Figures & data
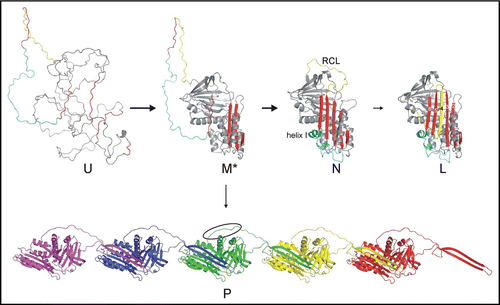
Figure 1 Serpin folding and polymerization. The pathway of serpin folding proceeds from the unfolded state (U) to the native state (N) via a stable intermediate (M*). The native conformation is the only active state, and is composed of a five-stranded A sheet (red) and a 20 residue reactive centre loop (RCL, yellow). Serpin inhibitory function requires the native conformation to be a kinetically trapped metastable state. Completion of sheet A by incorporation of the RCL as strand 4, to form the latent (L) state, results in the doubling of the serpin's thermodynamic stability (the six strands are labelled on L). Folding and unfolding of native serpins is known to proceed via a stable intermediate denoted M*, which also corresponds to the polymerogenic form.Citation24–Citation26 The key feature of the M* state is that strand 5 is not yet incorporated into sheet A, and can thus insert in an intermolecular fashion to form off-pathway polymers (P, each protomer of the pentamer is in a different colour). The polymers have complete A sheets and are thus hyperstable. As a consequence of polymerization, the linker region (cyan), containing helix I, remains unfolded. We hypothesize that the hydrophobic linker (indicated by the oval) is responsible for the lateral association of polymers into insoluble aggregates.