Figures & data
Figure 1 Chromatographic profiles obtained from a (A) IEC shown in full scale and (B) SEC shown in expanded scale for all charge variant fractions, starting material and buffer blanks. See for numerical values obtained from these analyses.
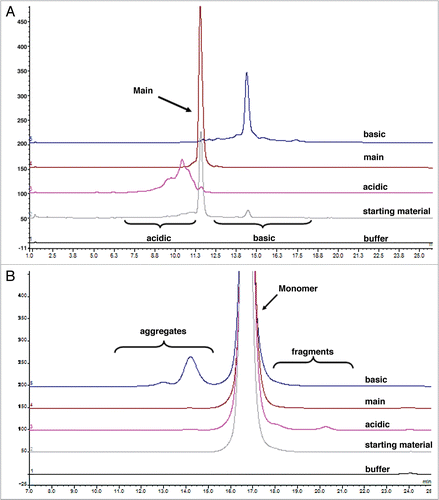
Figure 2 Representative sensorgrams of fitted data from the kinetic analysis of all charge variant fractions (A) acidic, (B) main, (C) basic and (D) starting material to immobilized rat FcRn. Black lines are fitted curves using a bivalent binding model. All sample concentrations (from bottom to top) are 62.5, 125, 250, 500, 1,000, 2,000 and 4,000 nM. Residual plot shows the difference between experimental and fitted data for every point in the sensorgram. Also see for clarity.
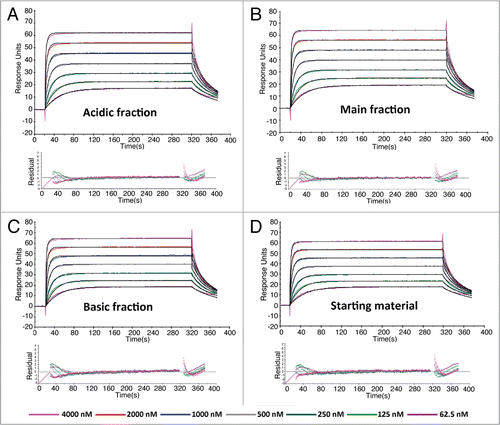
Figure 3 Linear changes in serum concentrations versus time for all charge variant fractions following single (A) intraveneous or (B) subcutaneous administration of 10 mg/kg in normal rats. The assay minimum quantifiable concentration was 0.4 µg/mL in rat serum. Results are mean ± SD (n = 12 rats/time point).
Table 1 Major chemical degradation pathways which are a common source of charge-related heterogeneity of therapeutic IgG1 mAbs
Table 2 Analytical results to assess all mAb charge variant fractions
Table 3 Analytical characterization of isolated charge variant fractions
Table 4 Comparison of all charge variant fractions to evaluate their kinetics of binding to rat FcRn, binding responses (in response unit) and their in vitro binding specific activity
Table 5 Pharmacokinetic parameters of all mAb fractions in normal rats