Figures & data
Figure 1. Study profile. MMRVnew WS: measles-mumps-rubella-varicella vaccine derived from newly established working seed virus stock. MMRV, combined measles-mumps-rubella-varicella vaccine derived from current working seed virus stock; ATP, according-to-protocol.
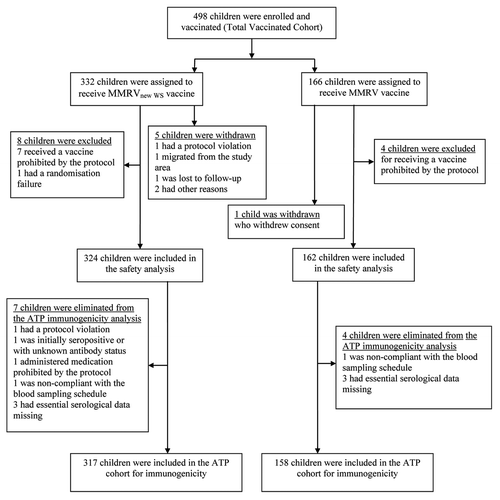
Table 1. Seroconversion rates and antibody GMTs in the MMRVnew WS and MMRV groups, 43 d after each dose (ATP cohort)
Table 2. Incidence of solicited local symptoms reported during the 4-d follow-up period in groups MMRVnew WS and MMRV (Total vaccinated cohort)
Table 3. Incidence of solicited general symptoms reported during the 43-d follow-up period in groups MMRVnew WS and MMRV (total vaccinated cohort)
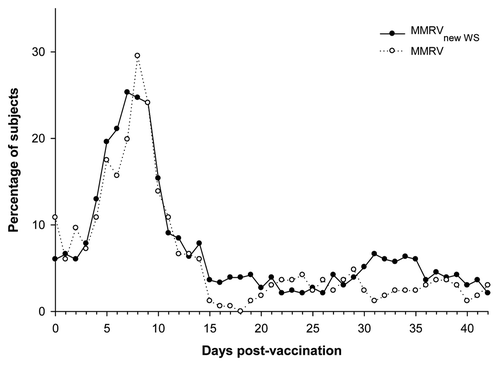
Figure 2. Prevalence of fever (any intensity) during the 43-d follow-up period after the first dose (Total vaccinated cohort). MMRVnew WS: measles-mumps-rubella-varicella vaccine derived from newly established working seed virus stock. MMRV, combined measles-mumps-rubella-varicella vaccine derived from current working seed virus stock