Abstract
Influenza A (H9N2) viruses are a genetically diverse population that infects wild and domestic avian species and mammals and contributed the internal gene segments to the A/H5N1 and A/H7N9 viruses associated with lethal human infections. Here we comprehensively assess the potential risk to mammals of a diverse panel of A/H9N2 viruses, representing the major H9N2 clades, using a combination of in vitro assays (e.g., antiviral susceptibility and virus growth in primary differentiated human airway cells) and in vivo assays (e.g., replication, transmission and/or pathogenicity of viruses in ducks, pigs, mice and ferrets). We observed that viruses isolated from humans, A/Hong Kong/1073/1999 and A/Hong Kong/33982/2009, had the highest risk potential. However, the A/swine/Hong Kong/9A-1/1998 and A/chicken/Hong Kong/G9/1997 viruses also displayed several features suggesting a fitness profile adapted to human infection and transmission. The North American avian H9N2 clade virus had the lowest risk profile, and the other viruses tested displayed various levels of fitness across individual assays. In many cases, the known genotypic polymorphisms alone were not sufficient to accurately predict the virus’ phenotype. Therefore, we conclude that comprehensive risk analyses based on surveillance of circulating influenza virus strains are necessary to assess the potential for human infection by emerging influenza A viruses.
Introduction
Outbreaks of influenza A viruses continue to cause morbidity and mortality worldwide in domestic birds and a wide variety of mammals, including humans. In recent years, a number of wholly avian influenza strains have crossed the species barrier and caused human infection and even death. These include the highly pathogenic avian H5N1 influenza viruses that are currently endemic in China, Vietnam, Cambodia, Indonesia, Bangladesh and Egypt and associated with significant mortality. More limited human infections have been associated with a highly pathogenic avian H7 influenza virus, which has been detected in poultry workers and veterinarians in the Netherlands and Mexico,Citation1 and the emerging avian H7N9 viruses in China.Citation2,Citation3 Less recognized are the mild human infections associated with the low-pathogenic avian influenza H9N2 viruses.Citation4,Citation5 Although much scientific and public health interest has focused on the H5N1 influenza viruses, the H7 and H9 viruses are also considered pandemic threats. The H9 viruses are of particular concern, given their extensive species tropism and distribution throughout many parts of Eurasia and the fact that they donated the internal genes to the currently circulating H5N1 and H7N9 viruses.Citation2,Citation6,Citation7
H9N2 viruses are widespread in nature. They are routinely isolated from wild birds and occasionally from pigs and other mammalian species.Citation8 They are also considered enzootic in poultry in some Asian and Middle Eastern countries.Citation9 In experimentally infected dogs, H9N2 viruses cause respiratory disease and are shed.Citation10 Antibodies against H9 influenza have been found in pet and performing monkeys in Southeast Asia.Citation11 Phylogenetic analysis has revealed that since 1997, multiple clades of H9N2 viruses have been circulating, including the G1-like lineage represented by A/quail/Hong Kong/G1/1997 and the Y280-like lineage represented by A/chicken/Hong Kong/G9/1997.Citation12 As a result of this cocirculation, multiple reassortant subtypes have been observed.Citation13 Given the wide distribution of H9N2 viruses, it is not surprising that sporadic cases of human infection have occurred, generally causing mild illness in Hong Kong and mainland China since the late 1990s.Citation5,Citation14 Currently, under investigation are the source and risk factors for H9 transmission to humans and the risk of infection with H9N2 viruses among persons working in live poultry markets, where these viruses are prevalent. One study in southern China reported that the prevalence of anti-H5 and -H9 antibodies among poultry market workers is 0.8% and 15.5%, respectively.Citation15 A recent study by Uyeki et al.Citation4 demonstrated that approximately 3% of poultry and non-poultry workers in Vietnam had antibodies against H9N2 viruses, suggesting that mild or subclinical human infections are quite common.
Few studies have compared the biological properties of different H9N2 clades and determined whether certain clades pose a higher risk to mammals. To fill this gap in knowledge, we undertook an extensive characterization and risk-assessment study of H9N2 viruses that included antigenic and genetic analyses, antiviral susceptibility, replication efficiency in primary normal human bronchial epithelial (NHBE) cells, and pathogenicity and transmission in key domestic animals and laboratory models, including ducks, pigs, mice and ferrets.
MATERIALS AND METHODS
Ethics statement
All animal studies were approved by the St Jude Children’s Research Hospital Animal Care and Use Committee (protocol NOs 428 and 513), following the guidelines established by the Institute of Laboratory Animal Resources, approved by the Governing Board of the US National Research Council, and carried out by trained personnel working in a United States Department of Agriculture-inspected Animal Biosafety Level 3+ animal facility.
Cell culture
Madin-Darby canine kidney cells were maintained in culture in minimum essential media supplemented with L-glutamine (2 mM) and 5% fetal bovine serum at 37 °C with 5% CO2. NHBE cells (Lonza, Walkersville, MD, USA) from a single healthy male donor were expanded, cryopreserved and maintained in culture in an air/liquid interface system, as previously described.Citation16 Briefly, cells were plated in 0.33 cm2 transwell inserts (Corning, Corning, NY, USA) and allowed to differentiate, as monitored by transepithelial resistance. An air/liquid interface was established in which the apical surface of the cells was exposed to a humidified 95% air/5% CO2 environment. The basolateral medium was changed every 2 days for a minimum of 5 weeks in culture.
Viral propagation and titers
The H9N2 influenza viruses were propagated in the allantoic cavities of 10-day-old embryonated chicken eggs at 35 °C for 48 h. Viral titers were determined either by injecting 100 µL of 10-fold dilutions of virus into the allantoic cavities of 10-day-old eggs and then calculating the 50% egg infectious dose (EID50) or by calculating the 50% tissue culture infectious dose (TCID50), as described.Citation17 These 50% end points were calculated according to the method of Reed and Muench.Citation18
RNA sequencing
RNA was isolated by using the MagMAXTM-96 AI/ND viral RNA Isolation Kit (Applied Biosystems/Ambion, Austin, TX, USA) with a Kingfisher Flex magnetic particle processor (Thermo Fisher Scientific, Rockford, IL, USA). RNA was eluted in 50 µL nuclease-free water. RT-PCR was carried out for individual fragments (primers available upon request), and Sanger sequencing was performed at the Hartwell Center at St Jude Children’s Research Hospital on an ABI capillary sequencer (Applied Biosystems).
Sequence analysis
The sequences obtained were aligned using Clustal WCitation19 and edited using Bioedit Software version 5.0.9.Citation20 Phylogenetic analysis was performed using the Neighbor-Joining algorithm (Kimura 2-parameter) and the MEGA version 5.05 program.Citation21 The number of bootstrap replications was set to 1000, and bootstrap values greater than 60 were labeled on major tree branches for reference. The virus strains were clustered based on the nucleotides in their sequences, and only dominant clusters were used to infer phylogenetic relationships.
Accession numbers
One hundred and seventy-one sequences have been deposited in Genbank under the accession numbers KF188234–KF188404.
Susceptibility to neuraminidase (NA) inhibitors
NA-inhibition (NAI) assays were performed using viruses standardized to equivalent NA activity and incubated with NAIs at concentrations of 5×10−6–500 µM for 30 min at 37 °C and with 2′-(4-methylumbelliferyl)-α-D-N-acetylneuraminic acid (Sigma-Aldrich, St Louis, MO, USA) as a substrate for 30 min at 37 °C before stopping the reaction and measuring the fluorescence.Citation22 Fluorometric determinations were quantified with a Synergy 2 multimode microplate reader (BioTek, Winooski, VT, USA) and were based on the release of the fluorescent product 4-methyl-umbelliferone using excitation and emission wavelengths of 360 and 460 nm, respectively. The concentration of NAI that reduced NA activity by 50% (IC50), relative to a control mixture with no NAI, was determined by plotting the percent inhibition of NA activity as a function of the compound concentrations calculated using GraphPad Prism 5 software (GraphPad Software, La Jolla, CA, USA).
Susceptibility to adamantanes
The genetic sequences of the transmembrane region of the M2 ion channel protein of the matrix gene were analyzed. Substitutions of residues L26, V27, A30, S31 and G34 were used to screen for predicted amantadine-resistant mutants.
Virulence and transmission in ducks
Male and female mallard hatchlings (1 day old) (Ideal Poultry, Cameron, TX, USA) were purchased, wing banded, provided food and water ad libitum, and raised to 5 weeks of age before use. Ducks (n=2/virus group) were infected via natural route (ocular, nasal, and oral) by inoculation with 106 EID50 units of virus in a total volume of 1 mL phosphate-buffered saline (PBS). Ducks were then monitored daily for signs of disease (i.e., conjunctivitis, depression, neurological abnormalities, diarrhea and disheveled appearance). Oropharyngeal and cloacal samples were collected every 2 days for 9 days post-infection (dpi), and virus titers were assessed by EID50. Seroconversion was monitored by hemagglutination inhibition (HI) assay at 14 dpi.
Viral infection of NHBE cells
Influenza A/California/04/09 (H1N1) or H9N2 viruses were diluted in 0.05% bovine serum albumin–bronchial epithelial cell basal medium (Lonza) to equivalent titers, as determined by TCID50 values. The apical surfaces of NHBE cells were washed 3 times with PBS to remove excess mucus secretion prior to infection. Fully differentiated NHBE cells were infected at multiplicity of infection=0.01 TCID50/cell. Viruses were allowed to adsorb for 1 h at 37 °C and were removed by aspiration. Cells were washed once with 0.9% aqueous NaCl solution (pH 2.2) to remove free infectious virus particles and twice with PBS to adjust the pH. Viruses released apically were harvested at the indicated times postinfection by the apical addition and collection of 300 µL 0.05% bovine serum albumin–bronchial epithelial cell basal medium allowed to equilibrate at 37 °C for 30 min. Samples were stored at –80 °C until assayed for viral titers. Viral titers were obtained by TCID50 analysis on MDKC cells and calculated according to the method of Reed and Muench.Citation18
Pathogenicity in mice
Six- to eight-week-old female BALB/c mice (Jackson Laboratory, Bar Harbor, ME, USA) were lightly anesthetized with isoflurane and intranasally inoculated with PBS or virus in 25 µL PBS, as previously described (n=10 per group).Citation17 Mice were monitored daily for clinical signs of infection and weighed every 24 h post-infection (hpi).Citation23 At 3 and 6 dpi, lungs were harvested (n=3 mice per group/day) and homogenized in 1 mL PBS. Viral titers were then determined by TCID50 analysis.Citation24
Virulence and transmission in pigs
Three-week-old Yorkshire/Hampshire mixed-breed pigs from a private farm were housed in easy-clean plastic tubs (n=4 pigs per tub) inside flexible-film isolators. Pigs were observed and fed twice daily to meet the thermal and nutritional requirements for their age and weight. Water was available ad libitum.Citation25 In each group of four animals, two pigs were removed from the tub, intranasally inoculated with 106 EID50 of virus in 1 mL PBS using a nasal spray, and immediately returned to their tubs. Pigs were weighed, and nasal swabs were collected on the indicated days. Viral titers were determined by TCID50. Sera were collected 14 dpi and treated with receptor destroying enzyme. Homologous and heterologous HI titers were determined using standard methods and 0.5% chicken red blood cells as described.Citation25
Virulence and transmission in ferrets
Three-month-old influenza-seronegative male ferrets (Triple F Farms, Sayre, PA, USA) were lightly anesthetized were isoflurane and intranasally inoculated with 106 TCID50 virus diluted in 0.5 mL PBS (n=2 per group). Ferrets were monitored daily for clinical signs of infection, and nasal washes were collected 2, 4, 6 and 8 dpi. For transmission studies, two ferrets were infected as described and housed individually in separate cages. For assessment of direct contact transmission, a naive ferret was introduced into each of the inoculated ferrets’ cages at 1 dpi. To assess aerosol-contact transmission, a naive ferret was introduced into an adjacent cage separated with a double-sided perforated screen at 1 dpi. The screen allowed airflow from the inoculated ferret’s cage into the aerosol-contact cage without allowing direct or indirect physical contact between the ferrets. Ferrets were monitored daily, and nasal washes were collected every 2 days, starting at 2 dpi. At 20 days post-contact, sera were collected from direct and aerosol contact ferrets to assess potential seroconversion. Ketamine was used to induce sneezing. A/Brisbane/59/2007 H1N1 virus was used as a positive control. Viral titers were determined by TCID50 in Madin-Darby canine kidney cells, as described.Citation24
Risk assessment scoring
A risk assessment score was assigned for each assay on a scale of 0–4, with a score of 4 representing the maximum growth, pathogenicity or transmission phenotype observed. The scores for each virus were then totaled and the virus given an overall risk as follows: low=5–8; intermediate (Int) =8–10 or >10 with Q226; high ≥10; very high >10 with L226.
Statistical analyses
Viral titers in supernatant from indicated H9N2 virus-infected NHBE cell cultures were compared to those in supernatant from A/California/04/2009 (H1N1) virus-infected NHBE cell cultures at each time point by analysis of variance (ANOVA). For the mouse experiments, data were analyzed by two-way ANOVA with virus and dpi as main effects with Student’s t-test used for post hoc comparison of groups. All analyses were performed in PRISM (Graphpad). Differences were considered significant at P<0.05.
RESULTS
Genetic analysis and selection of H9N2 influenza strains for investigation
To choose representative H9N2 influenza viruses, we sequenced and performed phylogenetic analyses on the hemagglutinin (HA) and NA genes of 63 H9N2 viruses (Supplementary Figure S1). These viruses were isolated from wild birds, domestic birds, humans and swine in different regions including Bangladesh, China (including Hong Kong SAR), Korea, Pakistan, United Arab Emirates and the United States, between 1978 and 2011. The viruses formed distinct clades in which the HA and NA genes from the Middle East, Pakistan, and Bangladesh strains clustered with previously identified G1-like viruses (Supplementary Figure S1). Most Chinese isolates clustered with the Y280-like viruses, except for a small cluster of Hong Kong and Shantou viruses. These included two of the candidate vaccine viruses, A/Hong Kong/1073/1999 (HK/1073) and A/Hong Kong/33982/2009 (HK/33982), both of which are G1-lineage viruses, and the chicken/Beijing-like viruses. As previously observed,Citation26,Citation27 the Korean isolates grouped separately, as did the North American isolates. Together, they formed a separate clade of North American/Old Eurasian viruses.
On the basis of the phylogenetic analysis, we selected 12 isolates representing distinct clades, animal species and geographic regions for full genomic analysis and in vitro and in vivo characterization. The isolates included shorebird/Delaware (North American lineage); chicken/Beijing (chicken-Beijing lineage); the following Y280 lineage viruses: duck/Nanchang, chicken/HK/G9, swine/HK, chicken/HK/TP38, chicken/Nanchang and guinea fowl/HK; and the following G1 lineage viruses: HK/33982, HK/1073, chicken/Dubai and quail/Bangl (). H9N2 viruses reassort readily with each other and other influenza subtypes.Citation28,Citation29 In our panel, we identified various gene constellations (Supplementary Table S1), and only four viruses conformed to ‘pure’ classic lineages: shorebird/DE (North American), chicken/Beijing (chicken/Beijing), guinea fowl/HK (Y280) and the G1-lineage virus HK/1073 (Supplementary Figure S2).
Table 1 Influenza H9N2 viruses used in these studies
Antigenic analysis of H9N2 influenza viruses
The availability of effective prophylaxis and treatment options against emerging biological agents are among the important risk assessment factors considered by government agencies for emergency preparedness and biological containment.Citation30 Therefore, the characterization of antigenic cross-reactivity (for vaccine selection) and phenotypic and genotypic susceptibility to currently available antivirals of H9 viruses is of clear public health importance. To monitor the antigenic similarity among the H9N2 viruses, we generated post-infection ferret antisera and performed HI assays using chicken red blood cells. Titers against the homologous viruses ranged from 1∶80 to 1∶20 480 (Supplementary Table S2). The shorebird/DE and chicken/Beijing viruses each reacted poorly against the heterologous antisera, thereby supporting the genetic data that these two viruses belong to distinct lineages. The majority (three of four) of the G1-lineage viruses reacted poorly to heterologous antisera, except the chicken/Dubai virus, which was inhibited by numerous non-G1 antisera. Finally, the Y280 viruses were inhibited by Y280 antisera, with some inhibition by heterologous antisera. Surprisingly, the guinea fowl/HK virus was cross-reactive with all of the antisera.
Antiviral susceptibility of influenza H9N2 viruses
To determine if our current antiviral therapies would be effective if these viruses were to emerge and spread, we examined the susceptibility of H9N2 viruses to the current Food and Drug Administration-approved antiviral drugs, e.g., NA inhibitors and adamantanes (M2 inhibitors). The susceptibility of H9N2 viruses to NA inhibitors oseltamivir carboxylate (the active form of oseltamivir), zanamivir and peramivir, was tested using a fluorescence-based NA enzyme-inhibition assay. All 12 H9N2 viruses were fully susceptible to the three NA inhibitors, with mean IC50 values ranging from less than 0.1 to 0.99 nM (). These IC50 values are similar to those of commonly circulating NA inhibitor-susceptible human H1N1 and H3N2 influenza A viruses.Citation31
To expand our analysis of NA-inhibitor susceptibility, we determined whether known molecular markers of resistance are present in NA sequences of all 63 H9N2 viruses described in Supplementary Figure S1. The screening did not reveal the presence of the NA mutations R292K and E119V/I/A/G, thus confirming the susceptibility of the H9N2 viruses to NA inhibitors. We applied the genotypic assay to detect known adamantane resistance-conferring M2 protein mutations in influenza viruses. Sequence analysis of the 12 viruses revealed that four possessed amino-acid substitutions in the M2 protein (V27A and V27A/S31N) that conferred resistance to adamantanes (). These results indicated that the tested H9N2 viruses, which represent multiple H9N2 lineages, are susceptible to the three NA inhibitors and that this class of antiviral drugs can be used for treatment or prophylaxis of infection caused by influenza H9N2 viruses. Because the level of resistance to adamantanes among the H9N2 viruses tested was lower than that in currently circulating human H1N1 and H3N2 influenza viruses, this class of drugs has little value as a single-agent therapy; however, they may still be useful in combination with other antiviral agents.
Table 2 Susceptibility of H9N2 viruses to NA inhibitors and adamantanes
Characteristics of influenza H9N2 virus infection in ducks
Influenza A H9N2 viruses have been isolated from poultry and wild birds, which serve as the reservoir for influenza viruses throughout many parts of the world. As seen with certain H5N1 viruses, poultry can serve as the intermediate host between wild birds and humans, although these viruses can remain fit for replication in wild birds.Citation32,Citation33 Given that the H5N1 viruses contain internal genes from H9N2 lineages, we were curious to determine if the viruses in our H9N2 panel had similarly maintained tropism for wild bird replication. To compare the pathogenicity and viral shed of the distinct H9N2 clades in ducks, we inoculated mallard ducks (n=2/experimental group) by intraocular, intranasal and oropharyngeal routes with 106 EID50 units of virus and then monitored the animals daily for morbidity and mortality. To determine the degree of viral shedding, we collected cloacal and oropharyngeal swabs every 2 dpi, beginning at 1 dpi. No clinical signs of disease were noted in any duck throughout the experiment (13 days).
Of the H9N2 clades, there appears to be a trend with the North American- and Y280-lineage viruses that mallard ducks were more productively infected as compared to the other viruses (Supplementary Table S3). Mallards inoculated with the North American-lineage shorebird/DE virus shed 103–105.5 EID50/mL predominantly via the cloacal route from 1 to 5 dpi, while only one duck shed via the oropharyngeal route at the limit of detection (i.e., 101 EID50/mL) at 1 dpi. The shorebird/DE virus induced the highest titers and most prolonged shedding. All of the Y280-lineage viruses were shed to differing degrees. The chicken/HK/G9 virus was shed via both the oropharyngeal (1–3 dpi) and cloacal (1 dpi) routes, with titers ranging from 102 to 103.5 EID50/mL. All ducks inoculated with swine/HK or chicken/Nanchang virus shed virus via the respiratory tract only at 1 dpi; the viral titers of these birds were 102.5 EID50/mL and 103.4 EID50/mL, respectively. The duck/Nanchang and guinea fowl/HK viruses were shed via the cloacal route by 50% of the birds at 1 dpi; the virus titers were 103 EID50/mL and 102.5 EID50/mL, respectively. Finally, the chicken/HK/TP38 virus was shed by 1 bird via the oropharyngeal route at the limit of detection on 3 dpi.
In contrast, ducks infected with most of the G1-lineage viruses failed to shed virus at any time post-infection (Supplementary Table S3). The chicken/Beijing virus was shed oropharyngeally at the limit of detection by 1 duck at 1 dpi and the G1-lineage chicken/Dubai virus was shed (102.5 EID50/mL) oropharyngeally by 1 duck at 1 dpi.
To ensure infection, we obtained sera samples at 13 dpi and performed HI assays using chicken red blood cells. Most ducks seroconverted, and 8 of 12 that had detectable virus shedding produced homologous HI titers ranging from 20 to 320 (Supplementary Table S4). Four animals that shed virus did not seroconvert to homologous antigen, but three of them showed HI titers higher than 10 against a heterologous antigen. Cross-reactivity of antigens was particularly prevalent among the Y280-lineage viruses, compared to that of the G1-lineage viruses (Supplementary Table S4, light gray vs. dark gray shading).
In summary, we found differential shedding of the H9N2 viruses in mallard ducks, with the shorebird/DE virus (North American lineage) being shed at the highest level and for the longest duration. The remaining viruses were shed for a minimal duration, if at all. One explanation is that, with the exception of the shorebird/DE virus, which was the only wild-bird isolate assessed, the H9N2 viruses used in our studies have adapted to gallinaceous poultry.
Differential replication of influenza H9N2 viruses in NHBE cells
To assess the potential risk of infection with influenza A H9N2 viruses in mammals, we evaluated the efficiency of viral replication in primary, well-differentiated NHBE cells grown at the air/liquid interface. To determine whether H9N2 influenza viruses infect differentiated NHBE cells, we infected cell cultures apically with the panel of H9N2 viruses or with the human A/California/04/2009 (CA/09) H1N1 virus, at a multiplicity of infection of 0.01 TCID50/cell. Viral titers were then determined at the indicated times after infection (). Within 24 hpi, the human H9N2 viruses HK/1073 and HK/33982 and the quail/Bangl virus had titers similar to the positive control CA/09 virus, while the swine/HK virus had titers that were 2 logs lower. By 48 hpi, the titer of the swine/HK virus was similar to that of the CA/09 and the human H9N2 isolates ().
Figure 1 Replication of H9N2 viruses in primary NHBE cells. Fully differentiated NHBE cells were inoculated with the designated viruses at MOI=0.01. H9N2 viruses of (A) human origin, (B) mammalian or avian origin or (C, D) avian origin were assessed. Viral titers were determined from apical washes taken at the indicated times post-infection. Error bars represent SEM and significance is indicated as *P<0.05; **P<0.01; ***P<0.001, by one-way ANOVA. MOI, multiplicity of infection.

The avian H9N2 influenza viruses we tested can be classified into two distinct groups characterized by their replication kinetics. Both groups replicated to significantly lower titers than did the CA/09 virus at earlier time points (8–24 hpi). The titers of the first group were similar to those of the CA/09 virus () by 48 hpi, while the titers of the second group did not reach levels similar to those of the CA/09 virus until 72 hpi (). Overall, all of the H9N2 viruses tested, regardless of species of origin, replicated in differentiated NHBE cells, though the kinetics and titers differed across the groups, with no consistent correlation to lineage.
Pathogenicity of influenza H9N2 viruses in mice
While the ferret model is considered the gold standard for influenza transmission studies, it is unclear how well it reflects disease pathogenesis. Additionally, the significant cost and outbred nature of the model limit the extent to which large numbers of viruses can be screened efficiently for multiple endpoints. The mouse model has been used as a more affordable, well-characterized proxy system, though it has its own limitations. Many viruses require adaptation to grow efficiently in mice and transmission is rare. Thus, as part of our risk assessment analysis, we included a characterization of our virus panel in mice to compare to data acquired in other models and provide a richer data set of phenotypes in mammalian species. To determine the in vivo mammalian phenotype of the H9N2 viruses, we inoculated six- to eight-week-old BALB/c mice with 103 or 105 TCID50 units of a virus in 25 µL PBS (n=5 mice per viral dose, thus, n=10 mice per isolate). The two doses of the chicken/Nanchang virus used, however, were 104 or 106 TCID50 units. Weight loss was monitored daily for 12 dpi (). Uninfected (PBS-inoculated) and A/Puerto Rico/8/34 (PR8, 102 TCID50)-inoculated mice served as controls. Of the H9N2 viruses tested, only the higher doses of HK/1073, swine/HK, chicken/Beijing (all 105 TCID50), or chicken/Nanchang virus (106 TCID50) induced significant weight loss compared to that seen in the PBS-inoculated mice (), as determined by one-way ANOVA. The mice lost 10%–15% of their initial weight by 7 dpi and were fully recovered by 12 dpi. In contrast, the control PR8-inoculated mice lost 25% of their starting weight by 7 dpi. None of the animals succumbed to infection.
Figure 2 Pathogenicity of H9N2 viruses in mice. Six- to eight-week-old BALB/c mice (n=5 mice/virus were intranasally infected with 105 TCID50 units of the indicated viruses, and weight loss was monitored for 12 dpi H9N2 viruses of (A) human origin, (B) mammalian or avian origin or (C, D) avian origin were assessed. (E) Lungs were collected at 3 and 6 dpi to measure viral titer by TCID50. Dashed line represents the limit of detection of the assay. Error bars represent SEM.
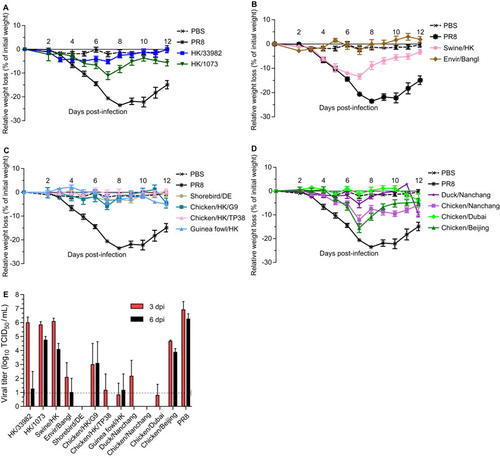
Viral titers were also determined in the lungs of the mice inoculated with the higher dose (105 TCID50) at 3 and 6 dpi (n=3 per time point). The mammalian viruses, HK/33982, HK/1073 and swine/HK, replicated to levels similar to PR8 virus-inoculated animals at 3 dpi (∼106 TCID50/mL) with some clearing by 6 dpi (). In contrast, mice inoculated with most of the avian strains had titers at or below the limit of detection at 3 dpi and were virus-negative by 6 dpi. Only the chicken/Beijing-inoculated mice had viral titers similar to the mammalian viruses, with minimal clearance at 6 dpi (). All mice seroconverted by 14 dpi, as measured by HI assay or enzyme-linked immunosorbent assay. Overall, these studies showed that only a few of the H9N2 viruses induced morbidity in BALB/c mice. Furthermore, only the mammalian viruses and the chicken/Beijing virus replicated above the limit of detection, though all viruses caused seroconversion.
Replication and transmission of influenza H9N2 viruses in pigs
Pigs have been identified as mixing vessels for influenza A viruses of human and animal origin.Citation34 The pandemic virus of 2009 evolved in pigs,Citation35 and the possibility that pigs were involved in the generation of the pandemic viruses of 1918 (H1N1), 1957 (H2N2) and 1968 (H3N2) has not been ruled out.Citation36,Citation37 Therefore, the ability of H9N2 viruses to replicate and shed in pigs increases the public health threat.Citation8 To assess this threat, we intranasally inoculated three-week-old pigs with an H9N2 virus at a dose of 106 EID50 (n=2 pigs per isolate). The inoculated pigs were then housed with two virus-naive pigs to assess infection by direct contact. Pigs were monitored daily for 14 dpi. Nasal swabs were collected every 2 days from 1 to 9 dpi, and viral titers were determined.
None of the pigs showed clinical symptoms (coughing, sneezing or malaise) at any time, and all gained weight with no significant differences between any of the groups (inoculated or direct contact) (). Despite the lack of clinical symptoms, one of the HK/33982 virus-inoculated pigs shed virus, reaching titers of 104 TCID50/mL and 102.7 TCID50/mL on 5 and 7 dpi, respectively. The virus also transmitted to one of the direct contact animals, which shed 103.3 TCID50/mL at 9 days post-contact. None of the pigs seroconverted by 14 dpi/post-contact, including the swine/HK virus-infected animals, as determined by HI assay. These results suggest that H9N2 virus replication is limited in pigs, though the HK/33982 (G1 lineage) virus can replicate to moderate titers and transmit by direct contact.
Figure 3 Pathogenicity of H9N2 viruses in pigs. Three-week-old pigs were inoculated intranasally with an H9N2 virus or were in DC with inoculated pigs (n=2 pigs/group), and weight was monitored every 2 days for 14 dpi. No significant differences in weight gain were identified across the groups. The data represent the average relative weight change compared to the animals’ weights at 0 dpi. Each pig is plotted as a separate line. DC, direct contact.
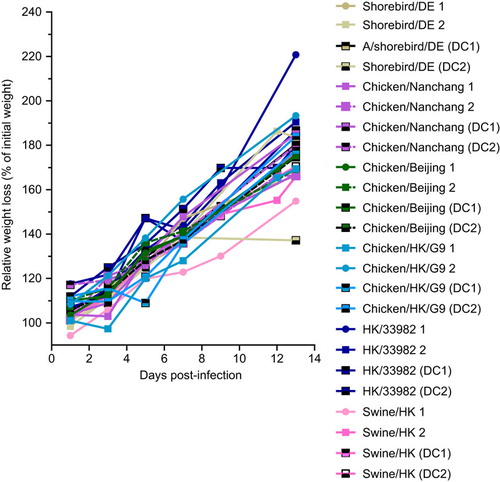
Replication and transmission of influenza H9N2 viruses in ferrets
To assess the ability of H9N2 viruses to replicate and transmit in ferrets, we intranasally inoculated three-month-old ferrets with 106 TCID50 of select H9N2 or A/Brisbane/59/2007 viruses (n=2 ferrets per group).We then assessed viral shedding in nasal washes at 2, 4, 6 and 8 dpi. Consistent with the efficient replication observed in NHBE cells, HK/33982 and HK/1073 viruses replicated to similar titers and kinetics as A/Brisbane/59/2007, the pre-pandemic seasonal H1N1 virus used as a positive control (). Similar replication efficiency and kinetics were observed with the swine/HK virus (). Surprisingly, many of the avian viruses, regardless of lineage, also replicated to similar titers and kinetics as the human-origin H9N2 and seasonal H1N1 viruses (). In fact, a subset of the avian-origin viruses (chicken/HK/G9, guinea fowl/HK, duck/Nanchang, chicken/Nanchang and chicken/HK/TP38) had 1–2 log10 TCID50/mL higher titers than did A/Brisbane/59/2007 at 6 dpi (). Only the shorebird/DE and quail/Bangl viruses replicated to titers that were significantly lower than that of the seasonal H1N1 virus and were cleared faster ().
Figure 4 Replication of H9N2 viruses in ferrets. Two ferrets each were intranasally inoculated with 106 TCID50 units of H9N2 virus of (A) human origin, (B) mammalian or avian origin or (C) avian origin. Brisbane/59 virus was used as a positive control. Viral titers in nasal washes were measured at 2, 4, 6 and 8 dpi. Each bar represents an individual ferret. The limit of detection was 10 TCID50/mL, and titers below that limit are shown at 0.5 log10 TCID50/mL.
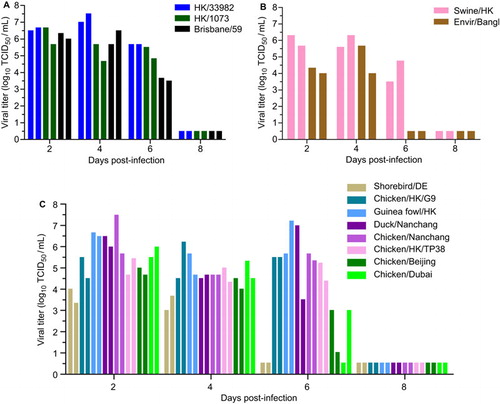
Despite viral replication, very few of the H9N2 viruses transmitted either by direct contact and none appeared to transmit by aerosol contact (). HK/33982 virus transmitted to both of the contact ferrets with similar viral titers as the inoculated animals in one contact (). The other mammalian-origin viruses HK/1073 and swine/HK transmitted to one of the direct contact ferrets (). Overall, human-origin H9N2 viruses displayed the fastest transmission dynamics, with titers observed in contact ferrets as early as 3 days post-contact, followed by the swine-origin and avian-origin viruses. Of the avian viruses, only chicken/HK/G9 transmitted to both of the direct contact ferrets; however, transmission was slightly delayed compared to mammalian-origin viruses, and the viral titers were lower than that in the inoculated animals (). Regardless, all of the infected direct contact ferrets seroconverted by 20 days post-contact (HI: 80–320; Supplementary Table S5). None of the viruses tested spread via aerosol contact, as evidenced by the lack of viral shedding () and the lack of seroconversion 20 days post-contact (Supplementary Table S5). These data contrast with that of the pre-pandemic seasonal H1N1 virus, which transmitted to 100% of the direct and aerosol contact animals ().
Figure 5 Transmission of H9N2 viruses in ferrets via direct or aerosol contact. Ferrets were infected intranasally with 106 TCID50 of virus (n=2 ferrets/virus) and housed in individual cages. H9N2 viruses of (A) human origin, (B) mammalian or avian origin or (C) avian origin were assessed. Brisbane/59 virus was used as a positive control. To assess direct contact transmission, a naive ferret was added to each cage containing an infected ferret, and to assess aerosol-contact transmission, another naive ferret was introduced into a cage adjacent to each infected ferret. Both naive ferrets were introduced at 1 dpi. Virus titers in nasal washes were measured 1, 3, 5, 7, 9, 11 and 13 days post-contact. Each bar represents an individual ferret. The limit of detection was 10 TCID50/mL. Titers below the detection limit are shown at 0.5 log10 TCID50/mL.
In summary, regardless of lineage, many of the H9N2 viruses replicated in inoculated ferrets, but only those of mammalian origin efficiently transmitted by direct contact. None of the H9N2 viruses tested transmitted by aerosol contact. Intriguingly, the chicken/HK/G9 virus was the only virus of non-mammalian origin that transmitted to both direct contact ferrets, though with delayed kinetics.
Risk assessment of H9N2 influenza viruses
To quantify our outcomes and rank the relative risk posed by each virus tested, we scored the results of each assay on an arbitrary fitness scale from 0 to 4, with 0 corresponding to the virus with the lowest measurement in an assay and 4 corresponding to viruses with the highest levels of replication or transmission (). Not surprisingly, the two human viruses of G1 lineage (HK/33982 and HK/1073) generated the highest cumulative score. This is consistent with the finding that viruses directly isolated from humans represent the greatest risk. However, the strength of this approach is demonstrated by the next two highest scores, swine/HK and chicken/HK/G9, neither of which would have been predicted to score well based on phylogeny alone. These viruses were moderately fit in NHBE cells and ferret experiments and transmitted via direct contact in ferrets.
Table 3 Risk assessment of H9N2 virus strains based on biological activity
DISCUSSION
H9N2 viruses are considered a pandemic risk, given their endemicity, ability to infect numerous species, repeated zoonotic infections of humans and lack of population-wide immunity. Recognizing this risk, the World Health Organization monitors the continual evolution of the H9N2 clades in order to provide candidate vaccine viruses for public health purposes. Most of the H9N2 viruses studied here appear to be adapted to gallinaceous poultry and replicate poorly in ducks, with the exception of the shorebird/DE strain. This result suggests that, unlike H5 viruses, H9-lineage viruses rapidly lose fitness in shorebirds once the virus adapts to terrestrial poultry or mammalian species.Citation38 A previous report found that a duck-derived H9N2 virus required experimental adaptation by serial passage through quail and chicken lungs in order to replicate efficiently in those species.Citation39 The shorebird-derived virus in our study had one of the lowest risk-assessment scores, driven largely by its failure to grow efficiently in the mammalian models.
The two isolates of human origin tested in the present study grew to higher titers in NHBE cells, appeared more virulent and/or more transmissible in the various animal models and ranked highest in our risk-assessment studies. The only consistent amino acid mutations that distinguished them from the other 10 tested viruses were as follows: I139T, K/A149R, D/N153G, D/N196Y and K321N in HA; S/G60N in non-structural protein 2 (NS2); and I/V147M and I401V in polymerase basic 2 (PB2) (data not shown). None of the isolates that we studied had a PB2 E627K substitution, but the HK/33982 virus contains the PB2 D701N mutation, a molecular marker for virulence and host adaptation (avian to mammalian).Citation40 The HA 226 position (in the receptor-binding pocket) has been previously associated with transmission of H9N2 in ferrets and described as a marker for increased pandemic risk.Citation41,Citation42 The viruses tested in the present study, however, show that this residue is not strictly determinative; HK/33982 and chicken/Beijing both carry the putatively non-permissive Q226 residue but transmitted as efficiently in ferrets as did the viruses carrying the permissive L226 residue ().
Previous studies of H9 and other viruses with pandemic potential have often focused on the possibility of the surface proteins of those strains being acquired by circulating human viruses in a reassortant event. Our studies strongly suggest that the internal genes of diverse H9-lineage viruses are fit (to various degrees) for replication in mammalian models and human cells. As such, these internal segments may also contribute to emerging pandemics. This possibility has recently been realized in the emergence of the H7N9 strains infecting humans in mainland China.Citation2 Those viruses contain surface proteins previously found mostly in avian influenza viruses with internal genes of H9N2-lineage viruses. Intriguingly, none of the H9-lineage viruses that have been characterized in public databases appear to cluster closely with all of the internal gene segments from the H7N9 lineage. This finding indicates that an as-yet-undocumented event occurred among H9 strains to generate the internal gene cassette of the novel H7N9 lineage.
Although swine viruses are often more intensely scrutinized due to the status of the pig as a mixing vessel, the chicken/HK/G9 virus stood out in comparison with the four other chicken viruses in our analysis, in terms of it growing reasonably well in multiple mammalian model systems. Understanding the genetic changes in this virus that improved its replication phenotype is an area of current investigation.
In summary, we have characterized a diverse array of H9-lineage influenza viruses, discovering a spectrum of fitness for replication and transmission in humans and relevant mammalian models. No cluster of sequence signatures fully predicted the fitness profiles observed, demonstrating the use and need for in vivo studies, as performed here. These measurements allowed us to quantify the relative risk of each virus. Multiple viruses isolated from non-human sources had moderate-to-high levels of fitness in mammalian models. This finding emphasizes the necessity for careful, continual and thorough surveillance paired with risk-assessment of circulating influenza viruses.
Author contributions
The SJCEIRS H9 Working group is: Tatiana Baranovich,1 Olga Bridges,1 Andrew Burnham,1 David Carey,2 Troy D. Cline,1 Jeri C. Crumpton,1 Jennifer DeBeauchamp,1 Susu Duan,3 Mariette F. Ducatez,1 Husni Elbahesh,1 Thomas P. Fabrizio,1 Heather L. Forrest,1 John Franks,1 Pamela Freiden,1 Elena A. Govorkova,1 Yi Guan,4,5 Trushar Jeevan,1 Jeremy C. Jones,1 Bryan S. Kaplan,1 Erik A. Karlsson,1 Lisa A. Kercher,2 Zeynep A. Koçer,1 Scott Krauss,1 Beth Little,2 Bindumadhav M. Marathe,1 Jennifer L. McClaren,3 Victoria A. Meliopoulos,1 Kevin B. O'Brien,1 Thomas H. Oguin III,3 Christine M. Oshansky,3 J.S. Malik Peiris,4,5 Kristi Prevost,1 Adam Rubrum,1 Charles J. Russell,1 Catherine J. Sanders,3 Patrick Seiler,1 Bradley J. Seufzer,1 Karthik K. Shanmuganatham,1 Stephanie Sonnberg,1 Terri D. Stoner,1 Jasmine Turner,1 Lee Ann Van de Velde,1,3 Nicholas C. Van de Velde,1,3 Richard J. Webby,1 Robert G. Webster,1 Sook-San Wong,1 Sun-Woo Yoon,1 Mark Zanin,1 Hassan Zaraket,1 Stacey Schultz-Cherry, Paul G. Thomas3. Z. Kocer led a team of J. Franks, O. Bridges, L. McClaren, K. Shanmuganatham, L. van de Velde, P. Freiden and B. Seufzer to grow and characterize the viral stocks. T. Baranovich and C. Oshansky led a team of S-S. Wong, M. Zanin, V. Meliopoulos, B. Kaplan, H. Elbahesh, C. Sanders, H. Zaraket, and S-W Yoon to perform the human epithelial cell experiments. E. Govorkova led a team of T. Stoner, S. Duan and B. Marathe to conduct the antiviral susceptibility studies. M. Ducatez and S-S. Wong led a team of T. Fabrizio, H. Forrest, J. Jones, B. Kaplan, Z. Kocer, A. Rubrum, K. Shanmuganatham, S. Sonnberg, J. Turner, S-W. Yoon and M. Zanin to perform all viral sequencing. H. Zaraket led a team of L. Kercher, E. Karlsson, S-S. Wong, L. McClaren, T. Jeevan, C. Russell, K. Shanmuganatham, S-W. Yoon, J. Jones, T. Cline, S. Duan, T. Baranovich, B. Kaplan, D. Carey, B. Little, and B. Marathe for the ferret studies. S. Sonnberg led a team of J. Jones, J. Franks, P. Seiler, S-S. Wong, S-W Yoon, P. Thomas, K. Shanmuganatham, H. Zaraket, T. Baranovich, B. Little, L. Kercher, D. Carey, J. Turner, T. Jeevan, S. Duan, T. Fabrizio, J. DeBeauchamp, and J-C Crumpton for the duck and pig studies. E. Karlsson led a team of A. Burnham, T. Cline, J. Crumpton, H. Elbahesh, L. Kercher, K. O’Brien, T. Oguin, C. Sanders, N. Van de Velde, and H. Zaraket to perform the mouse studies. M. Peiris and Y. Guan helped choose and provide viruses for study. R. Webby, R. Webster, E. Govorkova, C. Russell, P. Thomas, and S. Schultz-Cherry helped design and coordinate experiments. S. Schultz-Cherry and P. Thomas conceived of the project and wrote the paper with Z. Kocer, M. Ducatez, S-S. Wong, T. Barnaovich, C. Oshansky, E. Karlsson, S. Sonnberg, and H. Zaraket.
Supplementary information
Download MS Word (13.2 KB)Supplementary information
Download MS Word (48.3 KB)Supplementary information
Download MS Power Point (615 KB)Acknowledgments
We would like to thank Pamela McKenzie (St Jude Children’s Research Hospital) for administrative support and helpful conversations and Angela McArthur (St Jude Children’s Research Hospital) for manuscript editing. This work was supported by NIH/NIAID Contract HHSN266200700005C (St Jude Center of Excellence for Influenza Research and Surveillance) and American Lebanese Syrian Associated Charities at St Jude.
Supplementary information for this article can be found on Emerging Microbes & Infections' website (http://www.nature.com/EMI/).
Notes
Abbreviations: R, amantadine-resistant virus; S, amantadine-susceptible virus.
NA inhibition assay used 2′-(4-methylumbelliferyl)-α-D-N-acetylneuraminicacid (MUNANA) as a substrate at a final concentration of 100 µM. Results are the mean values (IC50, nM) of three experiments±standard deviation (SD).
Susceptibility to adamantanes was based on the presence of a substitution of one of five amino acids (positions 26, 27, 30, 31 and 34) within the transmembrane domain of the M2 protein. Amantadine resistance-associated mutations are shown in parenthesis.
Highest titers, the fastest kinetics indicated by score=4.
Highest titers, the longest duration indicated by score=4.
Seroconversion (SC) in ferrets was determined by HI assay.
1=direct contact (DC) transmission in one animal; 2=DC transmission in two animals; 3=aerosol-contact (AC) transmission in one animal; 4=AC transmission in two animals; 0, no transmission; ND, not done.
Seroconversion (SC) in mice was determined by enzyme-linked immuno sorbent assay.
1=2 ducks shed for 1 day; 2=2 ducks shed for 2 days.
Categories of risk were defined as follows: low=5–8; intermediate (Int) =8–10 or >10 with Q226; high ≥10; very high >10 with L226.
- Centers for Disease Control and Prevention. Notes from the field: highly pathogenic avian influenza A (H7N3) virus infection in two poultry workers—Jalisco, Mexico, July 2012 (n.d.). Atlanta: CDC, 2012. Available at http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6136a4.htm?s_cid=mm6136a4_w (accessed 17 May 2013).
- Li Q, Zhou L, Zhou Met al.Preliminary report: epidemiology of the avian influenza A (H7N9) outbreak in China. N Engl J Med; e-pub ahead of print 24 April 2013; doi: 10.1056/NEJMoa1304617.
- Centers for Disease Control and Prevention (CDC).Emergence of avian influenza A(H7N9) virus causing severe human illness—China, February–April 2013. MMWR Morb Mortal Wkly Rep2013;62:366–371.
- Uyeki TM, Nguyen DC, Rowe Tet al.Seroprevalence of antibodies to avian influenza A (H5) and A (H9) viruses among market poultry workers, Hanoi, Vietnam, 2001. PloS ONE2012;7:e43948.
- Cheng VC, Chan JF, Wen Xet al.Infection of immunocompromised patients by avian H9N2 influenza A virus. J Infect2011;62:394–399.
- Liu D, Shi W, Shi Yet al.Origin and diversity of novel avian influenza A H7N9 viruses causing human infection: phylogenetic, structural, and coalescent analyses. Lancet2013;381:1926–1932.
- Guan Y, Shortridge KF, Krauss S, Webster RG.Molecular characterization of H9N2 influenza viruses: were they the donors of the “internal” genes of H5N1 viruses in Hong Kong? Proc Natl Acad Sci USA1999;96:9363–9367.
- Yu H, Zhou YJ, Li GXet al.Genetic diversity of H9N2 influenza viruses from pigs in China: a potential threat to human health? Vet Microbiol2011;149:254–261.
- World Health Organization. Antigenic and genetic characteristics of A(H5N1), A(H7N3), A(H9N2) and variant influenza viruses and candidate vaccine viruses developed for potential use in human vaccines (n.d.).Geneva: WHO, 2013. Available at http://www.who.int/influenza/vaccines/virus/characteristics_virus_vaccines/en/ (accessed 17 May 2013).
- Amirsalehy H, Nili H, Mohammadi A.Can dogs carry the global pandemic candidate avian influenza virus H9N2? Aust Vet J2012;90:341–345.
- Karlsson EA, Engel GA, Feeroz MMet al.Influenza virus infection in nonhuman primates. Emerg Infect Dis2012;18:1672–1675.
- Lee DC, Mok CK, Law AH, Peiris M, Lau AS.Differential replication of avian influenza H9N2 viruses in human alveolar epithelial A549 cells. Virol J2010;7:71.
- Xu KM, Li KS, Smith GJet al.Evolution and molecular epidemiology of H9N2 influenza A viruses from quail in southern China, 2000 to 2005. J Virol2007;81:2635–2645.
- Shaw M, Cooper L, Xu Xet al.Molecular changes associated with the transmission of avian influenza a H5N1 and H9N2 viruses to humans. J Med Virol2002;66:107–114.
- Wang M, Fu CX, Zheng BJ.Antibodies against H5 and H9 avian influenza among poultry workers in China. N Engl J Med2009;360:2583–2584.
- Krunkosky TM, Martin LD, Fischer BM, Voynow JA, Adler KB.Effects of TNFalpha on expression of ICAM-1 in human airway epithelial cells in vitro: oxidant-mediated pathways and transcription factors. Free Radic Biol Med2003;35:1158–1167.
- Cline TD, Karlsson EA, Freiden Pet al.Increased pathogenicity of a reassortant 2009 pandemic H1N1 influenza virus containing an H5N1 hemagglutinin. J Virol2011;85:12262–12270.
- Reed LJ, Muench H.A simple method of estimating fifty per cent endpoints. Am J Epidemiol1938;27:493.
- Chenna R, Sugawara H, Koike Tet al.Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res2003;31:3497–3500.
- Hall T.BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser1999;41:95–98.
- Tamura K, Peterson D, Peterson Net al.MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol2011;28:2731–2739.
- Hurt AC, McKimm-Breschkin JL, McDonald Met al.Identification of a human influenza type B strain with reduced sensitivity to neuraminidase inhibitor drugs. Virus Res2004;103:205–211.
- Morton DB.A systematic approach for establishing humane endpoints. ILAR J2000;41:80–86.
- Szretter KJ, Balish AL, Katz JM.Influenza: propagation, quantification, and storage. Curr Protoc Microbiol2006; Chapter 15; doi: 10.1002/0471729256.mc15g01s3.
- Federation of Animal Science Societies (FASS). Guide for the Care and Use of Agricultural Animals in Research and Teaching, Third Edition, January 2010.Champaign: FASS, 2010. Available at http://www.fass.org/page.asp?pageID=216 (accessed 17 May 2013).
- Lee DH, Song CS.H9N2 avian influenza virus in Korea: evolution and vaccination. Clin Exp Vaccine Res2013;2:26–33.
- Lee CW, Song CS, Lee YJet al.Sequence analysis of the hemagglutinin gene of H9N2 Korean avian influenza viruses and assessment of the pathogenic potential of isolate MS96. Avian Dis2000;44:527–535.
- Bi Y, Lu L, Li Jet al.Novel genetic reassortants in H9N2 influenza A viruses and their diverse pathogenicity to mice. Virol J2011;8:505.
- Aamir UB, Wernery U, Ilyushina N, Webster RG.Characterization of avian H9N2 influenza viruses from United Arab Emirates 2000 to 2003. Virology2007;361:45–55.
- Centers for Disease Control and Prevention. Biosafety in Microbiological and Biomedical Laboratories (BMBL).5th ed.Atlanta: CDC, 2009. Available at http://www.cdc.gov/biosafety/publications/bmbl5/ (accessed 6 August 2013).
- Okomo-Adhiambo M, Sleeman K, Lysén Cet al.Neuraminidase inhibitor susceptibility surveillance of influenza viruses circulating worldwide during the 2011 Southern Hemisphere season. Influenza Other Respi Viruses2013;7:645–658.
- Hulse-Post DJ, Sturm-Ramirez KM, Humberd Jet al.Role of domestic ducks in the propagation and biological evolution of highly pathogenic H5N1 influenza viruses in Asia. Proc Natl Acad Sci USA2005;102:10682–10687.
- Sturm-Ramirez KM, Hulse-Post DJ, Govorkova EAet al.Are ducks contributing to the endemicity of highly pathogenic H5N1 influenza virus in Asia? J Virol2005;79:11269–11279.
- Ma W, Kahn RE, Richt JA.The pig as a mixing vessel for influenza viruses: human and veterinary implications. J Mol Genet Med2008;3:158–166.
- Smith GJ, Vijaykrishna D, Bahl Jet al.Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza A epidemic. Nature2009;459:1122–1125.
- Guan Y, Vijaykrishna D, Bahl Jet al.The emergence of pandemic influenza viruses. Protein Cell2010;1:9–13.
- Smith GJ, Bahl J, Vijaykrishna Det al.Dating the emergence of pandemic influenza viruses. Proc Natl Acad Sci USA2009;106:11709–11712.
- Kim JK, Negovetich NJ, Forrest HL, Webster RG.Ducks: the “Trojan horses” of H5N1 influenza. Influenza Other Respi Viruses2009;3:121–128.
- Hossain MJ, Hickman D, Perez DR.Evidence of expanded host range and mammalian-associated genetic changes in a duck H9N2 influenza virus following adaptation in quail and chickens. PloS ONE2008;3:e3170.
- Gabriel G, Herwig A, Klenk HD.Interaction of polymerase subunit PB2 and NP with importin alpha1 is a determinant of host range of influenza A virus. PLoS Pathog2008;4:e11.
- Wan H, Perez DR.Amino acid 226 in the hemagglutinin of H9N2 influenza viruses determines cell tropism and replication in human airway epithelial cells. J Virol2007;81:5181–5191.
- Wan H, Sorrell EM, Song Het al.Replication and transmission of H9N2 influenza viruses in ferrets: evaluation of pandemic potential. PloS ONE2008;3:e2923.