Figures & data
Figure 1. Heterochromatin redistribution during aging. Aging is associated with a loss of constitutive heterochromatin, as demonstrated by a decrease in H3K9 methylation and delocalization of HP1 proteins. Concurrently, increases in facultative heterochromatin occur at specific loci, particularly at Senescence-Associated Heterochromatin Foci (SAHFs) that depend on the activity of H3K9 methyltransferases, on recruitment of HP1 proteins and histone chaperones, and on the presence of macro H2A. The decrease in activity of the maintenance DNA methyltransferase Dnmt1 and the increase in de novo Dnmt3 methyltransferases during aging may also contribute to heterochromatin redistribution.
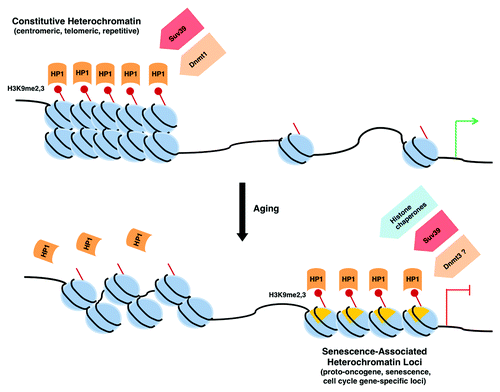
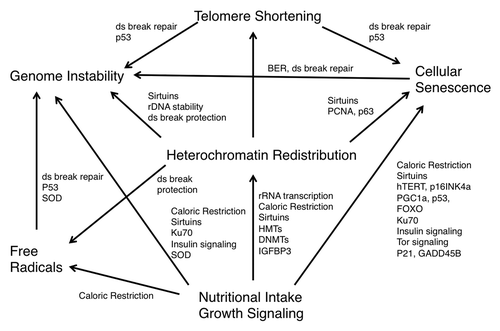
Figure 2. Epigenetic changes resulting in a decrease and redistribution of global heterochromatin may underlie the various models of aging. The models of free radical accumulation, genetically programmed senescence, telomere shortening, genomic instability, nutritional intake and growth signaling are distinct, yet overlapping theories of aging that may all be linked by heterochromatin redistribution.