Abstract
In March 2013, the cases of human infection with influenza A of H7N9 subtype were first reported. Preliminary data suggested that the H7N9 isolates are sensitive to neuraminidase inhibitors, such as oseltamivir, which is the recommended choice of treatment. On April 2nd, a 56-year-old male patient was presented with fever and cough to our hospital. He had previous history of close contact with another H7N9 patient. After caring for his wife (a confirmed H7N9 infection case died on April 3rd), this patient showed flu like symptoms on April 2nd. On the same day, oseltamivir (75 mg bid) treatment was started. Throat swab specimens were screened for H7N9 virus by real-time reverse transcriptase-polymerase chain reaction. The patient was hospitalized on April 4th. Initial specimens on April 4th and April 5th were negative for H7N9. But the specimen collected on April 10th was tested positive for H7N9. The result was confirmed by Shanghai Municipal Center of Disease Control and Prevention. By April 25th when we submitted this report, swab specimens of this patient were still positive for H7N9. This case calls for increased awareness of potential resistance of H7N9 to oseltamivir.
Introduction
Many subtypes of Influenza A viruses normally circulate in wild birds and generally do not cause diseases. But when they were transmitted into domestic poultry, the viruses can cause diseases in the new hosts.Citation1,Citation2 On rare occasions, the viruses can cause diseases in humans who were exposed to infected poultry.Citation1,Citation3 Influenza viruses are segmented RNA viruses. Subtypes of the viruses are defined according to the types of hemagglutinin (H1 to H17) and of neuraminidase (N1 to N9) encoded by two of the eight RNA segments.Citation2 When more than one subtype of influenza A virus are infecting a single animal cell, different segments of RNA from different influenza viruses can be packed in a single viral particle. This mixing of genetic materials results in a novel combination of RNA segments.Citation2 The novel influenza virus can have a new host-range.
In March 2013, the cases of human infection with influenza A of H7N9 subtype were reported.Citation4,Citation5,Citation6,Citation7 Genetic analysis of human H7N9 isolates indicates that the virus contains a novel combination of segments from duck and two wild birds.Citation4,Citation5 The H7N9 infections have a high rate of morbidity and mortality.Citation4,Citation5,Citation6,Citation7 As of April 17th, 21% (n=17) of the confirmed cases have died of acute respiratory distress syndrome or multiple organ failure; 73% (n=60) confirmed cases remained critically ill.Citation7 Preliminary data suggest that the H7N9 isolates are sensitive to treatment with neuraminidase inhibitors,Citation4,Citation5,Citation8 such as oseltamivir, which is the recommended choice of treatment.Citation9 Previous studies indicate that oseltamivir treatment within 8 days of symptom onset is effective to treat human infection with H5N1 virus, which is also an avian virus and causes severe illness.Citation10 Most of the reported H7N9 infections received oseltamivir treatment within 6 days (median) of disease onset.Citation7 Yet, nearly half of them developed acute respiratory distress syndrome.Citation7 In this article, we describe the clinical outcome of one confirmed case of H7N9 who has received oseltamivir treatment on the same day as the onset of illness.
MATERIALS AND METHODS
Infected subject: on April 2nd this patient developed systemic inflammatory response syndrome after an episode of nonproductive cough, fever >38.5 °C, and drowsiness. He was under close observation because he had taken care of his wife, who fell critically ill on March 27th, died on April 3rd and was confirmed on April 4th to have H7N9 infection. For precautious purpose, we initiated oseltamivir on April 2nd. Respiratory specimens obtained from the patient were tested for influenza and other respiratory viruses by real-time reverse transcriptase-polymerase chain reaction. We also obtained and analyzed clinical, epidemiologic, and virological data from this patient.
RESULTS
On April 2nd we administered oseltamivir as initial treatment. Initial specimens on April 4th (day 1 in hospital) and April 5th (day 2) were negative for H7N9. But the patient's symptoms worsened. On April 5th, the patient remained conscious but he was in a state of hypoxemia, tachypnea, tachycardia, and his body temperature fluctuated between 37.5 °C–39 °C. Hematological and biochemical laboratory tests showed pH 7.47, PaCO2 4.70 kPa, leukocyte count of 5480 with 68.0% neutrophils, prothrombin time 18.0 sec, activated partial thromboplastin time 49 sec, creatine kinase 290.0 U/L, and normal renal and hepatic function tests. Computed tomography (CT) on April 4th showed bilateral lower lung inflammatory exudation associated with acute lung injury (Figure ). On April 5th, the patient was treated with oseltamivir, moxifloxacin, piperacillin and tazobactam.
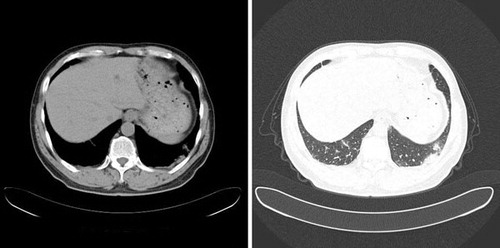
As the disease progressed further, the patient became critically ill. CT scan on April 6th (day 3) and April 15th (day 12) showed expanded lesions (Figures and ). The patient developed severe hypoxemia and was incubated for mechanical ventilation. On April 10th (day 7), throat swab specimens were tested positive for A/H7N9 influenza virus. This result was confirmed by Shanghai Municipal Center of Disease Control and Prevention. The patient continued to receive treatment and was tested daily from his throat swab specimens for H7N9 virus. At the time of writing (April 25th), the patient's specimen remained H7N9 positive and the patient's condition remained critical.
Figure 2 CT scan on April 6th, 2013 showed advanced bilateral lungs lesion with consolidation and exudation.
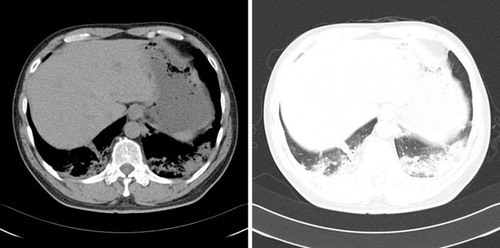
Figure 3 April 9th–15th chest radiographs: ground-glass opacity was observed on every graph. No significant improvement after treatment. (A): April 9th. (B): April 10th. (C): April 11st. (D): April 12nd. (E): April 14th. (F): April 15th.

This patient's wife had a confirmed H7N9 infection. She developed symptoms of fever and pneumonia beginning on March 27th and died on April 3rd. She was a house wife and visited a food market almost every day, where live poultry was sold. She had 31 close contacts who have been monitored for possible H7N9 infection. The patient and his sister-in-law presented with flu-like symptoms after caring for patient's wife. The sister tested negative for A/H7N9 influenza virus and her symptoms resolved shortly afterward. The patient and his sister-in-law did not have a history of frequent visits to the market. They did not report having close contact with live poultry before the onset of symptoms.
DISCUSSION
The disease caused by the novel avian influenza A (H7N9) is of significant public health concern because of the high mortality of the infection in humans.Citation5,Citation6,Citation7,Citation11 Oseltamivir is the first choice of treatment for human influenza A infection recommended by National Health and Family Planning Commission of China.Citation9 But its effectiveness on human influenza A (H7N9) is unknown. Here we report a confirmed case of human H7N9 infection that did not respond to oseltamivir very well. Clinical outcomes were not favorable despite the use of oseltamivir. H7N9 virus was detected from throat swab specimens on day 8 post treatment, while no virus was detected during the first six day of disease onset and treatment.
Chan et al. reported that initiation of oseltamivir before onset of respiratory failure is effective in treating human H5N1 infection, whereas oseltamivir initiation during the time of advanced respiratory failure that requires ventilator support is ineffective.Citation12 For the current case of human H7N9 infection, the patient did not experience any advanced respiratory failure when he was put on the oseltamivir treatment. Nevertheless, he went on to develop severe hypoxemia 11 days after the oseltamivir treatment and required ventilation support. A review of human H7N9 cases found that nearly half of the oseltamivir treated patients developed respiratory failure during treatment.Citation7 Thus the effectiveness of oseltamivir to treat human H7N9 is questionable. The median age of the human H7N9 cases (63 years old) reported so far is significantly higher than that of the individuals in the H5N1 study (18 years old).Citation7 Further investigation is necessary to determine whether age or other factors may contribute to the effectiveness of oseltamivir in treating human H7N9 infection.
Oseltamivir-resistant influenza A virus from humans and wild birds have been reported.Citation13 The sequence of the H7N9 virus isolated from our case is not available. But sequencing analysis of human isolates from other patients indicates that H7N9 should be sensitive to oseltamivir.Citation8
Preliminary epidemiological study of 82 confirmed human H7N9 cases provides no strong evidence for human-to-human H7N9 transmission.Citation7 The same study, however, acknowledges that limited human-to-human H7N9 transmission is possible after “prolonged, unprotected and close” contact with another suspected H7N9 individual.Citation7 The incubation period of human H7N9 infection is estimated to be six days.Citation7 In our case, the patient developed symptoms six days after his wife had fever. Both the patient and his wife had confirmed H7N9 infection. Study of human volunteer challenged with influenza A virus indicates that flu symptom and virus shedding initiate and peak together within 1 day.Citation14 Thus, the onset of patient's symptom was consistent with the possibility that patient have acquired the H7N9 infection from his wife. The patient's history of having unprotected, prolonged and close contacts with a confirmed H7N9 case further increased the likelihood that he acquired the H7N9 infection from another human. Two family clusters were reported.Citation7 However, H7N9 infection could not be confirmed in the index case of one cluster and in the initial case of another cluster.Citation7 In the current report, the patient and his wife both had confirmed H7N9 infection. The transmissibility of H7N9 among humans, however, is likely to be very weak, since only one of 31 close contacts beside the patient developed any flu-like symptoms.Citation7 That person was H7N9-negative and the symptom quickly resolved. We could not determine whether the patient and his wife were exposed to a common or a different source of H7N9, despite the fact that the patient reported no exposure to live poultry. Further investigation of this family cluster may help definitively determine the source of H7N9 infection of our present case.
In summary, we reported the first case of family cluster in which the initial case and the subsequent case both have confirmed H7N9 infection. We also described the ineffectiveness of oseltamivir to control disease progress in this case of human H7N9 infection, despite the fact that the treatment was initiated on the same day as the disease onset.
- Webster RG. Influenza virus: transmission between species and relevance to emergence of the next human pandemic. Arch Virol Suppl1997; 13: 105–113.
- Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y. Evolution and ecology of influenza A viruses. Microbiol Rev1992; 56: 152–179.
- Li KS, Guan Y, Wang Jet al . Genesis of a highly pathogenic and potentially pandemic H5N1 influenza virus in eastern Asia. Nature2004; 430: 209–213.
- Chen Yu, Liang Weifeng, Yang Shiguiet al . Human infections with the emerging avian influenza A H7N9 virus from wet market poultry: clinical analysis and characterisation of viral genome. Lancet2013 April 25; doi:https://doi.org/10.1016/S0140-6736(13)60903-4.
- Gao R, Cao B, Hu Yet al . Human Infection with a Novel Avian-Origin Influenza A (H7N9) Virus. N Engl J Med2013 Apr 11; doi:https://doi.org/10.1056/NEJMoa1304459.
- Yang F, Wang J, Jiang Let al . A fatal case caused by novel H7N9 avian influenza A virus in China. Emerg Microbes Infect2013; 2: e19.
- Qun Li, Lei Zhou, Minghao Zhouet al . Preliminary Report: Epidemiology of the Avian Influenza A (H7N9) Outbreak in China. N Engl J Med2013 Apr 24; doi:https://doi.org/10.1056/NEJMoa1304617.
- Kageyama T, Fujisaki S, Takashita Eet al . Genetic analysis of novel avian A(H7N9) influenza viruses isolated from patients in China, February to April 2013. Euro surveillance : European communicable disease bulletin 18(2013). Euro Surveill2013; 18.pii: 20453.
- National Health and Family Planning Commission. Diagnosis and treatment guideline for human infection with influenza A (H7N9) virus, 2nd ed. Beijing: NPFPC, 2013. Available at http://www.moh.gov.cn/mohgjhzs/s7952/201304/98ceede1daf74a45b1105f18c4e23ece.shtml.
- Adisasmito W, Chan PK, Lee Net al . Effectiveness of antiviral treatment in human influenza A(H5N1) infections: analysis of a Global Patient Registry. J Infect Dis2010; 202: 1154–1160.
- Wen YM, Klenk HD. H7N9 avian influenza virus - search and re-search. Emerg Microbes Infect2013; 2: e18.
- Chan PK, Lee N, Zaman Met al . Determinants of antiviral effectiveness in influenza virus A subtype H5N1. J Infect Dis2012; 206: 1359–1366
- Govorkova EA, Baranovich T, Seiler Pet al . Antiviral resistance among highly pathogenic influenza A (H5N1) viruses isolated worldwide in 2002–2012 shows need for continued monitoring. Antiviral Res2013; 98: 297–304.
- Carrat F, Vergu E, Ferguson NM. Time lines of infection and disease in human influenza: a review of volunteer challenge studies. Am J Epidemiol2008; 167: 775–785.