Figures & data
Figure 1. CD117 positive/hematopoietic lineage negative cells in the amniotic fluid of mice and human along the course of gestation. (A) Percentage of mouse AFS cells (mAFSC) as a function of gestational age. (B) Total number of mouse AFS cells (mAFSC) per embryo equivalent (EE) at different gestational ages. (C) Percentage of human AFS cells (hAFSC) as a function of gestational age. Means are represented by bars. Adapted from reference Citation37.
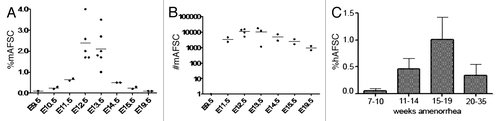
Figure 2. (A) Human AFS cells mainly display a spindle-shaped morphology during in vitro cultivation under feeder layer-free, serum-rich conditions. (B and C) Clonal human AFS cell lines retain long telomeres and a normal karyotype after more than 250 cell divisions. (B) Conserved telomere length of AFS cells between early passage (20 population doublings, lane 3) and late passage (250 population doublings, lane 4). Short length (lane 1) and high length (lane 2) telomere standards provided in the assay kit. (C) Giemsa band karyogram showing chromosomes of late passage (250 population doublings) cells. Adapted from reference Citation35.
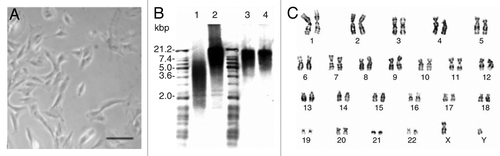
Table 1. Surface markers expressed by human AFS cells: results by different groups
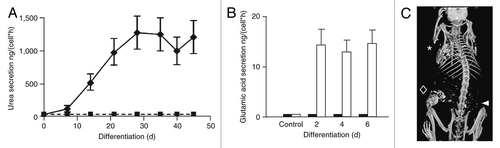
Figure 3. AFS cells differentiation. (A) Hepatogenic: urea secretion by human AFS cells before (rectangles) and after (diamonds) hepatogenic differentiation in vitro. (B) Neurogenic: secretion of neurotransmitter glutamic acid in response to potassium. (C) Osteogenic: mouse micro-CT scan 18 weeks after implantation of printed constructs of engineered bone from human AFS cells; arrow head: region of implantation of control scaffold without AFS cells; rhombus: scaffolds seeded with AFS cells. Adapted from reference Citation35.