Figures & data
Figure 1. Construction of a bacterial two-hybrid library and identification of DynA (YpbR) interation partner (A) Gene length distribution of B. subtilis. (B) XbaI and SacI restriction analysis of random clones (n = 14) isolated from the pUT18 genomic library. The average insert size is 1030 bp. P, plasmid backbone. (C) Validation of plasmids found in the two-hybrid screen. Plasmids were transformed into E. coli BTH101 carrying either pKT25_dynA (+) or empty pKT25 (−) as indicated in the bottom right position. (D) Second round of validation with full length genes cloned into pUT18 vector. Note, only three full length constructs show interaction with DynA. (E) Interaction matrix of YwpG, RNaseY, and YneK with the D1 and D2 subdomains of DynA and their nucleotide-binding mutants. D1M harbors the K56A mutation and D2M the K625R mutation, respectively.
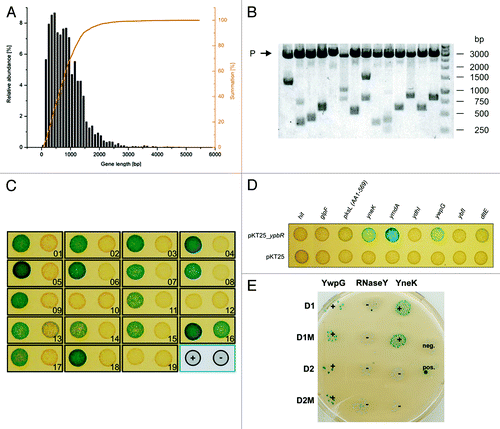
Table 1. Two-hybrid fusions encoded by the prey plasmids. Amino acid residues of the expressed fusion proteins are indicated in brackets
Figure 2. Localization of DynA interacting proteins. Strains (FBB018, PSB001–006) were grown in CH medium to mid-exponential phase and induced for 60 min with 0.1% xylose. YneK, RNaseY, and YwpG show similar localization compared with DynA. All proteins localize to the membrane and form foci that are often found at midcell positions. Scale bar 2 µm.
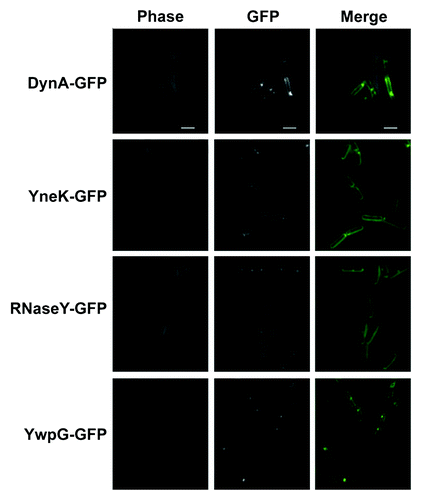
Table 2. Localization of DynA (ypbR) interaction partners into foci associated with the cell membrane
Figure 3.Localization of DynA interacting proteins in a minJ mutant background. Strains (PSB007–009) were grown in CH medium to mid-exponential phase and induced for 60 min with 0.1% xylose. YneK and RNaseY are localized to the membrane. Note that prominent foci are absent in ΔminJ strain backgrounds. YwpG localizes into discrete foci at the cell poles in absence of MinJ. Scale bar 5 µm.
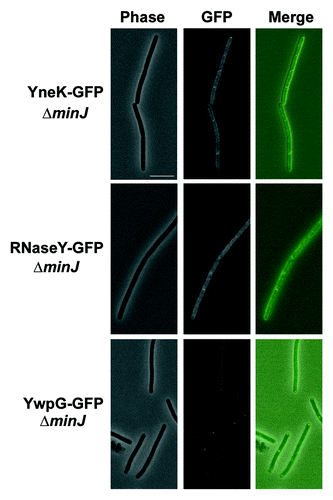
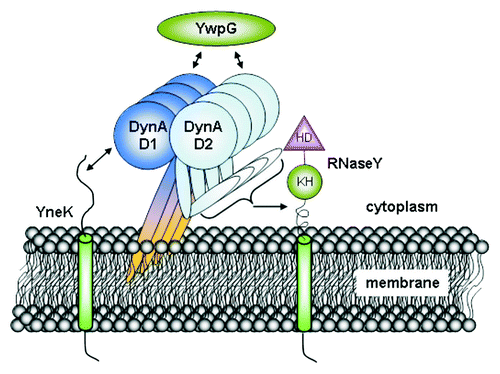
Figure 4. Interactome of DynA. B. subtilis DynA interacts specifically with YneK, YwpG and RNaseY. The YneK interaction is mediated by the D1 subdomain, while YwpG interacts with D1 and D2 domains. Interaction of RNaseY likely needs full length DynA.