Figures & data
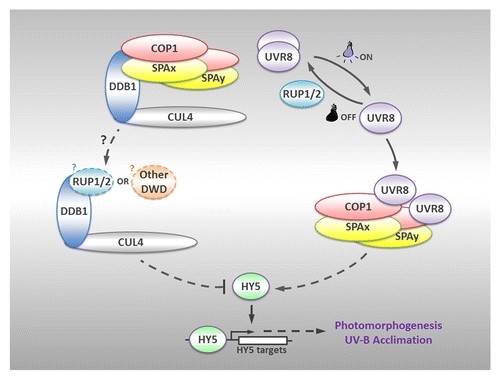
Figure 1. A model for the organization of protein complexes under photomorphogenic UV-B. Upon UV-B irradiation, UVR8 switches from dimer to monomer, and directly interacts with COP1 to form UVR8-COP1-SPA complexes, which promote the stability and activity of HY5. HY5 positively regulates downstream transcriptional responses so as to facilitate UV-B-induced photomorphogenesis and acclimation. On the other hand, the CUL4-DDB1 E3 apparatus, which releases the COP1-SPA core complexes sequestered by UVR8 monomers, might recruit alternative DWD proteins such as RUP1/RUP2 to repress UV-B-induced photomorphogenesis. Once the UV-B irradiation is removed, RUP1 and RUP2 mediate the redimerization of UVR8. Solid lines indicate direct effects, while dotted lines represent indirect regulation. Question marks denote the hypothesis, which awaits further investigation.