Figures & data
Figure 1. Histone post-translational modifications and variants. (A) Schematic drawing of a nucleosome with the four canonical histones (H2A, H2B, H3 and H4) and the linker histone H1. The covalent PTMs [methylation (Me), acetylation (Ac), ubiquitination (Ub), and phosphorylation (Ph)] are highlighted on the N- and C-terminal tails of each histone. (B) Graphical representation of histone PTM writers, which add covalent PTMs to histone tails, readers, which recognize and bind histone PTMs, and erasers, which remove histone PTMs. Protein families associated with these steps are listed on the left. In this example, the PRC2 complex adds (writes) tri-methylation on lysine 27 of histone H3. This is then recognized by the reader complex PRC1. The RING1A and RING1B subunits of PRC1 (writers) subsequently act to ubiquitinate lysine 199 of histone H2A. UTX-1 and JMJD3 can act to remove (erase) histone H3 K27 tri-methylation. (C) The known histone variants in ESCs are represented next to the canonical histones they replace.
![Figure 1. Histone post-translational modifications and variants. (A) Schematic drawing of a nucleosome with the four canonical histones (H2A, H2B, H3 and H4) and the linker histone H1. The covalent PTMs [methylation (Me), acetylation (Ac), ubiquitination (Ub), and phosphorylation (Ph)] are highlighted on the N- and C-terminal tails of each histone. (B) Graphical representation of histone PTM writers, which add covalent PTMs to histone tails, readers, which recognize and bind histone PTMs, and erasers, which remove histone PTMs. Protein families associated with these steps are listed on the left. In this example, the PRC2 complex adds (writes) tri-methylation on lysine 27 of histone H3. This is then recognized by the reader complex PRC1. The RING1A and RING1B subunits of PRC1 (writers) subsequently act to ubiquitinate lysine 199 of histone H2A. UTX-1 and JMJD3 can act to remove (erase) histone H3 K27 tri-methylation. (C) The known histone variants in ESCs are represented next to the canonical histones they replace.](/cms/asset/36d7b0a0-0258-41da-b9bb-266f1e430ea6/kepi_a_10921141_f0001.gif)
Figure 2. Pathways of DNA methylation and demethylation. (A) Passive DNA demethylation. DNA in a cell is 5′ methylated at CpG islands in a symmetrical fashion by DNA methyl transferases (DNMTs). If there is a loss of DNMT function, followed by DNA replication and cell division, cells containing asymmetric DNA methylation will arise. If there is continued loss of DNMT function and further DNA replication and cell division, this will give rise to cells with unmethylated DNA. This action is passive, as it relies on DNA replication and cellular division. (B) Active DNA methylation. Several pathways can lead to active demethylation of DNA without the need for DNA replication, and in the presence of DNMTs. Hydroxymethylation (1) of 5-methylcytosines (5-mC) is performed by TET proteins. 5-hydroxymethylcytosine (5-hmC) may act as a substrate for further modifications, or may itself be sufficient to prevent factors that interact with methyl-cytosine from having an effect. 5-hmC can be further oxidized by TET proteins to 5-formylcytosine (5fC) and 5-carbonylcytosine (5caC).Citation54 5caC is a substrate for TDG glycosylase, creating an abasic site through base excision, which is then repaired with an unmethylated cytosine.Citation54,Citation55 Both 5-mC and 5-hmC can act as substrates for deaminases (2). The deaminases AID/APOBEC can convert 5-hmC to 5-hydroxymethyl-uracil (5hmU).Citation57 This is then repaired by mismatch-repair pathways, beginning with base excision by the glycosylase UDG. AID may also be able to convert 5-mC to thymine, which is repaired beginning with the glycosylase MDB4. The presence of a bona fide DNA demethylase (3) is a controversial topic, but several groups have proposed candidates.Citation35
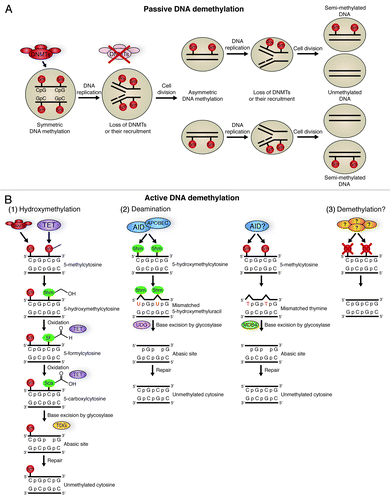
Figure 3. – Chromatin states. (A) Open, active euchromatin is characterized by histone H3 lysine 4 methylation (H3K4Me3) and lysine 9 acetylation (H3K9Ac). (B) Silent, repressive heterochromatin is characterized by methylation of histone H3 lysine 9 (H3K9Me3) and lysine 27 (H3K27Me3). Heterochromatic DNA is more densely packed than euchromatin, facilitated by heterochromatin protein 1 (HP1). (C) Regions of the genome have been identified with both activating (H3K4me3) and repressive (H3K27Me3) histone marks; termed bivalent chromatin. It is possible that these PTMs occur on neighboring nucleosomes, however, it is also possible that one allele of the gene is associated with heterochromatin, and the other euchromatin. This would give the appearance of bivalent chromatin.
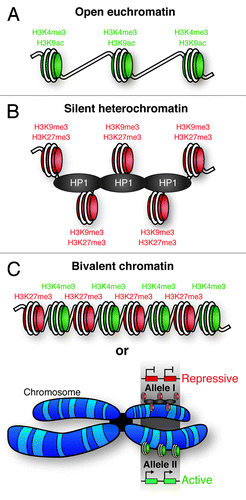
Figure 4. Mechanisms of SWI/SNF-mediated chromatin remodeling. (A) SWI/SNF binding to chromatin disrupts histone-DNA contacts. This allows for the creation of a loop of DNA that propagates around the nucleosome, ultimately resulting in repositioning of the DNA with respect to the nucleosome (sliding). This sliding can lead to eviction of adjacent nucleosomes. DNA looping also facilitates histone replacement with histone variants. (B) BRG1 containing mSWI/SNF in ES cells (esBAF) plays vital roles in the pathways promoting ES cell stemness and pluripotency. esBAF enforces repressive H3K27me3 marks deposited by the PRC2 complex at many lineage specific genes that would otherwise promote differentiation. In contrast, esBAF antagonizes PRC2 action at LIF targets, such as core pluripotency circuitry genes. By creating more accessible chromatin at these locations, esBAF prepares the chromatin environment for the cooperative action of phospho-STAT3 and master pluripotency regulators, such as OCT3/4 and SOX2.
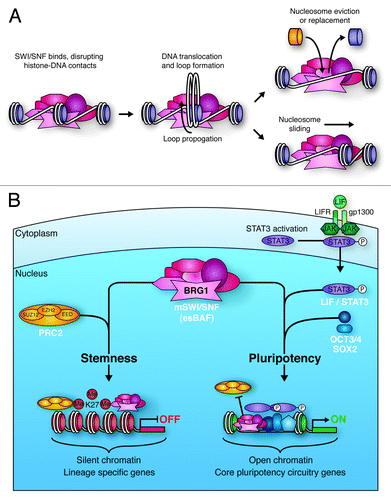
Table 1. Roles of mSWI/SNF complex components in mammalian development
Figure 5. miRNA biogenesis and function in ES cell differentiation. (A) Micro RNA biogenesis. Primary transcripts (pri-miRNAs) are generated through RNA pol II or pol III transcription, which then undergo RNase III cleavage, mediated by the DROSHA/DGCR8 complex, to generate ~70 nt pre-miRNAs. XPO5 exports these pre-miRNAs to the cytoplasm, where they are further cleaved by DICER to generate mature double-stranded RNA duplexes. One strand of these duplexes is then bound by one of four Argonaute proteins (AGO 1–4) to form active RISC complexes, which can modulate gene expression through translational inhibition, or mRNA deadenylation. If the miRNA is perfectly matched to the target sequence, endonucleolytic cleavage of the mRNA transcript can occur through the ‘slicer’ activity of AGO2.Citation139,Citation145 (B) miRNAs play roles in the maintenance of pluripotency (left section), the onset of differentiation (middle section), and the maintenance of terminal differentiation (right section). In order to maintain pluripotency, miRNAs act to promote maintenance of cell cycle progression (CDKN1A, RBL2 and LATS2 inhibition) and de novo DNA methylation (RBL2 inhibition), and suppress factors that promote differentiation (Let-7 inhibition). In order to promote differentiation, miRNAs act to block self-renewal and core pluripotency factor production. miRNAs maintain terminal differentiation by suppressing gene expression associated with other lineages (miR-1 and miR-133), block self-renewal (miR-124) and maintain the state of the specific lineage (miR-9/9*).
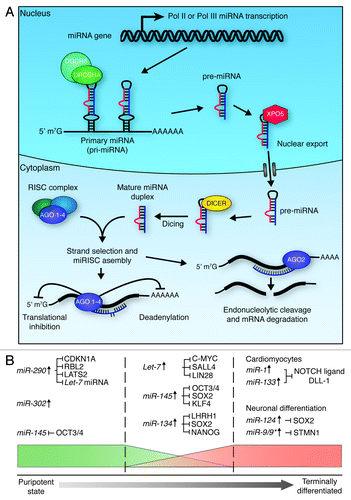
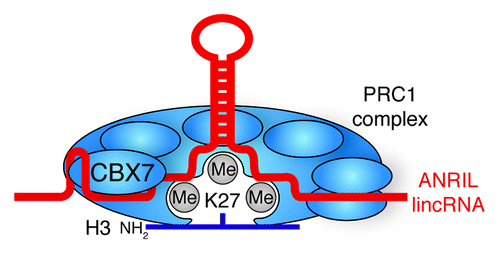
Figure 6. lincRNA stabilization of Polycomb Group complexes. ANRIL lincRNA bind and stabilizes the interaction of PRC1 with H3K27me3 through the CBX7 subunit.