Figures & data
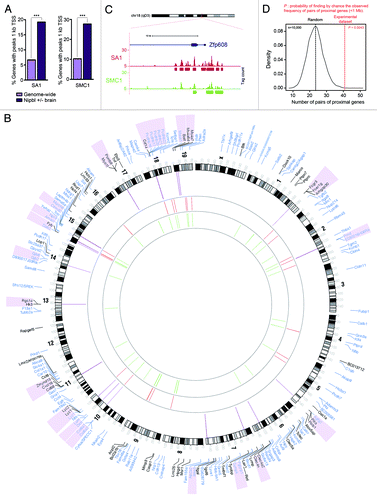
Figure 1. TEM8 structure and antibody binding. (A) TEM8 is a type I membrane protein containing a vWF type A (VWA) domain in its extracellular domain. (B) The image depicts the selective binding of anti-TEM8 blocking antibodies (blue) to TEM8 receptors (orange) that are overexpressed on the surface of tumor endothelium. Tumor cells are depicted in yellow.
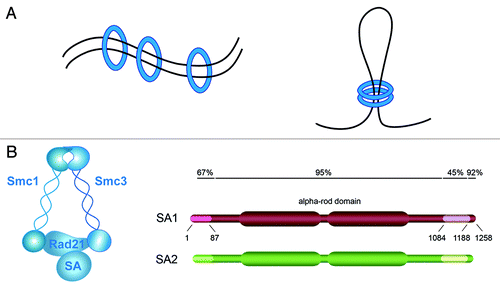
Figure 2. TEM8 antibodies recognize proteins in both Tem8 WT and KO mice. (A) The rabbit anti-TEM8 polyclonal antibody from AbCAM (cat # ab19387) generated against the TEM8 extracellular domain peptide LMKLTEDREQIRQGLE was used to detect TEM8 in various normal tissues from Tem8 WT and KO mice. Although the polyclonal antibody recognized both mouse TEM8 (mTEM8) and human TEM8 (hTEM8) in 293 cells transfected with the corresponding Tem8 genes (asterisk), when tested against lysates from various normal mouse tissues it reacted predominantly with proteins that were present in both the Tem8 WT and KO mice. (B) PCR genotyping assay used to confirm the Tem8 WT and KO status of the samples used in A.
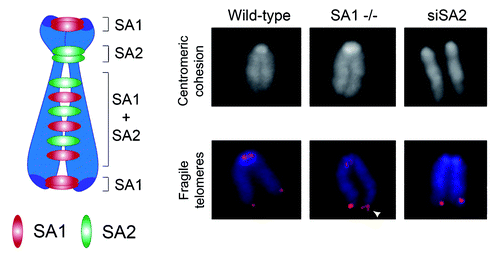
Figure 3. Amino acid starvation induces TEM8 expression. (A) Real-time quantitative PCR revealed a gradual increase in TEM8 mRNA expression following amino acid (AA) depletion from the growth medium. (B) Immunoblotting (IB) with anti-TEM8 antibodies (clone SB5) revealed an increase in TEM8 protein 24 h following removal of amino acids from the growth medium. β actin was used as a loading control.