Figures & data
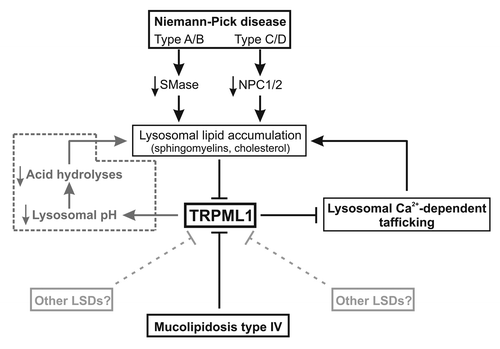
Figure 1. Comprehensive signaling events mediating lysosomal storage diseases. Under normal condition, lysosomal Ca2+ release through TRPML1 channel into the cytosol supports a transient Ca2+ elevation essential for lysosomal exocytosis and lipid trafficking. Under pathological conditions, lysosomal lipid accumulation (sphingomyelins, cholesterol) caused by alteration in sphingomyelinase (SMase) and NPCs activity (i.e., Niemann-Pick disease) inhibits TRPML1 channel activity and therefore lysosomal exocytosis and trafficking that in turn cause a secondary lysosomal storage. Similarly, direct alteration of TRPML1 activity (i.e., mucolipidosis type IV) results in the same lysosomal defects. Although H+ permeability of TRPML1 channel is still controversial, alteration in TRPML1 activity could also cause lysosomal H+ accumulation and pH acidification, decreasing acid hydrolases activity and causing lysosomal storage (pathway in dotted line).