Figures & data
Figure 1 NtWIF in the MAPK cascade. External signals such as wounding/pathogen and developmental stimulus activate the WIPK phosphorylation cascade. The initial component(s) that trigger this cascade are proteins belonging to the MEKK family, although their identity in this cascade remains to be determined. The second component(s) are MEKs, among which NtMEK2 was shown to directly phosphorylate WIPK. Activated WIPK then phosphorylates NtWIF, which recognizes the ARE motifs and transcriptionally activates diverse genes involved in defense response and developmental processes. When external signals are initiated from wound/pathogen stresses, the system mainly functions to activate defense-related genes (left side), and when siganls are related to developmental process, the system activates development-related genes (right side). However, since many genes are bifunctional, being transcriptionally activated during stress response and development, such a differential response my not be distinct. This cascade flexibly plays a critical role in a wide range of physiological responses.
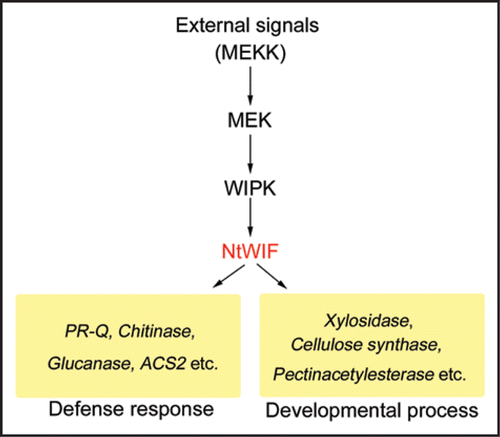
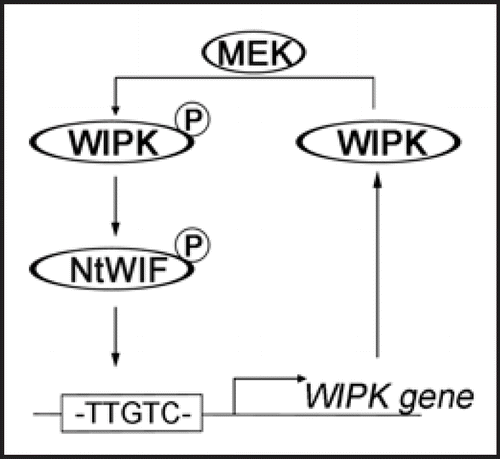
Figure 2 Self-amplification of WIPK protein. The 1.2-kb promoter region of WIPK gene contains one ARE motif, TTGTCT, which is recognized by NtWIF to induce WIPK transcription. Newly synthesized WIPK protein enters WIPK cascade, and is phosphorylated by upstream MEK(s). Resulting WIPK then phosphorylates and activates NtWIF, which again induces WIPK transcripts. This circuit helps a self-amplification of WIPK, thereby supplying enough amounts of WIPK proteins as far as up-steam signals through MEK(s) are available. Declining MEK activity may automatically shut-down this circuit. Hence, the MAPK cascade is auto-regulated through a feed-back system.