Abstract
Aims
The aim of this study was to investigate the antioxidant effects of l-carnitine on the oxidative stress parameters in oophorectomized rats.
Methods
Twenty-four female albino Wistar rats were used. Rats were divided into four groups: laparotomy-only (LOSALINE) group, oophorectomy plus l-carnitine 100 mg/kg/day (OXL100) group, oophorectomy plus l-carnitine 500 mg/kg/day (OXL500) group, and oophorectomy-only (OXSALINE) group. Experimental protocol was started on day 21 post-castration. Various dosage forms of l-carnitine or isotonic saline were administered intraperitoneally for 14 consecutive days. Nitric oxide (NO), malondialdehyde (MDA), total antioxidant status (TAS), total oxidative stress (TOS) and oxidative stress index (OSI) were evaluated in the tissues including kidney, liver and heart, and sera.
Result(s)
In the heart tissue samples, there was no difference in the levels of NO, OSI and TOS between the groups. However, MDA levels in OXSALINE group were significantly higher than OXL500 group. On the other hand, there was no statistically significant differences between the groups in terms of levels of NO, MDA, TAS, TOS and OSI in liver, kidney and sera samples.
Conclusion(s)
Levels of MDA in the heart tissue were significantly higher in OXSALINE group compared to OXL500 group. Thus, it may be suggested that l-carnitine reduces oxidative stress at least in the heart of oophorectomized rats.
1 Introduction
Menopause is defined as the permanent cessation of menstruation resulting from the loss of ovarian follicular activity. A variety of physiological changes occur in the body; some of them are related to cessation of ovarian functions, and others are because of the aging process.Citation1 It is also known that level of lipid peroxide, a marker of free-radical production and cell membrane lipid peroxidation (LPO), increases after menopause.Citation2 In various studies, it has been shown that the menopause impairs oxidative systems and causes increase in LPO.Citation2,Citation3 LPO is a process mediated by free oxygen radicals. During this process, the polyunsaturated fatty acids that are the structure of phospholipid, glycolipid, glyceride or sterol present in cell membrane are converted into various noxious products such as peroxides, alcohols, aldehydes, and hydroxy fatty acids. One of the major end products of lipid peroxidation, is malondialdehyde (MDA).Citation4
Oxygen-free radicals more generally are known as reactive oxygen species (ROS) along with reactive nitrogen species (RNS), and one of them is also nitric oxide.Citation4 Harmful effects of oxidative stress are counteracted by the action of antioxidants. It is well known that various estrogens scavenge reactive oxygen species (ROS) efficiently in both aqueous and lipophilic cellular components.Citation5
Furthermore, many in vitro and animal studies have reported that l-carnitine is a free radical scavenger, which protects antioxidant enzymes from oxidative damage.Citation6 l-carnitine is a quaternary amine containing seven carbon atoms with a molecular mass of 161.2 g/mol, water-soluble, small, highly polar compound which is widely distributed in nature.Citation7 Carnitine exists in two isomeric forms: l-carnitine and d-carnitine. Only the l-isomer of carnitine is metabolically active and synthesized primarily in liver also in the kidney. Tissues containing high levels of l-carnitine are unable to synthesize it. Tissues, other than liver and kidney, are dependent on the active uptake of carnitine from blood into tissues.Citation8,Citation9 It has been reported that l-carnitine is a powerful antioxidant, and l-carnitine exerts its antioxidant effect by preventing LPO.Citation10–Citation12
In this study, we aimed at investigating the protective effects of l-carnitine against oxidative stress in various tissues at the surgically menopausal rats.
2 Material and method
This study was conducted in the Experimental Medical Research Unit (DETAB, Kırıkkale University Medical Faculty, Kırıkkale, Turkey). The study protocol was approved by Kırıkkale University Animal Research Ethical Committee on 22.08.2011 (Approval Number 11/8-11-210).
2.1 Animals
A total of 24 adult female Wistar albino rats weighing 200–250 g were used in the study. The rats were given ad libitum access to a standard rat chow and tap water, and were housed as a group per cage under standard controlled temperature (20–22 °C), humidity (40–60%), and light (12 h light/12 h dark) in a facility for each group. All animal procedures compiled with the items of Helsinki Final Act (1986) related to experimental animals.
2.2 Study protocol
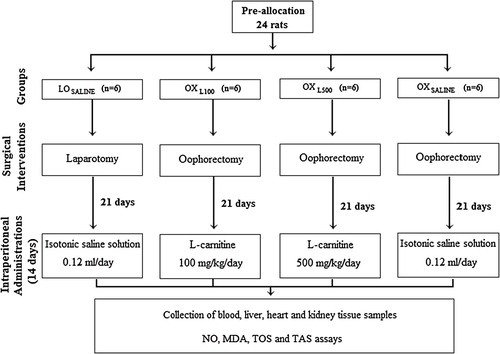
The rats were randomly allocated to 4 groups: laparotomy-only group (LOSALINE group; n = 6), oophorectomized-only group (OXSALINE group; n = 6), oophorectomy plus 100 mg/kg/day l-carnitine group (OXL100 group; n = 6) and oophorectomy plus 500 mg/kg/day l-carnitine group (OXL500 group; n = 6). The rats were anaesthetized with intraperitoneal injections of ketamine 80 mg/kg (Ketalar®, 50 mg/ml vial; Pfizer Pharmaceuticals, Istanbul, Turkey) and 10 mg/kg xylazine (Rompun®, 100 mg/ml vial; Bayer, Leverkusen, Germany) cocktail. All the interventions were performed under aseptic conditions and the rats were spontaneously breathing during surgical procedure. Surgery on each animal was performed through a ventral midline incision. No intervention other than incision was done in the SL group. The ovaries were exposed and bilateral oophorectomy was performed in the other three groups. Abdominal layers were closed anatomically, using 4/0 atravmatic Vicryl® and the animals were allowed to recover from anesthesia.
Experimental protocol was started on day 21 post-castration. In rats, antioxidant dosage recommendations for l-carnitine were 100 mg/kgCitation13,Citation14 and 500 mg/kgCitation15,Citation16 in the literature.
LOSALINE group was administered 0.12 ml/day of isotonic saline solution, OXL100 group was administered 100 mg/kg/day of l-carnitine, OXL500 group was administered 500 mg/kg/day of l-carnitine and OXSALINE group was administered 0.12 ml/day of isotonic saline solution, intraperitoneally, for 14 consecutive days. On day 35 post-castration, all the animals were anesthetized again and blood samples were obtained via intra-cardiac puncture before the animals were sacrificed. The blood samples were centrifuged at 10.000 rotations per minute (rpm), at +4 °C for 10 min (Eppendorf; 5804/R, Germany). Sera obtained from blood samples were transferred to polyethylene tubes. After removal the liver, heart and kidney tissue samples were immediately washed with cooled isotonic saline and placed on ice-cold plate. Sera and tissue samples (liver, heart and kidney) were stored at −80 °C until they were used for analysis of nitric oxide (NO), malondialdehyde (MDA), total antioxidant status (TAS), and total oxidative stress (TOS).
Flowchart of the study is presented in .
2.2.1 MDA measurement
The evaluation of MDA was based on the modified version of Yagi’s fluorometric method.Citation17 MDA levels were calculated using a standard curve and values are expressed as nmol/g protein.
2.2.2 NO measurement
Nitrates/nitrites were assayed according to the method of Miranda et al.Citation18 NO (nitrates/nitrites) levels were calculated using a standard curve and multiplying by the dilution factor. Values are expressed as μmol/g protein.
2.2.3 TOS measurement
Erel’s method was used for measuring TOS.Citation19 TOS was assayed with TOS commercial kit (Rel Assay Diagnostics®, Gaziantep, Turkey, 2010) adapted to an Olympus® AU 480 autoanalyzer (Fully Automatic Chemistry Analyser [AU480] Olympus Diagnostics, Japan). The results are expressed as micromolar hydrogen peroxide equivalents per g protein (μmol H2O2 equiv/g protein).
2.2.4 TAS measurement
Erel’s method was used for measuring TAS.Citation20 TAS was assayed with TAS commercial kit (Rel Assay Diagnostics®, Gaziantep, Turkey, 2010) adapted to an Olympus® AU 480 autoanalyzer (Fully Automatic Chemistry Analyser [AU480] Olympus Diagnostics, Japan). The results are expressed as micromolar Trolox equivalents per g protein (μmol Trolox equiv/g protein).
2.2.5 Calculation of oxidative stress index (OSI)
For calculation, the resulting unit of TAS was converted to μmol/L, and the OSI value was calculated according to the following formula: OSI (arbitrary unit) = 100 ∗ TOS (μmol H2O2 equivalent/L)/TAS (μmol Trolox equiv/L).Citation21
3 Statistical analysis
The Statistical Package for Social Sciences (SPSS) 11.5 for Windows software (SPSS Inc., Chicago, IL, United States) was used for statistical analysis. We used the Shapiro Wilk test to test variables for normality of distribution. Levene’s test was used for testing homogeneity of variances. Results of descriptive analysis were presented as mean (±standard deviation) or median (minimum–maximum). The parametric data were statistically analyzed by one-way ANOVA and the inter group comparison of means was conducted using a post hoc Tukey’s HSD (Honestly Significant Difference) test. The nonparametric data were analyzed by Kruskal-Wallis rank sum tests followed by Conover’s multiple comparison tests. Results are considered significant when p < 0.05.
4 Results
In the kidney tissue samples, levels of NO, MDA, and TOS in OXSALINE group were higher (but not significantly) than those in OXL100 and OXL500 groups (p > 0.05 for all comparisons). There was no difference in TAS, and OSI levels of the groups (p > 0.05 for all comparisons) ().
Table 1 Levels of NO, MDA, TOS, TAS, and OSI in the kidney tissue samples.
In the heart tissue samples, there was no difference in the levels of NO, OSI and TOS between the groups (p > 0.05 for all comparisons). MDA levels in OXL500 group were significantly lower than those in OXSALINE group (p = 0.002) (, ).
Table 2 Levels of NO, MDA, TOS, TAS, and OSI in the heart tissue samples.
In the liver tissue samples, there was no difference in the levels of NO, MDA, TAS, TOS and OSI between the groups (p > 0.05 for all comparisons) ().
Table 3 Levels of NO, MDA, TOS, TAS, and OSI in the liver tissue samples.
In the serum samples, levels of NO, MDA, and TOS in OXSALINE group were higher (but not significantly) than those in OXL100 and OXL500 groups (p > 0.05 for all comparisons). There was no difference in TAS and OSI levels of the groups (p > 0.05 for all comparisons) ().
Table 4 Levels of NO, MDA, TOS, TAS, and OSI in the serum samples.
5 Discussion
Estrogen, a natural antioxidant, exhibits radical scavenger properties Citation22,Citation23 and protects vascular smooth muscle cell membrane phospholipids against peroxidation.Citation24 After menopause, the level of estrogen decreases in sera. Menopause impairs oxidative system, and causes increase in LPO and oxidative stress.Citation2,Citation3
In our study, we found as expected that MDA levels of oophorectomized groups were higher compared to LOSALINE group, although these differences were not statistically significant level. Bednarek-Tupikowska et al. found that the level of lipid peroxide increases after menopause and can be reduced by Hormone Replacement therapy (HRT).Citation2 And also in other study, it has been determined that oxidative stress markers (MDA, glutation) and protein’s redox level increase in the brain, liver and plasma of oophorectomized rats and all these processes could be prevented by HRT.Citation5 It seems to be HRT might be protective oxidative stress in menopause. However, sometimes we cannot use these drugs and we are supposed to find other choices.
It has been shown that exogenous l-carnitine is a free-radical scavenger and it protects cells against oxygen radicals.Citation10,Citation11 To our knowledge, there is no study in the literature that assessed the effects of l-carnitine on oxidant and antioxidant status in sera or other tissues, particularly in menopause or in experimental/surgical menopause.
In our study, NO, MDA, TAS, TOS and OSI levels in the various tissue specimina and sera of surgically menopause rats were detected. These oxidative stress markers were mostly increased in the kidney tissue specimina of OXSALINE group when compared with the other groups, although these differences were not significant level. Oxidative stress parameters’ levels were similar in OXL100, OXL500 and LOSALINE groups. Contrary to our hypothesis, we could not find protective effect of l-carnitine in kidney tissues. In the literature, there is no study on the effects of carnitine on antioxidant system and LPO about kidney during menopause. However, Fatouros et al. have investigated the effects of l-carnitine on oxidative stress response in patients with renal disease and they have observed that l-carnitine reduces exercise-induced oxidative stress, increases levels of antioxidants, and improves performance in patients with end-stage renal failure.Citation25
Furthermore when we examined liver tissue specimina, NO, MDA, TAS, TOS and OSI levels were mostly increased in OXSALINE group when compared with the other groups. However these differences were not significant as in the kidney specimina. Oxidative stress parameters’ levels were similar in l-carnitine treated groups and the LOSALINE group. In reviewing of the literature, it could be seen that there are no menopause-specific liver changes. However, it was known that oxidative stress and LPO have important roles in occurence of liver injury. l-carnitine exhibits antioxidant effect by reducing metabolic stress and the use of l-carnitine has recently come into question in the treatment of many diseases such as liver disease.Citation25 While there is no study on menopause and the use of l-carnitine, there are studies that investigate the effects of carnitine on liver disease in the literature. In a study investigating the protective effects of l-carnitine against hydrogen peroxide cytotoxicity in hepatocytes, it has been reported that l-carnitine that exhibits antioxidant effects by reducing MDA and increasing catalase (CAT) and superoxide dismutase (SOD), reduces cytotoxic effect of hydrogen peroxide.Citation26
It is accepted that menopause is an important risk factor for cardiovascular disease. In cases of estrogen deficiency such as menopause, deterioration in oxidative system resulted in increasing of lipoprotein oxidation which causes cardiovascular disease.Citation27 In this study, we found that MDA (marker of LPO) levels in heart tissue were significantly lower in OXL500 group compared to OXSALINE and this situation supports our hypothesis that high dose l-carnitine reduces oxidative stress in heart tissue. Conversely, there were differences in TAS levels between the groups. This suggests that high oxidative stress may be suppressed by antioxidants which increase reactively or distinct antioxidant mechanism. Suppression of oxidative stress by l-carnitine may play an important role in the heart. These results may be related to treatment duration or dose. In the literature, carnitine and its derivative have been subjected to numerous studies which have shown promising results.Citation28–Citation32 Carnitine that exerts cardio-protective effect against hypoxia and oxidative stress, is a specific treatment choice.Citation30 In experimental studies, it has been determined that carnitine administration before or during acute ischemia increases ATP levels, while reducing oxidative stress, improves hemodynamic parameters, and reduces the degree of ST segment elevation.Citation13,Citation28,Citation31,Citation32 In a rat study conducted by Gomez-Amores et al., it has been shown that chronic l-carnitine treatment reduces systemic oxidative stress related to hypertension and strengthens antioxidant defense.Citation10
According to our study, NO, MDA, TAS, TOS and OSI levels in the sera were similar between all the groups. In parallel with the literature, serum levels of NO and MDA were higher in the OXSALINE group compared to the LOSALINE group, and serum levels of NO, MDA and TOS were lower in the l-carnitine treated groups compared to the OXSALINE group, although these differences were not statistically significant. This can be explained by the rapid uptake of exogenous carnitine via active transport from plasma to tissues.Citation33 Gumral et al. have examined the effects of carnitine and selenium on oxidative stress in blood of rat induced by 2.45-GHz electromagnetic radiation, and they have found that LPO levels were higher in the radiation exposure group compared to controls and also glutathione levels were lowest in the l-carnitine receiving group. They have concluded that l-carnitine exhibited protective effect via its free radical scavenging activity in the blood, but no such effect has been demonstrated for selenium.Citation34
In accordance with our hypothesis, the level of MDA (a LPO product) in the heart tissue was higher in OXSALINE group when compered to the OXL500 group. We couldn’t find similar results in other tissues and sera. These results may be related to tissue-specific antioxidant effect of l-carnitine. Moreover, Topcuoglu et al. have reported similar tissue-specific antioxidant effect for estrojen.Citation5 Probably, l-carnitine can act in different ways on different tissues. In our study, l-carnitine exhibits statistically significant reduction in MDA levels in heart tissue in surgically menopausal rats. This effect may be dose dependant.
In conclusion, l-carnitine 500 mg/kg/day exhibits antioxidant effect in the heart by reducing oxidative stress. l-carnitine may be an alternative for menopausal supportive treatment, because it is naturally occurring in human body, easy-to-obtain, easy-to-administrate, cheap and has low incidence of adverse events. However, there is a need for large scale, prospective and long-term studies that evaluate the effects of carnitine in menopause.
Conflict of interest
There is no conflict of interest.
Notes
Peer review under responsibility of Alexandria University Faculty of Medicine.
Available online 10 August 2016
References
- WHO scientific group. Research on the menopause in the 1990s. In: WHO technical report series, 866. Geneva: World Health Organization; 1996. p. 1–107.
- G.Bednarek-TupikowskaU.TworowskaI.JedrychowskaB.RadomskaK.TupikowskiB.Bidzinska-SpeichertEffects of oestradiol and oestroprogestin on erythrocyte antioxidative enzyme system activity in postmenopausal womenClin Endocrinol (Oxf)642006463468
- J.E.CastelaoM.Gago-DominguezRisk factors for cardiovascular disease in women: relationship to lipid peroxidation and oxidative stressMed Hypotheses7120083944
- M.ValkoC.J.RhodesJ.MoncolM.IzakovicM.MazurFree radicals, metals and antioxidants in oxidative stress-induced cancerChem Biol Interact1602006140
- A.TopcuogluH.UzunH.BalciM.KarakusI.CobanT.AltugEffects of estrogens on oxidative protein damage in plasma and tissues in ovariectomised ratsClin Invest Med322009E133E143
- J.KolodziejczykJ.Saluk-JuszczakB.WachowiczL-carnitine protects plasma components against oxidative alterationsNutrition272011693699
- L.L.BieberCarnitineAnnu Rev Biochem571988261283
- G.DenizKarnitin: sentez, metabolizma, fonksiyon ve iskemik kalpte terapötik önemiT Klin Tıp Bilimleri1919995562
- M.A.ArsenianCarnitine and its derivatives in cardiovascular diseaseProg Cardiovasc Dis401997265286
- L.Gomez-AmoresA.MateJ.L.Miguel-CarrascoL.JimenezA.JosA.M.CameanL-carnitine attenuates oxidative stress in hypertensive ratsJ Nutr Biochem182007533540
- S.SushamakumariA.JayadeepJ.S.KumarV.P.MenonEffect of carnitine on malondialdehyde, taurine and glutathione levels in heart of rats subjected to myocardial stress by isoproterenolIndian J Exp Biol271989134137
- T.ThangasamyP.JeyakumarS.SittadjodyA.G.JoyeeP.ChinnakannuL-carnitine mediates protection against DNA damage in lymphocytes of aged ratsBiogerontology102009163172
- Y.SuzikiT.KamikawaA.KobayashiEffects of L-carnitine on tissue levels of acyl carnitine, acyl coenzyme A and high energy phosphate in ischemic dog heartsJpn Circ J451981687694
- F.BodeaA.BoceaN.DeceaL-carnitine decreases oxidative stress induced by experimental hypobaric hypoxiaPediatr Endocrinol Diabetes Metab1620107881
- J.KellerR.RingseisS.PriebeR.GuthkeH.KlugeK.EderEffect of L-carnitine on the hepatic transcript profile in piglets as animal modelNutr Metab (Lond)8201176
- M.M.Sayed-AhmedM.A.EissaS.A.KenawyN.MostafaM.CalvaniA.M.OsmanProgression of cisplatin-induced nephrotoxicity in a carnitine-depleted rat modelChemotherapy502004162170
- K.YagiLipid peroxides in hepatic, gastrointestinal, and pancreatic diseasesAdv Exp Med Biol3661994165169
- K.M.MirandaM.G.EspeyD.A.WinkA rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitriteNitric Oxide520016271
- O.ErelA novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cationClin Biochem372004277285
- O.ErelA new automated colorimetric method for measuring total oxidant statusClin Biochem38200511031111
- M.HarmaM.HarmaO.ErelIncreased oxidative stress in patients with hydatidiform moleSwiss Med Wkly1332003536563
- R.K.DubeyE.K.JacksonEstrogen-induced cardiorenal protection: potential cellular, biochemical, and molecular mechanismsAm J Physiol Renal Physiol2802001F365F388
- M.B.Ruiz-LarreaC.MartinR.MartinezR.NavarroM.LacortN.J.MillerAntioxidant activities of estrogens against aqueous and lipophilic radicals; differences between phenol and catechol estrogensChem Phys Lipids1052000179188
- R.K.DubeyY.Y.TyurinaV.A.TyurinD.G.GillespieR.A.BranchE.K.JacksonEstrogen and tamoxifen metabolites protect smooth muscle cell membrane phospholipids against peroxidation and inhibit cell growthCirc Res841999229239
- I.G.FatourosI.DouroudosS.PanagoutsosP.PasadakisM.G.NikolaidisA.ChatzinikolaouEffects of L-carnitine on oxidative stress responses in patients with renal diseaseMed Sci Sports Exerc42201018091818
- J.L.LiQ.Y.WangH.Y.LuanZ.C.KangC.B.WangEffects of L-carnitine against oxidative stress in human hepatocytes: involvement of peroxisome proliferator-activated receptor alphaJ Biomed Sci19201232
- T.AkcayY.DincerR.KayaliU.ColgarE.OralU.CakatayEffects of hormone replacement therapy on lipid peroxides and oxidation system in postmenopausal womenJ Toxicol Environ Health A59200015
- J.D.FoltsA.L.ShugJ.R.KokeN.BittarProtection of the ischemic dog myocardium with carnitineAm J Cardiol41197812091214
- J.L.Miguel-CarrascoA.MateM.T.MonserratJ.L.AriasO.AramburuC.M.VazquezThe role of inflammatory markers in the cardioprotective effect of L-carnitine in L-NAME-induced hypertensionAm J Hypertens21200812311237
- D.F.PaulyC.J.PepineThe role of carnitine in myocardial dysfunctionAm J Kidney Dis412003S35S43
- A.L.ShugJ.H.ThomsenJ.D.FoltsN.BittarM.I.KleinJ.R.KokeChanges in tissue levels of carnitine and other metabolites during myocardial ischemia and anoxiaArch Biochem Biophys18719782533
- J.H.ThomsenA.L.ShugV.U.YapA.K.PatelT.J.KarrasS.L.DeFeliceImproved pacing tolerance of the ischemic human myocardium after administration of carnitineAm J Cardiol431979300306
- J.HarmeyerThe physiological role of L-carnitineLohmann Inf2720021521
- N.GumralM.NazirogluA.KoyuK.OngelO.CelikM.SayginEffects of selenium and L-carnitine on oxidative stress in blood of rat induced by 2.45-GHz radiation from wireless devicesBiol Trace Elem Res1322009153163