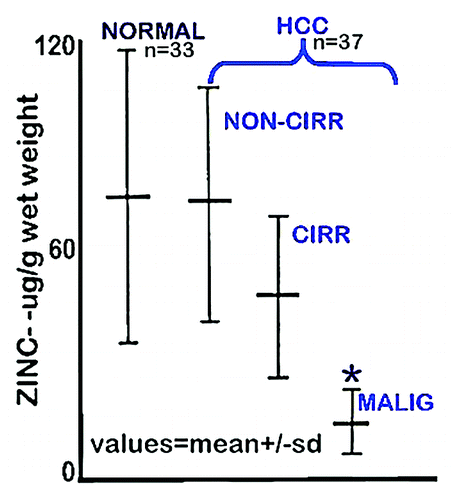
Figure 2. In situ zinc staining of normal vs. HCC tissues. (A) Dithizone staining showing black ZnDTZ stain of high levels of cellular zinc in normal parenchyma, and loss ZnDTZ in HCC. (B) Enlargement of normal liver and Grade1 HCC to show the ZnDTZ throughout the parenchymal hepatocyte component; and loss of zinc stain in well-differentiated hepatoma cells. (C) Zinquin stain showing high fluorescence by zinc in normal hepatocytes and marked decrease in hepatoma cells. (Taken and modified from ref. Citation14.)
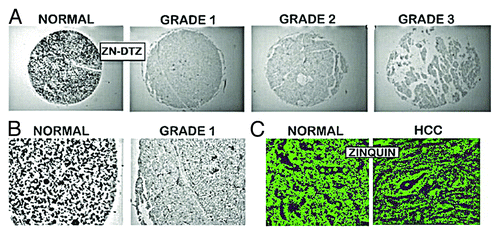
Figure 3. In situ identification of ZIP14 in normal liver vs. HCC. (A) ZIP14 IHC showing high transporter abundance in the normal hepatocytes and its loss in HCC hepatoma cells. (B) Higher magnification showing ZIP14 transporter localized to the normal hepatocyte plasma membrane, and the absence of plasma membrane transporter in the hepatoma cells. (C) In situ RT-PCR showing high expression of ZIP14 in normal hepatocytes, and silencing of expression in the well-differentiated Grade 1 hepatoma cells. (Taken and modified from ref. Citation14.)
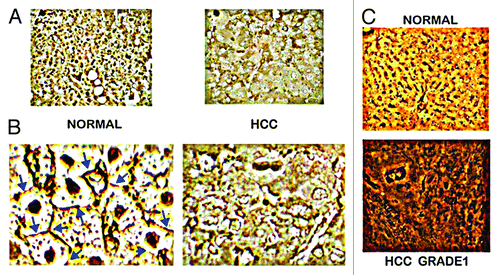
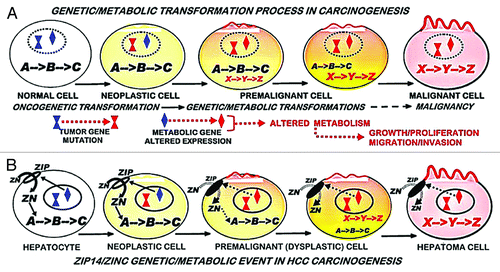
Figure 4. Concept of (A) the involvement of the genetic/metabolic transformation in carcinogenesis, and (B) the involvement of ZIP14 gene expression and decreased zinc in hepatocarcinogenesis.