Figures & data
Figure 1 Capacitance measurement of cell concentrations in tissue constructs. (A) Capacitance at 0.4 MHz of fibroblasts cells at different concentrations in a fibrin gel, showing a linear relation between capacitance and cell concentration. (B) Constantly monitoring the capacitance at 0.4 MHz allows the cell concentration in the artificial tissue to be followed in time. The gel was inoculated with 100.000 human fibroblast cells/ml in DMEM growth medium with 10% fetal calf serum. Capacitance measurements were performed with an Aber Instruments model 220 Biomass Monitor (Aber Instruments, Aberystwyth, UK).
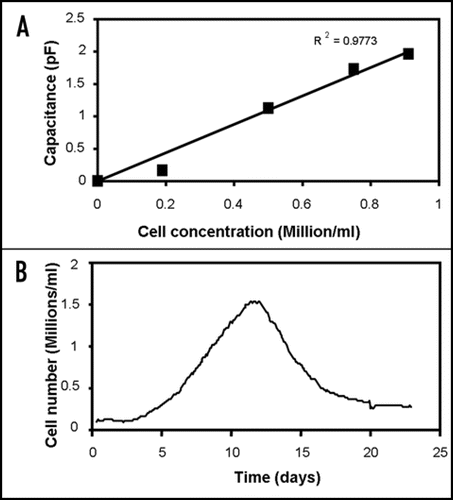
Figure 2 Electro-orientation of (fission) yeast cells. The electrodes used had a spacing and width of 40 µm; the applied voltage was 10 V peak-to-peak. (A) Orientation along electric field lines at 1 MHz; (B) Orientation perpendicular to electric field at 50 MHz.

Figure 3 Outline of the principle of an electro-rotation setup, with the phases of the electric field signal applied to each electrode. The electro-rotation method can be used for orienting polarized cells in any desired direction.
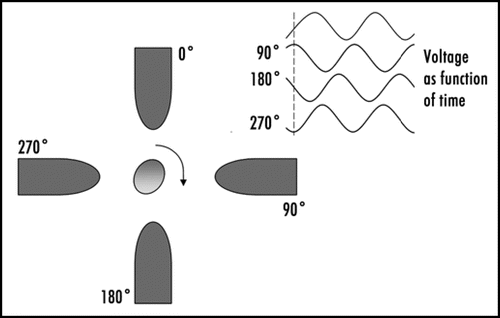
Figure 4 Principle of dielectrophoresis. The dashed lines represent electric field lines. A dipole is induced by the electric field in the particle, and a force is exerted on the induced charges on both sides of the particle. Because the electric field strength is stronger near the smaller electrode on the right than near the larger electrode on the left the particle will experience a net force, pulling the particle towards the smaller electrode. If the charges on the electrodes are reversed the distribution of charges in the dipole will also reverse; dielectrophoresis will therefore occur in both AC and DC electric fields. If the surrounding medium is more polarisable than the particle the net force will be in the opposite direction (negative dielectrophoresis rather than the positive dielectrophoresis depicted in ).
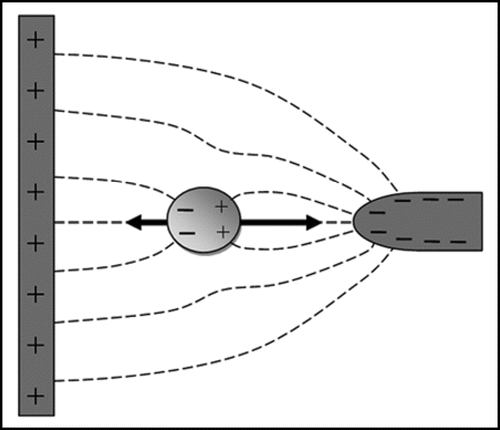
Figure 5 Artificial haematopoietic stem cell microniches created using dielectrophoresis. (A) Top view; (B) side view; (C) actual aggregate. The aggregate shown is made from successive layers of osteoblast (bottom), stromal (middle) and Jurkat (top) cells, attracted between interdigitated oppositely castellated electrodes. The aggregates, which are approximately 500 µm across, mimic the osteoblast haematopoietic stem cell niche in bone marrow. Vascular haematopoietic stem cell niches, which include endothelial cells, can be created using a similar approach.Citation51
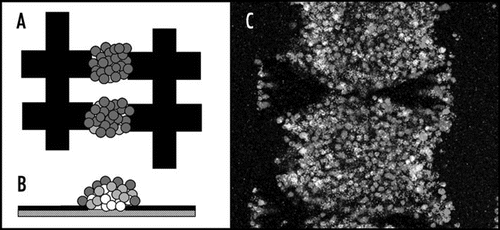
Figure 6 Textiles can be created which can be used for constructing cellular arrays using dielectrophoresis on large scale.Citation55
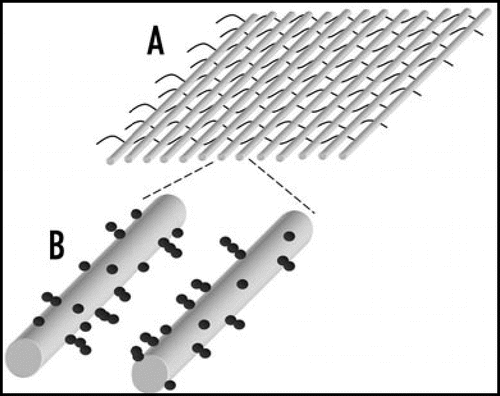
Table 1 Comparison of different physical force field-based techniques for cell manipulation