Figures & data
Figure 1. Confocal fluorescence microscopic analysis of iodine stained starch in leaves. Green fluorescence emission of iodine-stained starch granules occurring in chloroplasts of WT, aps1, ss3/ss4, gbss and sus1/sus2/sus3/sus4. Plants were grown in pots at ambient CO2 (350 ppm) at 20°C under a 16 h light (90 μmol photons sec–1 m−2) / 8 h dark regime. Leaves were harvested at the end of the light period, fixed, stained as described in the main text, and examined using a D-Eclipse C1 confocal microscope with Ar 488 nm excitation using BA515/30 filter (detector gain setting 7.2). Note that green fluorescence associated only with oval/round structures that were identified as starch granules. Bar = 5 µm.
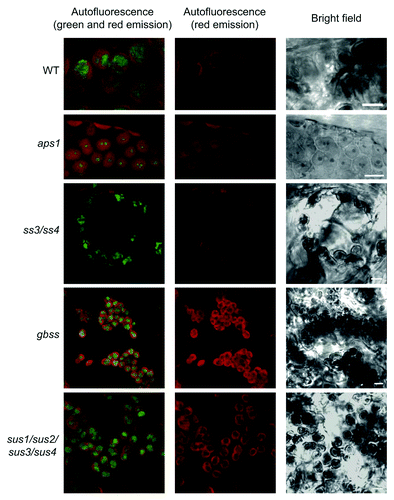
Figure 2. Confocal fluorescence microscopic observation of iodine stained glycogen granules in E. coli. E. coli cells were cultured in M9 minimal medium supplemented with glucose, harvested at the end of the exponential growth and stained as described in the main text. Note the presence of green fluorescence dots in the poles of WT cells, but not in the glycogen-less ΔglgBXCAP cells lacking the whole glycogen biosynthetic machinery. Bar = 2.5 µm
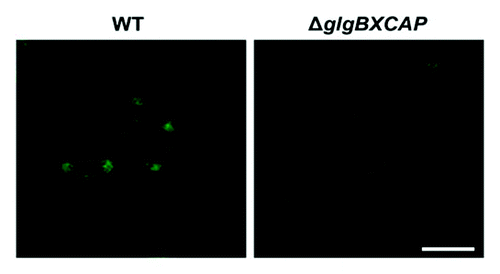
Figure 3. Confocal fluorescence microscopic observation of iodine stained pure potato starch, amylopectin and amylose. Pure, commercially available potato starch (Roche), amylose (Sigma A00512) and amylopectin (Sigma A8515) were stained with iodine solution, rinsed gently in distilled water for about 1 min, mounted on microscopic slides, and examined using a D-Eclipse C1 confocal microscope with Ar 488 nm excitation using BA515/30 filter (detector gain setting 7.2–7.5). Bar = 10 µm for starch granules and 20 µm for amylose and amylopectin.
