Figures & data
Table 1. Adverse reactions registered in the 14 d after immunization
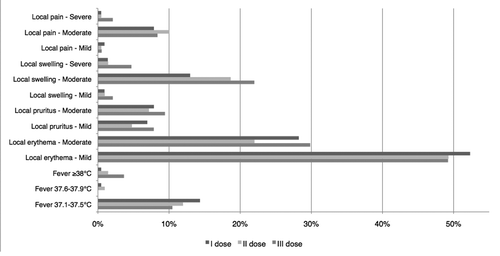
Table 2. Adverse reactions registered in the 14 d following the administration of each dose
Table 3. Assessment of difference in tolerability after each dose. Significant values (p < 0.05) are indicated in bold type
Table 4. Adverse reactions registered in the 14 d following the administration of each dose in the subset of 186 subjects who received all 3 doses of vaccine and completed and returned all three diary cards
Table 5. Assessment of difference in tolerability after each dose in the subset of 186 subjects who received all 3 doses of vaccine and completed and returned all three diary cards. Significant values (p < 0.05) are indicated in bold type
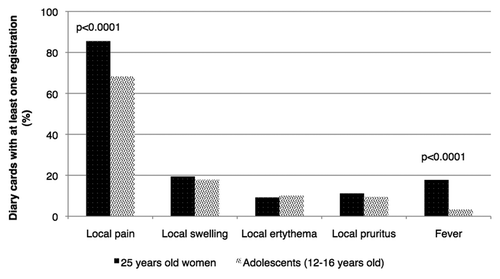
Figure 2. Comparison between our findings and those previously published with regards to a sample of adolescent girls aged 12–16.Citation13