Figures & data
Figure 1 The molecular cascade that leads to the formation of amyloid fibrils. The formation of amyloid fibrils is a sequential process that proceeds from monomeric species into well-ordered amyloid fibrils. It is still unclear whether the formation of soluble oligomers such as the Aβ*56Citation17 and the annular amyloid pores,Citation8 is an on-pathway or an off-pathway process.
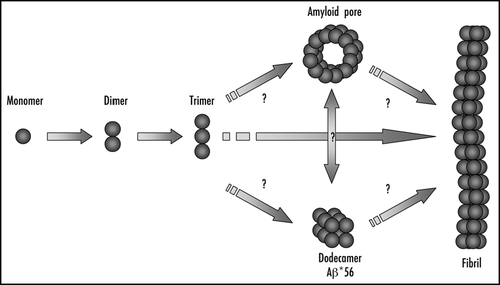
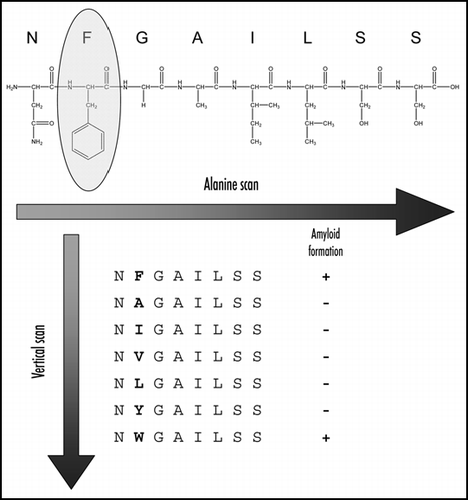
Figure 2 Systematic study of amyloid formation. Alanine scan was first used to identify the phenylalanine as the most important residue in amyloid formation by this fragment. This was followed by vertical scan in which the role of aromatic as compared to aliphatic residues was revealed.