Abstract
Objective
This study aimed to check the sensitivity of multiple newly developed 3T MRI breast sequences using CAD software, in pre-sampling diagnosis of breast cancer, in an attempt to minimize unnecessary invasive sampling or surgical procedures.
Patients and methods
This was a prospective study, included 120 female patients, presented with debatable or malignancy suspected mammo-sonographic results. The study protocol was approved by the ethics committee in Al-Mana General Hospital.
Results
36 patients’ tumors were reported as benign and 84 were reported as malignant. Biopsy approved 33 tumors as benign and 87 as malignant tumors. These results gave indices of 93.1% sensitivity and 90.9% specificity.
Conclusion
3T MRI breast with CAD is very sensitive imaging tool, that can help to avoid unnecessary invasive procedures.
Abbreviations:
- AGH
- Almana General Hospital
- BI-RADS
- Breast Imaging Reporting and Data System
- CAD
- computer aided diagnosis
- CC
- Cranio-caudal
- DCE
- dynamic contrast enhancement
- DWI
- diffusion-weighted images
- KSA
- Kingdom of Saudi Arabia
- T2w
- T2 weighted
- ML
- medio-lateral
- MIP
- maximum intensity projection
- PHT
- postmenopausal hormonal replacement therapy
- 3T
- 3 Tesla
1 Introduction
Breast cancer has a tremendous impact on women’s morbidity and mortality, worldwide. It is the most common cancer in females, as it accounts for about 22% of all malignant tumors in women. In Saudi Arabia, the incidence of breast cancer is about 19.9%. Average age of presentation is considerably variable from area to area; in the United States, 50% of breast cancer occurs in women over the age of 65 years, while in Middle East countries, including Saudi Arabia, it is significantly presented at earlier age groups; average below 52 years.Citation1,Citation2
It is the second leading cause of cancer deaths in KSA. This malignancy carries tremendous worldwide health care, socio-economic, emotional and public health implications. There are many risk factors classifying some women as high risk groups for developing such disease, some of these factors are avoidable and others are not.Citation1
1.1 Unavoidable risk factors
1.1.1 Gender and age
Breast cancer occurs nearly 100 times more often in women than in men. Risk of developing breast cancer increases for women that are getting older. About 1 out of 8 invasive breast cancers are found in women younger than 45, while about 2 of breast cancers out of 8 are found in women age 55 or older.Citation3,Citation4
1.1.2 Genetic risk factors and family history
5–10% of breast cancer cases are thought to be hereditary, due to an inherited chromosomal anomaly in the BRCA1 and BRCA2 genes. Breast cancer risk is higher among women with close relatives’ history of breast cancer. Having one first-degree relative with breast cancer approximately doubles a woman’s risk. Having 2 first-degree relatives increases her risk about 3-fold.Citation5,Citation6
1.1.3 Personal history and racial ethnicity
A woman with cancer in one breast has 3- to 4-folds increased risk of developing a new cancer in the other breast or in another part of the same breast. Overall, western women are slightly more likely to develop breast cancer than African women, but African women are more likely to die of this cancer. Asian, Hispanic, and Native-American women have a lower risk of developing and dying from breast cancer.Citation7–Citation9
1.1.4 Dense breast tissue and certain benign breast conditions
Breasts are made up of dense glandular and fibrous tissue and less fatty tissue, have a higher risk of breast cancer than women with less dense breasts. Unfortunately, malignant lesions can be easily missed in dense breast mammograms. A number of factors can affect breast density, such as age, menopausal status, the use of drugs (e.g. Post menopausal hormonal therapy), pregnancy, and genetics. Women with multiple fibroadenomas have an increased risk of breast cancer – about 1½ to 2 times. Also, sclerosing adenosis, papillomatosi and radial scars slightly increase the risk of developing breast cancer.Citation10–Citation12
1.2 Avoidable risk factors
1.2.1 Nonpregnant and nonlactating females
Women who had no children or who had their first child after age 30 have a slightly higher breast cancer risk. Pregnancy reduces a woman’s total number of lifetime menstrual cycles, which may be the reason for this effect. Also, some studies suggest that breast feeding may slightly lower breast cancer risk, especially if it is continued for 1½ to 2 years. One explanation for this possible effect may be that breastfeeding reduces a woman’s total number of lifetime menstrual cycles.Citation13
1.2.2 Birth control and postmenopausal hormonal replacement therapy (PHT)
Oral contraceptives have been proven to slightly increase the risk of breast cancer, than women who have never used them, especially if taken by BRCA1&2 carriers. Also PHT, although relieving many of postmenopausal complaints e.g. osteoporosis, it significantly increases the risk of getting breast cancer, as well as the chances of bad prognosis.Citation14
1.2.3 Alcohol
Compared with non-drinkers, women who have 2–5 drinks daily have about 1½ times the risk of women who do not drink alcohol. Excessive alcohol consumption is also known to increase the risk of developing several other types of cancer.Citation15
1.2.4 Breast imaging
Still mammography is the first and most commonly used imaging tool for initial breast evaluation. In 2000, the Dutch Institute for Health Care Improvement had established a mammographic imaging protocol called, Breast Imaging Reporting and Data System (BI-RADS) for breast imaging. This system was formatted to have standard items including, indication for the study, class of breast composition, all positive findings, type of calcification and associated changes e.g. skin thickening, and axillary lymph nodes, if present and changes from older studies. Thereafter, breast will be classified under one of 6 standard BI-RAD classes; ranging from normal breast (Type 1) to proven malignancy (type 6). Finally, suitable management is suggested according to the reported class, e.g. biopsy for grade 4.Citation16
Annual screening mammography for females is recommended, starting at age 40, for early discovery of malignant lesions. However, there is one great disadvantage of breast cancer mammographic screening, which is the false-positive result. This disadvantage may lead to unnecessary work out and sometimes invasive procedures, thus, not cost effective and provoking anxiety in women before malignancy could be ruled out. Regardless, there is significant improvement of the results, after advent of the digital mammography into practice, there is still false positive results in considerable percentage of patients. In an article published by Hobson K, in Wall Street Journal at October 2011, a wide USA statistic study detected that 60% of women following the schedule of annual mammograms were falsely recalled for additional imaging e.g. other mammographic views, or Ultrasound, or MRI, or biopsy procedures.Citation17
Regardless, MRI is very sensitive soft tissues imaging tool, its application in breast imaging is not frequent, especially in Middle East. Some institutes use MRI breast, only as preoperative imaging tool, in pathologically proven cases of cancer breast, in attempt for clear demonstration of lesions, and if there are another mammographically missed lesions that may change the plan of management. Also, it may be recommended in postoperative evaluation for residual or recurrent lesions or response to neoadjuvant therapy.Citation18 Due to its relative high cost, there is no many researches for the evaluation of the use of MRI as periodic screening tool, instead of mammography.
1.2.5 Objective
This study aimed to test the highest possible accuracy of breast MRI using combined standardized quantitative and qualitative analysis of imaging data and its diagnostic potential. This may have potential impact of reduction of unnecessary intervention procedures and patients’ stress for benign lesions with controversial mammographic findings, especially in high risk patients.
2 Patients and methods
2.1 Population of the study
In Almana General Hospital (AGH)-Eastern Province, Saudi Arabia- from June 2012 until March 2014, 120 patients were found to have either controversial or malignant looking breast lesions, through both mammographic and sonographic examinations i.e. consistent with BI-RAD 4 or 5 classes, respectively. After installation of 3T MRI, we had applied a new protocol for these suspected breast lesions, which includes MRI examination before biopsy procedure.
All patients had signed informed consent in compliance with the hospital review board. MRI was performed shortly after mammographic-ultrasound examination, dated at the first two weeks of menstrual cycle for premenopausal ladies.
2.2 Methods
All MRI examinations were done through Magnetom Verio 3T machine, Siemens AG. Wittelsbacherplatz 280333, Muenchen, Germany. Imaging was performed using a bilateral breast array dedicated surface coil. Contrast was given through power injector (MRI compatible Bracco Empower Injector- Siemens AG Henkestrasse 127 D-91052 Erlangen-Germany). The patients were imaged in prone position, with both breasts centralized in the breast coil. In an attempt to avoid aliasing artifacts, which may occur in large breasts, smooth compression with sponge pads was done. On the contrary, if breasts were too small, foam wedge supports were inserted inside the coil, in order to limit respiratory movement artifacts.Citation19
Our imaging protocols were consisted of axial STIR, coronal T2w and sagittal T1w sequences. Afterward, DWI of both breasts was performed in the axial plane using single-shot echo-planar sequence, with 3 different b values; 0, 800 and 1500 s/mm2, respectively 2°. Finally, dynamic contrast enhancement sequence (DCE) was applied, as one pre- and 4 postcontrast axial T1w fat sat sequences. A single contrast dose (0.2 mL/kg) was injected at a rate of 2 mL/s, followed by 20 mL normal saline flush, at a rate of 1 mL/s.
Average imaging parameters in DCE sequences were: 5 mm slice thickness interpolated to 2.5 mm intervals; matrix: 317 × 512; field of view: 16–18 cm2, flip angle, 25°. The total duration of the dynamic study was approximately 8–10 min. The patient afterward were removed from MRI, and scheduled for mammographic stereotactic or free hand ultrasound guided biopsy within one week.
2.3 Post-processing
This study applied standard evaluation system for all examined patients, including qualitative and quantitative parameters. These Qualitative items were: shape and borders of the lesion, Tw2 signal intensity, being single or multifocal or multicentric or bilateral, edema if present; including its degree of extension, septations if present and their enhancement pattern, inner architecture distortion if present, diffusion restriction, nipple retraction, skin and areolar thickening, vascularity of the lesion and axillary lymphadenopathy.
Quantitative post-processing was done through computer aided diagnostic (CAD) software, aiming to achieve the possible highest MRI examinations diagnostic sensitivity. This included evaluation of DCE sequences through recording post-contrast peak enhancement percentage value, time, pattern of enhancement (homogenous or heterogeneous or ring) and DCE curve type. Three types of DCE curves could be obtained, giving three different scores for evaluation. In type-I (continuous) curve; there is continuously increasing enhancement with no decline allover sequence time. In type-II (plateau) curve, the post-contrast enhancement shows peak enhancement, then it becomes fixed over time with no or less than 10% changes. In the Type-III (washout) curve; enhancement significantly decreases after initial rise.
Also one of the useful CAD software used in this study is the post-processing subtraction applied to pre-and post-contrast dynamic sequences in all phases i.e. subtracting pre-contrast from post-contrast images. This gave 4 subtracted sequences, eliminating all non-enhanced tissues e.g. Residual nonsuppressed fatty tissues and glandular tissue that may distort the clear visualization and evaluation of the enhancing lesions. Vascular scoring was made through maximum intensity projection (MIP) filter, which enables measurements of length and diameter of neoplastic feeding vessels. This gave four grades of vascularity of the examined breast tumors, ranging from 0 to 3.
Diffusion-weighted MRI (DWI) had been applied, including both qualitative and quantitative measures. This sequence is sensitive for diagnosis and estimation of the degree of diffusion restriction by tumor cells. It can help in diagnosis by differentiating condensed cellularity of malignant tumors from less condensed benign lesions. For quantitative analysis of the data acquired from DWI, ADC maps were automatically created, using software provided by the MRI system manufacturer (Syngo, Siemens Healthcare) using three b values (50, 800, and 1500 s/mm2). Tumors having ADC values less than (1.05 × 10−3 mm2/s) were widely considered malignant.Citation20
In an attempt for limitation of subjective MRI readers’ false results, we adapted scoring system for all MRI examination signs from Malich et al. who adapted Fischer’s scoringCitation21 with some little modification adding DW sequence results instead of blooming sign. These evaluation criteria were standardized for all patients with given points ranging from −3 to 3 for each character according to its benign or malignant features, respectively. If the total scores ⩾5, the examined lesion was considered malignant, in contrast to lesions with scores <5, that were considered benign lesions ().
Table 1 Scoring points and sum for biopsy proven (BP) benign and malignant lesions.Citation21
2.4 Results
Through a standard scoring system; post-imaging evaluation through items of scoring gave the following results: ().
T2 signal was given score ranging from −3 to 1, inversely proportionate to its signal intensity. Average of this point was −2.2 for proven benign lesions and 0.2 for proven malignant lesions (–). Edema was scored −1 if not found, 2 if bilateral or multifocal or diffuse and 2 if localized unilateral. Average for benign lesions was −0.8, while for malignant lesions was 1.3 (A). Hook or dendrite sign; if positive, it was given 1, if not it was given 0. It is not seen in any benign lesions, while average score was 0.3 for malignant swellings (B and A).
Figure 1 (A) Axial T2w image showing left breast hypointense (proven malignant) nodule (arrow) and right breast incidentally discovered small well defined benign looking hyperintense nodules (arrowheads). (B) Another patient with right breast malignant T2w hypointense infiltrative tumor with dendritic outline (arrow).
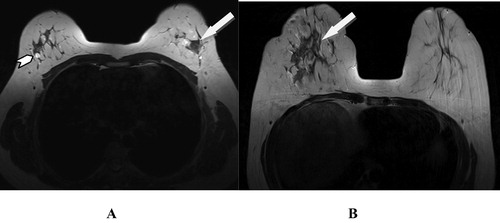
Figure 2 (A and B). Left breast proven malignant tumor: axial STIR images show left breast long TR hypointense swelling (arrowhead) with localized perifocal edema, asymmetrical skin thickening (arrow in B), dendritic infiltrative outline and positive hook sign (curved arrow).
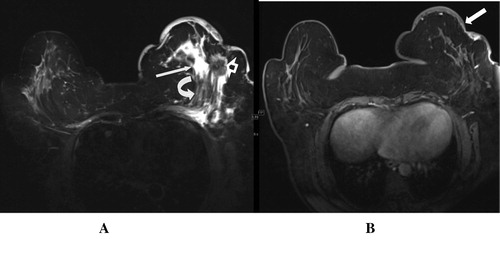
Figure 3 (A) T2w image, of right breast proven malignant tumor, shows irregular hypointense lesion (arrow), (B) ADC the lesion shows remarkable restricted diffusion, seen as low signal intensity. (C) Another patient with left breast benign swellings showing T2w Hyperintense signal (arrows). (D) No restricted diffusion at all in DWI.
Skin thickening was scored 1 if present and 0 if absent; averaging 0.1 in benign lesions and 0.6 for malignant ones ( and B). If the tumor showed restricted diffusion at DWI, it was scored 2, if not it was scored 0 giving average of 0.6 for benign lesions and 1.6 for malignancy (B and D). Vascularity of lesions was classified into 4 grades ranging for absent (0) to high (4); average score was 1.3 for benign lesions and 2.4 for malignant lesions ( and ).
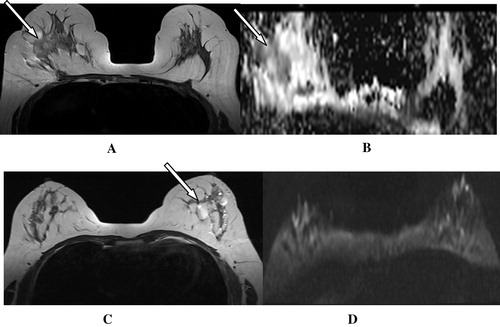
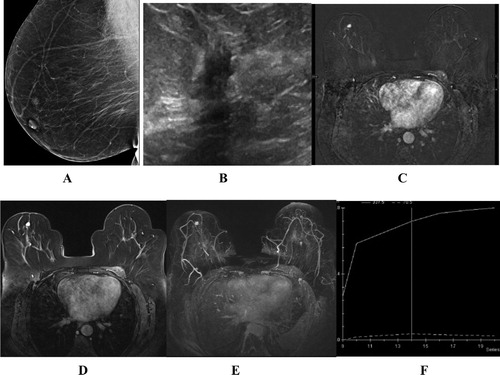
Figure 4 (A) US left breast showing central hypoechoic soft tissue lesion with highly suspicious malignant features (arrow). (B) STIR axial MRI image showing associated skin thickening with the lesion (arrow), and epsilateral axillary lymphadenopathy (notched arrow) together with diffusion restriction seen in ADC image (arrow in C) raise the malignant suspicion. (D) MIP for post-contrast axial T1w fat sat image shows grade I vascularity as relatively engorged local vascularity with slightly increased caliber. (E) DCE curve shows continuously raising curve consistent with benign nature of the swelling, chronic inflammatory process (granulomatous mastitis) was the pathological result of US guided biopsy.
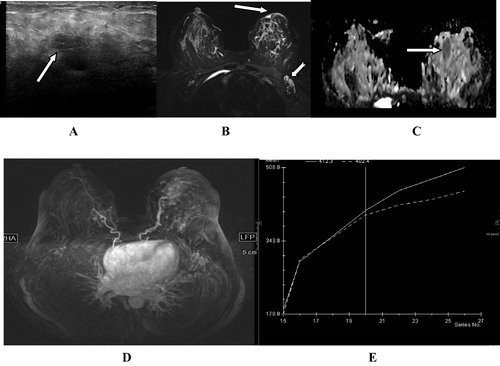
Figure 5 (A and B) Right breast ML mammogram and US breast show central nodule which was suspected to be malignant due to dense acoustic shadow in US. (C) MRI T1 postcontrast T1 fat sat showed avid post-contrast enhancement of the lesion. MIP images of early (D) and delayed (E) phases of dynamic postcontrast sequence show low vascular score (1) with no new angiogenesis. This together with benign continuously raising DCE curve (F) had supported the benign radiological diagnosis. (Pathologically proven traumatic fat necrosis with fibrous scar).
Postcontrast scoring had included wall definition of the swelling; 0 if well-defined and 1 if not well defined. Average was 0.3 for benign and 0.6 for malignant swellings. Also, postcontrast shape of the swelling; 0 if smoothly rounded or macrolobular and 1 if irregular or speculated; averaging 0 for benign and 0.7 for malignant lesions. Enhancement pattern, if homogenous was given 0 if heterogeneous it was given 1, averaging 0.1 for benign and 0.7 for malignant lesions.
Enhancement intensity peak was expressed as percent in correlation with initial precontrast intensity; giving 0 if < 50%, 1 if 50–100% and 2 if > 100%. Average score was 0.5 for benign lesions and 1.8 for malignant lesions. DCE curve shape was scored 0, if it was continually raising curve, 1 if plateau and 2 if showing washout. This gave average score 0.3 for benign tumors and 1.7 for malignant tumors (–). Axillary lymphadenopathy if not seen or smaller than 10 mm, it got 0 score; if more than 10 mm it was scored 1. This resulted in average 0.6 for benign lesions and 0.7 for malignant lesions. All these parameters collectively gave sum of 1.3 for benign lesions and 11.6 for malignant lesions.
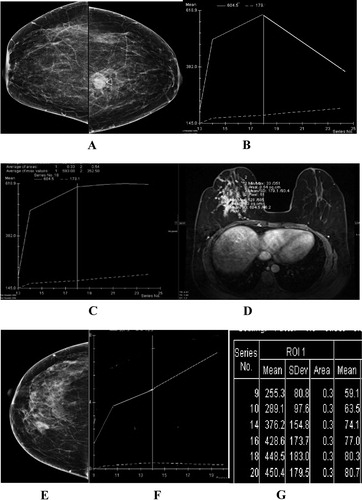
Figure 6 (A) CC mammogram showing left breast pathologically proven malignant nodule. (B) DCE curve of the lesion seen in A showing early peak with rapid washout (malignant curve). (C) Plateau curve for another malignant lesion of the right breast (D). (E) CC mammogram showing right breast pathologically proven benign nodule, (F) DCE curve demonstrating continuos rising enhancement (benign enhancement curve), (G) table of sequential ROI measures in DCE study showing continuos raising figures.
Before biopsy, MRI results had been interpreted for 36 patients’ swellings as benign tumors (MRI scoring points sum < 5 points). The other 84 patients’ tumors were reported malignant; having score ⩾ 5 points. 30 of these 36 MRI had been approved to be benign tumors, to be benign, while 6 had pathological diagnosis of being malignant. Of those 84 imaging reported malignant tumors, 81 tumors had pathological confirmation of being malignant. This means that there were 81 true positive and only 3 false positive cases, as well as 30 true negative and 6 false negative cases. These results gave indices of 93.1% sensitivity, 91.9% specificity, 96.4% positive predictive value (PPV) and 83.3% negative predictive value (NPV) .
Table 2 Results of the MRI examinations.
3 Discussion
MRI is expected to be sensitive tool of imaging for diagnosis, staging, and post-therapeutic follow up of breast cancer. Some newly applied imaging sequences had noticeably increased accuracy and sensitivity of MRI breast imaging. In an attempt to obtain the best results, we used combination of data obtained from morphology of the lesions and associated surrounding tissues changes in different sequences, as well as postcontrast dynamic changes i.e. utilizing the high spatial and high temporal resolution criteria of MRI examinations.
First of the evaluating items was T2w signal intensity of the lesion; T2w hypointensity favors the diagnosis of malignancy, while T2w hyperintensity supports benign diagnosis. This could be explained by the fact that, T2w hyperintensity reflects high water and/ or fat contents that are usual components of benign lesions. T2w low signal intensity might be secondary to collagen-rich fibrous tissue with low water and fat contents, which is known histopathological sign of malignancy.Citation22,Citation23 Scores for this item was arranged from −3 to 1 according to signal intensity percentage ().
Localized perifocal edema had been more commonly seen with malignant lesions. This might be attributed to the fact that, the malignant neoplastic vessels induced by neoangiogenesis have less intact basal membranes, leading to higher penetration of fluid and perifocal edema. Also, neoplastic proliferating cells produce large amounts of endothelium and vascular permeability destroying agents; with secondary increased permeability of neoplastic blood vessels.Citation24 Epsilateral asymmetrical skin thickening with breast tumors had got higher scores in association with malignancy in comparison with low score of benign lesions. This is due to subcutaneous lymphatic system clog by tumor cells. Asymmetrical skin thickening more than 5 mm should be considered suspicious for malignancy.Citation25,Citation24
Edema may be found in other different types of breast lesions e.g. inflammatory lesions, however, it will be more diffuse and not strictly limited to the focal lesion area.Citation25
DWI is very informative sensitive sequence with valuable qualitative and quantitative analysis, because malignant tumors usually had restricted water molecular diffusion. The diffusivity of water molecules is restricted in malignant tumors, due to compact high cellularity, intracellular and extracellular edema, and high viscosity.Citation24,Citation26 In study published by Woodhams et al. Citation20 for comparison between ADC values of benign and malignant breast tumors, ADC values were significantly lower in malignant tumors being less than 1.6 × 10(−3) mm2/s with 95% sensitivity. In our study, we standardized this figure for DWI in differential diagnosis of malignant from benign lesions. Proven malignant lesions had significantly higher scores of diffusion restriction in comparison with benign lesions .
A sure sign of aggressive infiltrating extension of malignant lesions is the speculated deeply extended irregular linear dendrites (hook sign), it had never been detected in any benign lesions. So, this hook sign can be confidently reported as sign of malignancy.Citation24 Vascular scoring is another moderately effective differentiating point. Malignant lesions had higher average scores, in comparison with benign lesions. This could be explained by new angiogenesis of malignant lesions, as angiogenesis is a key event in the uncontrolled progression of malignancy. Tumor angiogenesis involves multiple cellular processes including endothelial cell proliferation, migration and reorganization of extracellular matrix.Citation26
One of the most valuable sequences is the dynamic enhancement sequence (DCE), with many published literatures supporting its accuracy and sensitivity.Citation27,Citation28 Also, use of CAD software facilities that apply subtraction for post-contrast images, allowing kinetic analysis of the soft tissue tumors at, had significantly improved the sensitivity of DCE sequence. Early post-contrast enhancement peak (before 3 min), with high signal percentage value (more than 100% increase of initial tumor signal) and early washout (more than 20% signal intensity loss) within the time of the dynamic sequences are highly sensitive signs for diagnosis of malignancy.Citation26
This specific malignant pattern of enhancement could be explained by the abnormal development of malignant neovascularity, which lacks capillary network and contains multiple microscopic arteriovenous fistulae, causing rapid washout of contrast without capillary phase. Plateau type of DCE curve has relatively lower scoring points for malignancy, than the washout pattern. As documented in published researches, kinetic values of DCE curve, washout and early enhancement peak favor diagnosis of malignancy with sensitivity more than 80%. While, plateau curve is consistent with only 60% malignant possibility, in comparison with the continuous raising curve, which supports diagnosis of benign swellings, with less than 20% malignant possibility.Citation21,Citation27–Citation29
Axillary lymphadenopathy is relatively weak differentiating point with score ranging from −1 to 1, according to its absence or presence and size. This is because they can coexist with either benign of malignant lesions. Only if it shows the criteria which favors diagnosis of nodal metastasis, it will be supportive sign. These criteria include size if > 2 cm at its short axis, lost fatty hilum and rounded contour. As regards such parameter in the study, benign lesions had average score point of 0.6, in comparison with malignant lesions score which was 0.7, therefore it was not a considerable differentiating parameter in discrimination of the lesions.Citation30
Based on our experience through this study, magnetic resonance imaging (MRI) is highly sensitive imaging modality for breast cancer diagnosis. It clearly detected primary lesions and show their extent, as well as additional malignant foci that may be missed by mammographic imaging. Also, continued advances in computer software had much improved MRI’s diagnostic sensitivity, allowing to avoid unnecessary interventional breast procedures.
4 Conclusion
| • | MRI is very sensitive imaging tool for diagnosis of mammographic and or sonographic debatable lesions. | ||||
| • | To attain such successful results, there are standardized technical protocols for breast MRI examination should be carefully followed. | ||||
| • | This necessitates application of different MRI sequences with correlation of each sequence positive data to standard scoring system. | ||||
| • | Post-processing CAD software tools had undoubtedly increased diagnostic sensitivity of MRI examinations of breast tumors. | ||||
5 Recommendation
MRI breast examination is recommended to be applied not only for evaluation of debatable breast lesions seen, but also as screening tool at least once every two years for breast cancer high risk females. This protocol aims for early detection of cancer breast, which may be missed for long time of repeated scanning with conventional mammogram.
Conflict of interest
The authors have no conflict of interest to declare.
Notes
Peer review under responsibility of Alexandria University Faculty of Medicine.
Available online 14 January 2015
References
- US Breast Cancer Statistics <http://www.breastcancer.org/symptoms/understand_bc/statistics> [modified 26.09.13].
- <http://www.moh.gov.sa/en/HealthAwareness/Campaigns/Breastcancer/Pages/stat.aspx>.
- C.LehmanJ.BlumeP.WeatherallD.ThickmanN.HyltonScreening women at high risk for breast cancer with mammography and magnetic resonance imagingCancer1039200518981905
- American Cancer Society. Detailed guide: breast cancer; 2012 <http://www.cancer.org/Cancer/BreastCancer/DetailedGuide/index>.
- A.M.MartinB.L.WeberGenetic and hormonal risk factors in breast cancerJ Natl Cancer Inst92200011261135
- M.RobsonT.GilewskiB.HaasD.LevinP.BorgenBRCA-associated breast cancer in young womenJ Clin Oncol165199816421649
- Masaru.UejiEi.UenoDouglas.Osei-HyiamanTomoko.SaitoHideto.TakahashiKatsumi.KanoRisk Factors for breast cancer among Japanese women: a case-control study in Ibaraki, JapanBreast Cancer541998351358
- MP1MadiganR.G.ZieglerJ.BenichouC.ByrneR.N.HooverProportion of breast cancer cases in the United States explained by well-established risk factorsJ Natl Cancer Inst8722199516811685
- Mayo-clinic <http://www.mayoclinic.org/diseases-conditions/breast-cancer/basics/riskfactors%20/con-20029275>.
- J.A.TiceE.MearaD.L.WeaverC.VachonR.B.BarbashK.KerlikowskeBenign breast disease, mammographic breast density, and the risk of breast cancerJNCI J Natl Cancer Inst10514201310431049
- L.C.HartmannT.A.SellersM.H.FrostW.L.LingleBenign breast disease and the risk of breast cancerN Engl J Med3532005229237
- R.LakshmiR.AthiraJ.MarVijayalakshmiBreast cancer risk factors: preventive and non-preventiveIRJP31020124852
- C.C.HsiehM.GoldmanM.PaviaA.EkbomE.PetridouH.O.AdamiBreast cancer risk in mothers of multiple birthsInt J Cancer54119938184
- S.A.NarodM.P.DubeJ.KlijnOral contraceptives and the risk of breast cancer in BRCA1 and BRCA2 mutation carriersJ Natl Cancer Inst9423200217731779
- R.BaborJ.RehmAlcohol and public healthThe Lancet3652005519
- W.BergC.CampassiP.LangenbergM.SextonBreast imaging reporting and data system inter- and intraobserver variability in feature analysis and final assessmentAJR1746200017691777
- Hobson K. What’s the real risk of breast-cancer screening false positives? The Wall Street J [Europe Edition. Saturday, October 17, 2011, As of 5.17 pm].
- C.NealZ.YilmazM.NoroozianK.Klein1B.SundaramE.KazerooniImaging of breast cancer-related changes after surgical therapyAJR2022014262272
- E.HendrickBreast MRI: fundamentals and technical aspectsArtifacts and Errors in breast magnetic resonant imagingvol. 112007Springer Science LCC Business MediaNew York, USA190192
- R1.WoodhamsS.KakitaH.HataK.IwabuchiS.UmeokaC.E.MountfordDiffusion weighted imaging of mucinous carcinoma of the breast: evaluation of apparent diffusion coefficient and signal. Intensity in correlation with histologic findingsAJR Am J Roentgenol1931200926026610.2214/AJR.08.1670
- A.MalichD.R.FischerS.WurdingerJ.BoettcherC.MarxM.FaciusPotential MRI interpretation model: differentiation of benign from malignant breast massesAJR Am J Roentgenol18542005964970
- P.ProvenzanoD.InmanK.EliceiriJ.KnittelL.YanC.RuedenCollagen density promotes mammary tumor initiation and progressionBMC Med611200810.1186/1741-7015-6-11
- Wang Z, Sun L, Li Y, Li N. Relationship between elasticity and collagen fiber content in breast disease: a preliminary report. Ultrasonics, http://doi:10.1016/j.ultras.2014.10.016 [online 28.10.14].
- Werner A.KaiserSigns in MR-mammography2nd ed.2009Springer VerlagHeidelberg, Berlinp.80–88
- C.AnneKushwaha1J.GaryWhitman1B.CarolStelling1Primary inflammatory carcinoma of the breast. Retrospective review of mammographic findingsAJR1742000535538
- N.NishidaH.YanoT.NishidaT.KamuraM.KojiroAngiogenesis in cancerVasc Health Risk Manage232006213219
- L.TudoricaK.OhN.RoyM.KettlerY.ChenA feasible high spatiotemporal resolution breast DCE-MRI protocol for clinical settingsMagn Reson Imaging309201212571267
- X.LiL.R.ArlinghausG.D.AyersA.B.ChakravarthyR.G.AbramsonV.G.AbramsonDCE-MRI analysis methods for predicting the response of breast cancer to neoadjuvant chemotherapy: pilot study findingsMagn Reson Med714201415921602
- G.S.DhillonN.BellD.T.GinatA.LevitS.DestounisA.O’ConnellBreast MR imaging: what the radiologist needs to knowJ Clin Imaging Sci120114810.4103/2156-7514.85655
- S.SongB.SeoS.LeeA.YieK.Y.LEEClassification of metastatic versus non-metastatic axillary nodes in breast cancer patients: value of cortex-hilum area ratio with ultrasoundJ Breast Cancer1512012657010.4048/jbc.2012.15.1.6
