Figures & data
Figure 1. Vaccination with Lm-LLO-E7 induces the expression of chemokines by TC-1 tumors. (A) cDNA from three mice that were treated twice with either Dulbecco’s PBS (DPBS) or Lm-LLO-E7 bacteria were pooled and applied to chemokine PCR arrays. Data are reported as fold changes in the expression of the indicated mRNA species in TC-1 tumors isolated from Lm-LLO-E7-treated mice relative to DPBS-treated animals. Data are from a single experiment conducted once. (B and C) Conventional quantitative RT-PCR assays were conducted on tumor RNA samples (n = 3) (B) and tumor lysates were analyzed using chemokine ELISAs in order to confirm the presence of vaccine-induced TH1 chemokines (CXCL9 and CXCL10, n = 8, pooled from 2 experiments; CCL4 and CCL5, n = 3) (C). Mean RQ (B) or protein concentration values (C) ± SEM are reported (*P≤ 0.05, ** P ≤ 0.01, ***P < 0.001). Conventional quantitative RT-PCR primer assays and ELISAs were conducted twice with similar results.
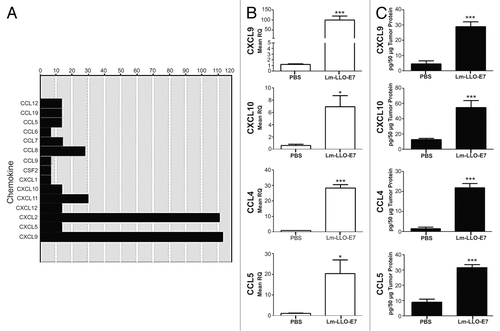
Figure 2. CD4+ and CD8+ T cells express CXCR3. (A and B) Lymph node (LN) cells were isolated from 3–5 tumor bearing mice that were vaccinated with live Lm-LLO-E7 bacteria, pooled and labeled with a panel of antibodies to identify multiple T-subsets. Labeled cells were analyzed by flow cytometry for the expression of CXCR3 and CCR5. Representative density plots are shown. CXCR3+ (A) and CCR5+ (B) T cells were identified among live, single TCRβ+ cells.
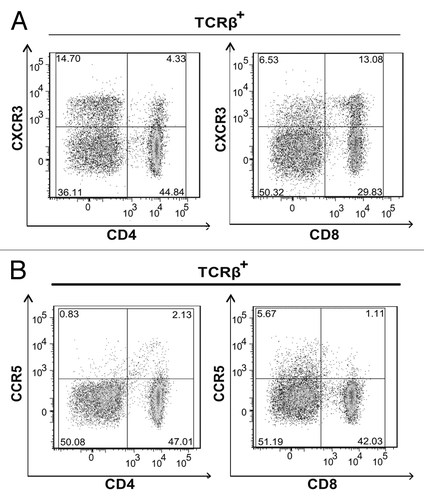
Figure 3. Vaccine-induced chemokine expression is affected by anti-interferon γ antibody administration. TC-1 tumor-bearing mice (n = 3-5 mice per group) were treated with anti-interferon γ (IFNγ) or IgG control antibodies and vaccinated with Lm-LLO-E7 bacteria. CCL4, CCL5, CXCL9 and CXCL10 expression levels were quantified by quantitative RT-PCR or ELISA, as indicated. Mean RQ or protein concentration values ± SEM are reported (*p ≤ 0.05). These experiments were conducted twice with similar results.
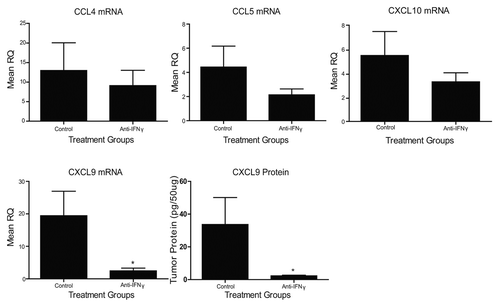
Figure 4. Interferon γ induces the expression of chemokines by - cells. (A–C) Samples from TC-1 cells that were maintained under control conditions or cultured with tumor necrosis factor α (TNFα) and interferon γ (IFNγ), were collected and then processed for chemokine PCR arrays (A), CXCL9-specific quantitative RT-PCR assays (B) or CXCL9-specific ELISAs (C). In (A and B) data are reported as fold changes in mRNA expression relative to cells maintained in control conditions. Mean RQ or protein concentration values ± SEM (of 2 independent experiments) are reported in (B and C) respectively. nd, not detected.
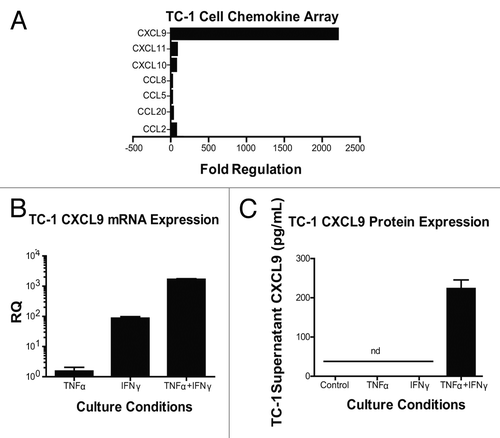
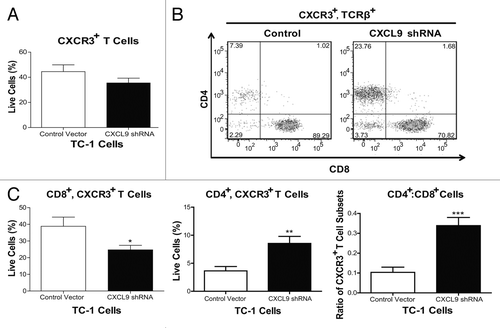
Figure 5. Knockdown of CXCL9 in TC-1 cells alters T-cell subset distribution in the tumor plugs of vaccinated mice. (A–C) TC-1 cells bearing a control plasmid or a shRNA plasmid for the downregulation of CXCL9 were co-injected with basement membrane extract (BME) and allowed to form plugs for 7 d. On day 7, mice were vaccinated with Lm-LLO-E7 bacteria and, 7 d later, tumor plugs were removed and processed for the cytofluorometric measurement of the percentages of CXCR3+TCRβ+ as well as CD8+CXCR3+TCRβ+ and CD4+CXCR3+TCRβ+ T lymphocytes (among live single cells). The percentage of CXCR3+TCRβ+ cells is reported in panel (A) whereas representative density plots of the relative abundance of CD8+CXCR3+TCRβ+ and CD4+CXCR3+TCRβ+ T lymphocytes is depicted in (B) In (C) quantitative data on the percentages of CD8+CXCR3+TCRβ+ and CD4+CXCR3+TCRβ+ T cells as well as on their ratio are reported. In (A and C) columns represent mean values ± SEM of pooled data (n = 7) from two independent experiments (2–5 mice per group) (*P ≤ 0.05, **P ≤ 0.01, ***P < 0.001).