Figures & data
Table 1. The effect of gene methylation in MDS
Table 2. Histone modification in MDS
Table 3. Downregulated microRNAs in MDS patients
Table 4. Upregulated microRNAs in MDS
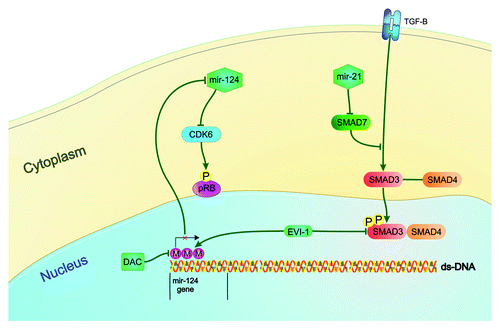
Figure 1. Epigenetic alteration of microRNAs and MDS pathogenesis Mir-124, which is hypermethylated and under-expressed in MDS patients, participates in MDS pathogenesis. It down-regulates the cyclin dependent kinase 6 (CDK6), which in turn phosphorylates and inactivates the tumor-suppressor protein Rb. EVI1 represses miR-124 expression by promoter methylation. It also down-regulates the expression of SMAD3, a member of TGF-β pathway that regulates cell differentiation, proliferation and apoptosis. Mir-124 activity can be restored by the treatment with the DNA methylation inhibitor, Decitabine. The TGF-β pathway is also regulated by mir-21 which down-regulates the expression of SMAD7, which in turn is a negative regulator of TGF-β receptor.