Figures & data
Figure 1 Injection of human mesenchymal stem cells (hMSCs) into growing rat embryo at the sit of nephrogenesis. Using mouth pipette, hMSCs were injected into the intermediate mesoderm between the somite and the lateral plate at the level of somite 29, which we previously estimated by in situ hybridization for c-ret, to be the ureteric budding site. Asterisk indicates chorioallantoic placenta and arrow indicates yolk sac.
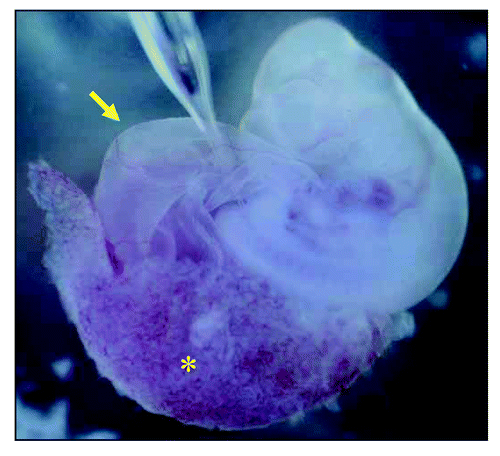
Figure 2 Appearance of the incubator for whole embryos. This system can rotate 15-ml culture bottles to achieve a continuous oxygen flow over the entire culturing period. The optimal gas-exchange schedules for rat embryo was determined previously.Citation76

Figure 3 Generation of vascularized neo-kidney from hMSCs. LacZ-positive hMSCs were differentiated in vivo using the modified relay culture system (A). Yellow dotted lines indicate the outline of the developed kidney in the omentum (top). These were dissected out and compared with native kidney (bottom). The resulting neo-kidney was subjected to X-gal assay to trace the transplanted hMSCs (B, top). The morphology of these LacZ-positive cells (shown under high magnification, bottom) and the renal structures to which they contributed was consistent with their being glomerular cells (left) and tubular cells (right). High magnification imaging revealed that several vessels from the omentum were integrated into the no-kidney (C) and electron microscopic analysis showed that the glomerular vasculature contained red blood cells (arrows), which might have derived from the host circulation (D). Reproduced with permission.Citation77
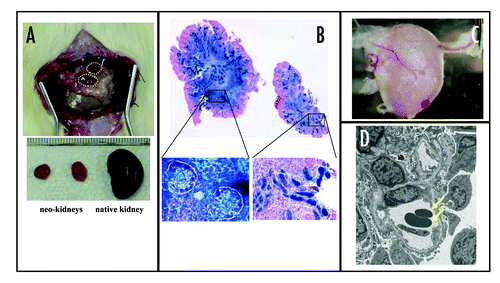
Table 1 List of minimum requirements for regenerated artificial kidney for chronic renal failure