Figures & data
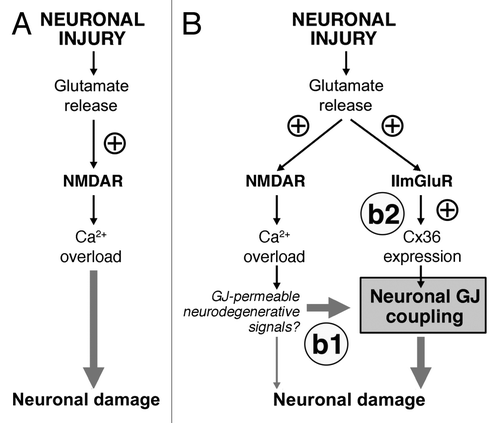
Figure 1. Glutamate-dependent excitotoxicity during neuronal injuries. (A) Traditional model of the mechanisms for glutamate-dependent excitotoxicity. (B) Novel model of the mechanisms of glutamate-dependent excitotoxicity. b1: Existing neuronal gap junctions (GJ) contribute substantially to neuronal death caused by overactivation of NMDARs. b2: New neuronal gap junctions are induced by activation of group II mGluRs (IImGluRs) and also contribute to glutamate-dependent neuronal death. ⊕, this sign indicates the increase in the receptor activity or expression of Cx36. See text for details. Figure reprinted with permission: Wang Y, Song J-H, Denisova JV, Park W-M, Fontes JD, Belousov AB. Neuronal gap junction coupling is regulated by glutamate and plays critical role in cell death during neuronal injury. J Neurosci 2012; 32:713-25; PMID:22238107; 10.1523/jeurosci.3872-11.2012.