Figures & data
Figure 1. Nomenclature and localization of the major external and internal protein antigens of the influenza virus.
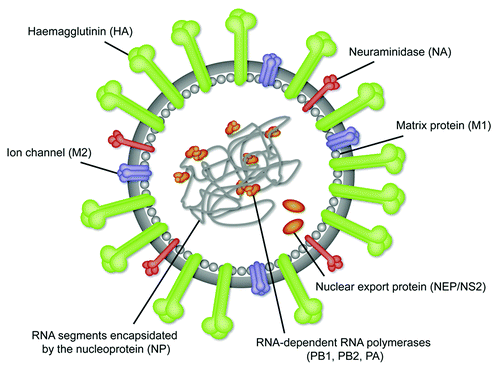
Table 1. Approved seasonal and (pre)pandemic influenza vaccines
Table 2. Novel influenza vaccine approaches in clinical development categorized according to the primary mode of protection intended to be induced, vaccine type, and antigen targets
Table 3. Established and emerging COPs for influenza vaccine development
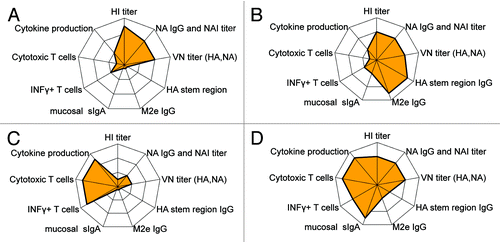
Figure 2. Connectograms showing both type and strength of a spectrum of immune responses induced by principally different influenza vaccine concepts: (A) Classical split or subunit vaccines mostly inducing vaccine specific HA and NA antibodies, (B) Vaccines specifically targeting a more broad antibody response with potential for cross-protection, (C) Vaccines specifically targeting conserved antigens for cross-reactive T cell responses, and (D) LAIV covering a broad range of both humoral and cellular immune responses.