Abstract
Hepatitis C virus (HCV) infection affects approximately 3% of the world’s population and causes chronic liver diseases, including liver fibrosis, cirrhosis, and hepatocellular carcinoma. Although current antiviral therapy comprising direct-acting antivirals (DAAs) can achieve a quite satisfying sustained virological response (SVR) rate, it is still limited by viral resistance, long treatment duration, combined adverse reactions, and high costs. Moreover, the currently marketed antivirals fail to prevent graft reinfections in HCV patients who receive liver transplantations, probably due to the cell-to-cell transmission of the virus, which is also one of the main reasons behind treatment failure. HCV entry is a highly orchestrated process involving initial attachment and binding, post-binding interactions with host cell factors, internalization, and fusion between the virion and the host cell membrane. Together, these processes provide multiple novel and promising targets for antiviral therapy. Most entry inhibitors target host cell components with high genetic barriers and eliminate viral infection from the very beginning of the viral life cycle. In future, the addition of entry inhibitors to a combination of treatment regimens might optimize and widen the prevention and treatment of HCV infection. This review summarizes the molecular mechanisms and prospects of the current preclinical and clinical development of antiviral agents targeting HCV entry.
Introduction
Hepatitis C virus (HCV) belongs to the family Flaviviridae and infects more than 180 million people worldwide. HCV infection is considered as a major public health problem and consumes millions of dollars in medical expenses every year.Citation1,Citation2 HCV has a total of seven identified genotypes, with more than 50 subtypes and millions of quasispecies. The high variability and complexity of the virus make it difficult to manufacture effective prophylactic or therapeutic vaccines to prevent the pathogen from spreading. Approximately 70% of acutely infected patients will ultimately develop chronic infections despite the implementation of advanced medical care and intervention.Citation3 Due to its biological characteristics, HCV infection is one of the leading causes of liver-associated diseases, such as cirrhosis, steatosis, and hepatocellular carcinoma, whose end-stage patients require liver transplantation to stay alive.Citation4 Unfortunately, the reinfection of a graft is difficult to avoid due to the lack of preventive strategies.Citation5
The previously recommended treatment for HCV infection was a combination therapy consisting of PEGylated interferon alpha and ribavirin.Citation3 In recent years, HCV treatment has undergone a groundbreaking evolution. Direct-acting antivirals (DAAs), such as protease inhibitors (boceprevir or telaprevir in 2011), have revolutionized the current status of HCV treatment. Triple-combination therapy improves sustained virological response (SVR) rates in naive genotype 1 patients by more than 70%. However, the two first-generation protease inhibitors that are typically used easily lead to the development of drug-resistant variants, and concomitant adverse reactions such as fatigue or anemia unavoidably reduce patient compliance with the regimen.Citation4,Citation6,Citation7 A second-wave first-generation protease inhibitor, simeprevir, and a nucleotide analog, sofosbuvir, were approved by the United States in 2013 via the FDA and by Europe in 2014 for the treatment of hepatitis C (HC).Citation7,Citation8,Citation9 In October 2014, the use of ledipasvir/sofosbuvir was approved by the FDA, and in December, an interferon-free regimen including an ombitasvir/paritaprevir/ritonavir combination tablet and dasabuvir was also approved for the treatment of genotype 1 patients.Citation10,Citation11,Citation12,Citation13,Citation14,Citation15 A number of other DAAs and host-targeted agents (HTAs) are undergoing clinical trials. Daclatasvir is an NS5A inhibitor and is currently being evaluated in an advanced clinical trial as a component of a combination therapy.Citation16 In fact, the combination of daclatasvir and asunaprevir (an HCV NS3/4A protease inhibitor) has been approved for the treatment of genotype 1 patients in Japan.Citation16 The future of HCV therapy is likely to be consist of interferon-free regimens with pan-genotypic activity, higher antiviral efficiencies, shorter treatment durations, and fewer adverse reactions. The emerging novel antivirals should optimize the treatment options, especially for difficult-to-treat patients, such as those who are suffering from advanced liver diseases or other co-infections and who have poor response rates to current regimens.Citation17,Citation18
HCV entry represents the beginning of viral infection, which is highly orchestrated and essential in initiating viral infection and spread. HCV entry includes the initial recruitment and attachment of the virus to hepatocytes, post-binding interactions with host entry factors, clathrin-mediated endocytosis, and a final low pH-triggered membrane fusion to release viral RNA into the cytosol (). The blocking of viral entry can efficiently eradicate HCV infection at the very first step, before viral genomes start to emerge, and might prevent cell-to-cell transmission, which is also required for viral spread. The current antiviral agents that are on the market or being evaluated in clinical trials mainly focus on targeting HCV nonstructural protein maturation or viral RNA synthesis. Although the currently used cocktail therapy is believed to cure more than 90% of infected patients, the appearance of viral resistance, null responders or treatment failure, superimposed with the adverse effects caused by the drugs, is still a major limitation that must be resolved.Citation19 As an RNA virus, HCV very easily develops a resistance to antiviral treatments due to its error-prone replication property. Most entry inhibitors target host components, such as receptors or key enzymes, which are required for HCV entry and definitely have high genetic barriers to resistance due to their conserved nature. Therefore, these inhibitors tend to not only have pan-genotypic activity against virus infection but also have a greater risk of simultaneously causing cellular toxicity. Moreover, as the initial step of the viral life cycle has been blocked, entry inhibitors also have prophylactic properties. Because entry inhibitors target a different stage of the viral life cycle than the currently used anti-HCV drugs, they might have a synergistic effect when combined with the current regimens, especially with DAAs, providing multiple novel targets and new insight into antiviral strategies and complementary existing antiviral interventions, such that viral clearance may finally be achieved. This article reviews the entry inhibitors that are currently in development.
Figure 1 HCV entry into hepatocytes and antiviral agents targeting entry factors. The HCV lipoviral particle (LVP) is recruited and binds to glycosaminoglycans and low-density lipoprotein receptor on host cells. After binding, the virions interact with a series of entry factors. SRB1 plays a role in both binding and post-binding. CD81 interacts with HCV E2, forms a complex with claudin-1 (CLDN1), and mediates HCV movement to the tight-junction areas. This process is regulated by the receptor tyrosine kinase (RTK) family, including epidermal growth factor receptor (EGFR) and ephrin receptor A2 (EphA2). The virions internalize into host cells by clathrin-mediated endocytosis. Transferrin receptor 1 (TfR1) facilitates viral entry after CD81, possibly during HCV particle endocytosis. Niemann Pick C1-like 1 (NPC1L1) plays an important role in cholesterol transportation and is a cofactor for HCV entry during post-binding steps. Low pH-dependent membrane fusion between endosome and HCV particle. Red words and lines indicate the antiviral agents targeting different stages and factors of HCV entry.
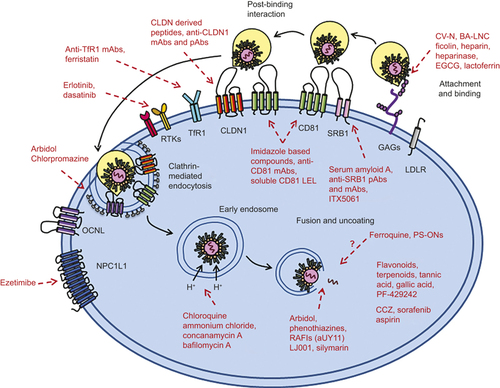
HCV entry into hepatocytes
Initial low-affinity attachment and binding
In vivo, circulating HCV particles reach the basolateral surfaces of hepatocytes, where the virus first binds to several receptors with low affinity, allowing it to become concentrated on the host cells’ surfaces to enable subsequent interactions with other essential entry factors (). These attachment factors include glycosaminoglycans (GAGs) on heparan sulfate (HS) and low-density lipoprotein receptor (LDLR). Both of these receptors are able to interact with viral envelope proteins and apoE on lipo-virion particles (LVPs).Citation20,Citation21,Citation22 HCV exists as LVP with LDL and VLDL in the circulatory systems of chronically infected patients. Recent studies have demonstrated that LDLR plays an important role in HCV attachment to target cells.Citation23 The knockdown of this receptor by small interfering RNA (siRNA) potently reduces virus infectivity, and a soluble form of LDLR can impair virus binding by interacting with HCV particles.Citation24 Although this receptor is dispensable for lipid/cholesterol-free HCVpp entry, productive HCV infection, including viral entry and replication, requires the involvement of LDLR.Citation24,Citation25
The lectin cyanovirin-N (CV-N) is a type of carbohydrate-binding agent that has potent antiviral activity against HCV. This small compound impairs virus binding by interacting with viral envelope glycoproteins at their high-mannose oligosaccharides ().Citation26 Recent studies have identified boronic acid-modified lipid nanoparticles (BA-LNC) as potent inhibitors of HCV entry through a mechanism similar to that of lectins. BA-LNCs could be used as a pseudolectin-based therapeutic agent to develop novel HCV entry inhibitors.Citation27 Ficolins are a type of serum protein related to collectins.Citation28 Neutralizing concentrations of L-ficolin can be found in the sera of HCV-infected patients. Additionally, a recent study shows that recombinant human L-ficolin can neutralize HCV particles directly and inhibit virus attachment by neutralizing the viral glycoproteins E1 and E2.Citation29 Heparin is a structural analog of HS that can competitively inhibit virus binding to host cells. A series of heparin-derived molecules are undergoing evaluation for their potential to serve as anti-HCV agents.Citation21,Citation90 Heparanase, an enzyme that degrades HS on host cell surfaces, can also impede both HCV E2 and HCVcc binding to host cell surfaces ().Citation21
Table 1 The process of viral entry and targets for antiviral agents with their development stage
Epigallocatechin gallate (EGCG) and its derivatives are natural polyphenol compounds that are abundant in green tea extracts and have long been considered to regulate lipid metabolism, thereby having the potential to affect a variety of diseases.Citation91 Studies suggest that EGCG and its derivatives impair virus binding to the host cell by interfering with virion E1/E2 function and simultaneously blocking cell-to-cell transmission in vitro ().Citation30,Citation31,Citation32 Additionally, limited sampling estimates of EGCG in HCV patients suggest that a single oral dose of up to 400 mg of this green tea extract is safe and well tolerated.Citation92 Additional experiments are needed to evaluate the possibility of using this compound as a candidate for anti-HCV therapy.
Lactoferrin (LF) is an iron-binding glycoprotein that belongs to the transferrin family; it is abundant in milk and most biological fluids.Citation93 The antiviral activity of LF is relatively well understood. LF is thought to function by directly interfering with HCV particles to prevent their attachment to host cells both in vitro and in vivo.Citation33 Bioactive peptides, such as the N-lobe or C-lobe of LF, also inhibit virus infection.Citation35 Among all species, camel lactoferrin (cLF) shows the most effective antiviral property and is now being evaluated in a clinical trial ().Citation34
The p7 is a polypeptide of the HCV-encoded protein in the endoplasmic reticulum membrane and is essential for infectious viral production in vivo.Citation94,Citation95 A recent study revealed that a p7 ion channel-derived peptide H2-3 potently inhibits HCV entry by directly affecting virus binding to the cell surface and interfering with the virus–host interaction ().Citation36
Post-binding interactions with specific entry factors
Subsequent to virus binding, LVPs start to form contacts with a series of entry factors on host cells. Targeting these relatively conserved factors, which are indispensable for the early life cycle of the virus, builds up genetic barriers against antiviral agents. HCV entry requires several host factors, including the tetraspanin molecule CD81, scavenger receptor class B type 1 (SRB1), the tight junction (TJ) proteins claudin-1 (CLDN1) and occludin (OCLN), transferring receptor 1 (TfR1), the receptor tyrosine kinases (RTKs) epidermal growth factor receptor (EGFR) and ephrin receptor A2 (EphA2), and Niemann-Pick C1-like 1 (NPC1L1) cholesterol uptake receptor ().
CD81 was the first of these factors to be identified and is the best understood HCV entry factor. It is a ubiquitously expressed, 26 kDa transmembrane protein that consists of a small extracellular loop (SEL) and a large extracellular loop (LEL).Citation96,Citation97,Citation98 The CD81 LEL is believed to interact with the HCV E2 protein, which contributes directly to HCV infection. Imidazole-based compounds simulate the D-helix of CD81 and are relatively safe small molecule inhibitors of HCV. They selectively abrogate the function of CD81 during HCV entry, while the remaining physiological functions of CD81 are basically preserved ().Citation37 Specific anti-CD81 monoclonal antibodies (mAbs), such as JS-81 or the newly developed K04, counteract E2-CD81 interactions, interfering with viral entry during a post-binding process and inhibit HCV infection in humanized mice ().Citation38,Citation39,Citation40,Citation41,Citation42,Citation43 A soluble recombinant form of CD81 LEL shows effective anti-HCV activity and is able to inhibit the entry of HCVpp, HCVcc, and serum-derived HCV, as well as HCV infection, in vivo ().Citation42,Citation44,Citation45,Citation46,Citation47,Citation48 However, because CD81 is widely distributed in all tissues, the toxicity issues that are associated with using CD81-based antibodies or compounds should be evaluated carefully.
SRB1 is a horseshoe-shaped glycoprotein that is closely related to lipid metabolism. SRB1 binds diverse lipoproteins, including HDL, LDL, and oxLDL and plays key roles in bidirectional cholesterol transport, possibly modulating HCV entry into host cells.Citation99,Citation100 The extracellular loop of SRB1 interacts with the HCV E2 HVR1 region and is required for viral entry during both binding and post-binding steps.Citation51,Citation101 Serum amyloid A (SAA) is an acute-phase protein that is produced by the liver.Citation102,Citation103 There is a close relationship between SAA and HDL in modulating HCV infectivity.Citation50 SRB1 binds to and internalizes SAA, and SAA inhibits HCV entry by interacting with the virus ().Citation49,Citation50 Antibodies targeting SRB1 inhibit virus infection and spread both in vitro and in a humanized mouse model ().Citation51,Citation52,Citation53,Citation54 The preclinical compound ITX5061 is a small-molecule antiviral that impedes the uptake of HDL through SRB1, thus blocking the uptake of viral particles.Citation55,Citation56 An in vitro study indicated that ITX5061 functions synergistically with DAAs, making it a promising candidate for future combination therapy.Citation57 This compound has just finished evaluation in a phase Ib study and is now undergoing a phase II clinical trial in HCV-positive patients ().Citation58
CLDNs and OCLNs are components of TJs. CLDN1 is believed to form a complex with CD81 and to contribute to efficient HCV internalization.Citation104,Citation105,Citation106 The expression of CLDN1, CD81, and SRB1 can confer HCVpp entry into HEK293 cells.Citation107 CLDN1 is highly expressed in hepatocytes, making it an ideal and promising target for the development of specific prophylactic antiviral agents.Citation108 A human CLDN1-derived peptide (CL58) was screened out from an overlapping peptide library and was confirmed to have antiviral activity during a late post-binding step without affecting TJ integrity ().Citation59 CLDN1 mAbs and pAbs show potent inhibitory effects on HCV infection, probably because they can neutralize E2-CD81-CLDN1 associations with low toxicity in primary human hepatocytes (PHHs) and humanized mice ().Citation60,Citation61,Citation62 However, a recent study has suggested that broad CLDN tropism permits its escape from CLDN1 Abs because the virus can utilize CLDN6 proteins in the same cell, providing new insight into CLDN usage during HCV infection.Citation109 OCLN is also a key entry factor for HCV, as the expression of human OCLN and CD81 in mouse liver leads to viral permissivity in this originally non-susceptible animal model, while the silencing of this receptor perturbs HCV entry during a late post-binding step.Citation110,Citation111,Citation112 A recent study found that the overexpression of miR-122 can decrease HCV entry by downregulating OCLN.Citation113
EGFR and EphA2 are two well-understood RTKs and have recently been identified as host cofactors of HCV entry by a functional siRNA kinase screen.Citation63 These two kinases are highly expressed in human liver, and their specific inhibitors erlotinib (an EGFR inhibitor) and dasatinib (an EphA2 inhibitor), two clinically approved anticancer compounds, significantly impair viral entry in both polarized hepatoma cells and PHHs; erlotinib has also been effective in human-liver chimeric mice ().Citation63 RTKs modulate viral entry at post-binding steps by interfering with CD81–CLDN1 complex association and blocking cell-to-cell transmission, all of which makes these two RTKs promising targets for developing anti-HCV agents, especially for the prevention of graft reinfection in chronic HCV patients who must undergo liver transplantation.Citation114 Still, the specific efficacy and safety of using the currently licensed inhibitors of RTKs in HCV treatment requires further clinical evaluation.
Clinical observational data suggest that an iron metabolic disorder might occur in HCV-positive patients.Citation115,Citation116,Citation117 Transferrin receptor 1 (TfR1), an iron uptake receptor, is widely expressed in mammalian cells, including hepatocytes, and its trafficking protein (TTP) is involved in HCV entry. Both a specific anti-TfR1 mAb and a TfR1 inhibitor, ferristatin, impede HCV infection without affecting viral RNA replication when treatment is applied before virus incubation ().Citation64 Kinetic experiments suggest that TfR1 facilitates HCV entry at a post-binding step, most likely after CD81.Citation64 Further studies are needed to identify the functional domain of TfR1 that is used during HCV entry, which will enable the development of specifically targeted antiviral agents. However, HCV cell-to-cell transmission is not completely reduced after treatment with either the anti-TfR1 antibody or the TfR1 inhibitor, indicating that a different potential mechanism might be involved in this process.
NPC1L1 is a 13 transmembrane cholesterol transport receptor that is highly expressed on the apical surfaces of human enterocytes and the canalicular membranes of human hepatocytes.Citation118,Citation119 The main function of it is to modulate cholesterol homeostasis in the body.Citation120 A correlation exists between the NPC1L1 level and HCV infection.Citation66 Specific antibodies, especially ones that target the NPC1L1 large extracellular loop 1 (LEL1), effectively eradicate HCV entry in a manner similar to that of CD81 antibodies, indicating the potential action mode of this receptor.Citation65 Ezetimibe is an FDA-approved NPC1L1 antagonist that is clinically used to treat hypercholesterolemia. The application of ezetimibe inhibits HCV entry and cell-to-cell transmission in vitro, while in vivo, this drug delays the establishment of genotype 1 HCV infections in severe combined immunodeficiency (SCID) mice with humanized hepatocytes ().Citation66 The therapeutic window in humans has not yet been determined.
Clathrin-mediated viral endocytosis and membrane fusion
After interacting with a series of receptors, HC virions are internalized into cells via a highly coordinated process, most likely through clathrin-mediated endocytosis together with some cell entry factors, such as CD81 and CLDN1.Citation70 The trafficking of the CD81–CLDN1 receptor complex promotes simultaneous virus internalization and fusion.Citation60,Citation104,Citation106 During this process, the internalized vesicles form early and late endosomes, which are then prepared for the subsequent virion-cell fusion process.Citation69 The use of either siRNA molecules targeting the clathrin heavy chain or the specific inhibitor chlorpromazine, which effectively interferes with clathrin-coated pit formation, impairs HCVpp entry and HCVcc infection in host cells ().Citation67 Arbidol is an indole-derivative molecule that is licensed as an anti-influenza drug in China and Russia.Citation121,Citation122 This broad-spectrum antiviral uses several approaches to inhibit HCV cell entry. One of its antiviral mechanisms involves affecting clathrin-mediated endocytosis by impairing clathrin-coated pit release and dynamin-2-induced membrane scission ().Citation68
Membrane fusion between the virus and host cell is the final step before the viral genome is released into the cytosol to start translation and replication.Citation123 The envelope proteins are primed by virus–receptor interactions (most likely CD81) to become sensitive to low pH, and the fusogenic conformational changes of the related peptides are activated by the acidic environment and proper temperature of the endosomal lumen.Citation71,Citation90,Citation124 Lipids, including cholesterol and sphingomyelin (SM), are essential components that facilitate HCV fusion.Citation125,Citation126 Studies have shown that the addition of cholesterol enhances the fusion efficiency of the HCV particle. Additionally, among virions of different densities, the lowest-density particle, HCVcc, exhibits the highest specific fusogenicity.Citation127
HCV fusion inhibitors are basically characterized into the following three groups. The first group targets the acidification-triggering mechanism of virion–cell membrane fusion. Currently available acidification inhibitors include concanamycin A and bafilomycin A, two potent inhibitors of vacuolar ATPases ().Citation69 Chloroquine and ammonium chloride are also considered to disturb endosome acidification and to suppress the occurrence of membrane fusion in a dose-dependent manner ().Citation70,Citation71 The second group focuses on the lipid compositions of both virus and host cells, which are indispensable throughout the process of virus fusion. In addition to its inhibitory effect on viral endocytosis, the indole-derivative arbidol also inhibits HCV membrane fusion, most likely via a dual-binding mode that involves aromatic residues in viral glycoproteins and phospholipids at the membrane–water interface, thus impeding the required conformational changes of fusion peptides during virus fusion ().Citation72 Phenothiazines are small-molecule compounds containing nitrogen and sulfur tricyclic structures. Recently, three phenothiazine compounds have been identified as potent HCV entry inhibitors (); they suppress virion–cell membrane fusion by incorporating into target membranes and increase the fluidity of cholesterol-rich membranes, destabilizing the pre-fusion state of the virus.Citation73 Rigid amphipathic fusion inhibitors (RAFIs) are a class of synthetic rigid amphiphiles that are similar to phospholipids. RAFIs can interact with envelope lipids and increase the activation barrier of viral proteins, leading to the blockage of increased negative curvature during the initial viral fusion stage ().Citation74 AUY11 is a representative arabino-based RAFI.Citation75 LJ001 is a small-molecule compound that specifically intercalates into viral membranes to inactivate the virion from a pre-fusion state, thereby blocking virus fusion ().Citation76 Silymarin is a compound mixture of several flavonolignans and flavonoid taxifolins; it inhibits HCV infection both in vitro and in vivo in a similar pattern to that of arbidol ().Citation77,Citation78 The third group of fusion inhibitors includes several compounds with unclear mechanisms, including ferroquine, a chloroquine analog, and phosphorothioate oligonucleotides (PS-ONs), which act as amphipathic DNA polymers.Citation79,Citation80 PS-ONs inhibit HCV infection both in vitro and in vivo, possibly at the fusion step ().
Some natural, plant-derived compounds, such as the flavonoid ladanein, the terpenoids saikosaponin, oleanolic acid, tannic acid or gallic acid, or the small molecule SKI-1/SIP inhibitor PF-429242, have antiviral activities against HCV during an early stage of viral infection ().Citation81,Citation82,Citation83,Citation84,Citation85,Citation86 However, their exact antiviral mechanisms and potential applications in clinical practice require further investigation and evaluation.
Moreover, several FDA-approved drugs that have already been qualified for clinical application have shown antiviral activities against HCV infection. Chlorcyclizine HCl (CCZ) is an over-the-counter anti-histamine drug for allergy symptoms. CCZ blocks late-stage of HCV entry before viral replication and is now undergoing a phase Ib clinical trial.Citation87 Sorafenib is a multi-kinase inhibitor that has been approved for the treatment of hepatocellular carcinoma.Citation128 A recent study shows that sorafenib inhibits both HCV entry and production and affects CLDN1 expression and localization.Citation88 Aspirin, a commonly used analgesic and anti-platelet drug, blocks HCV entry by downregulating CLDN1 ().Citation89 These drugs are relatively accessible and affordable agents with an established clinical safety profile, thereby becoming promising candidates for drug repurposing for the treatment HCV infection.
Conclusions and perspectives
The unveiling of the molecular mechanisms of HCV entry in recent years has largely promoted the development of entry inhibitors targeting different stages of the early viral life cycle. Because viral entry is essential for the initiation, spread, and maintenance of HCV infection, great potential and numerous prospects exist for entry inhibitors to be applied as members of cocktail therapy in future HCV treatments. Although newly identified entry inhibitors have emerged continuously in recent years, most of them still remain in the in vitro stages of testing. Until now, only a few have entered clinical trials, including the most advanced entry inhibitor, ITX5061, which targets the host entry factor SRB1 to interfere with virus infection. This compound is currently undergoing phase II clinical trials and appears to be a promising option for future combination therapy.Citation9,Citation56,Citation63 Regardless of its outcome, ITX5061 provides a good example for the study and development of entry inhibitors. Moreover, several compounds with favorable anti-HCV potencies have already been approved to treat other diseases in the clinic and provide a good method of screening for novel entry inhibitors of HCV. summarizes the targets and developmental stages of current antiviral agents during the HCV entry process.
Unlike the currently marketed anti-HCV DAAs, which target viral proteins of high variability, most entry inhibitors are HTAs with high genetic barriers, which are valuable features for avoiding viral escape, due to their conserved nature. Because all of the major HCV genotypes are believed to enter host cells via the same cellular pathways, the antiviral activities of most entry inhibitors tend to be genotype-independent.Citation129 Moreover, entry inhibitors are also bestowed with the unique advantage of preventing cell-to-cell transmission as long as their antiviral targets cover common factors in both cell-free and cell-to-cell viral spread.Citation130 Additionally, such drugs will help end-stage HCV patients resist graft reinfection after undergoing liver transplantation. A series of novel antiviral agents is currently being tested in in vitro assays or in vivo animal models. Some of these agents have entered the clinical stage for evaluation in patients. The satisfactory outcomes of this class of entry inhibitors should complement current treatment approaches and lead to more efficient, economical and better tolerated options for HCV patients, especially difficult-to-treat patients.
Hepatitis C: blocking viral entry
Developmental drugs that inhibit viral entry into host cells could enhance existing treatment for hepatitis C virus (HCV) infection. Following a review of potential treatments, Zhong-Tian Qi and co-workers at the Second Military Medical University in Shanghai, China, recommend incorporating drugs known as entry inhibitors into HCV treatment regimens. A leading cause of liver disease, HCV is a highly complex virus with multiple strains in circulation. The HCV inhibitors currently undergoing trials target earlier stages of the viral life cycle than current antivirals. Existing treatments target viral proteins and are effective in most cases, but they are expensive and can have side effects. There are also issues with drug-resistant HCV strains and reinfection following liver transplants. The new inhibitors work either by impairing viral binding to host cells or making genetic changes to prevent cell-to-cell transmission.
Acknowledgments
This work was supported by the National S&T Major Project for Infectious Diseases Control (2012ZX10002003-004-010), Natural Science Foundation of China (81171564, 81273557, 81302812 and 81221061), Medical Youth Science Program (13QNP100), and Shanghai Municipal Natural Science Foundation (13ZR1449300).
- LavanchyD.The global burden of hepatitis C.Liver Int2009; 29:74–81.
- AlterMJ.Epidemiology of hepatitis C virus infection.World J Gastroenterol2007; 13:2436–2441.
- GhanyMG,StraderDB,ThomasDL,SeeffLB,American Association for the Study of LiverD.Diagnosis, management, and treatment of hepatitis C: an update.Hepatology2009; 49:1335–1374.
- LiangTJ,GhanyMG.Current and future therapies for hepatitis C virus infection.N Engl J Med2013; 368:1907–1917.
- MederackeI,Von HahnT.Survival of the fittest: selection of hepatitis C virus variants during liver graft reinfection.Hepatology2011; 53:705–708.
- BuhlerS,BartenschlagerR.New targets for antiviral therapy of chronic hepatitis C.Liver Int2012; 32:9–16.
- AghemoA,De FrancescoR.New horizons in hepatitis C antiviral therapy with direct-acting antivirals.Hepatology2013; 58:428–438.
- GaneEJ,StedmanCA,HylandRHet al.Nucleotide polymerase inhibitor sofosbuvir plus ribavirin for hepatitis C.N Engl J Med2013; 368:34–44.
- IssurM,GotteM.Resistance patterns associated with HCV NS5A inhibitors provide limited insight into drug binding.Viruses2014; 6:4227–4241.
- AlqahtaniSA,AfdhalN,ZeuzemSet al.Safety and tolerability of ledipasvir/sofosbuvir with and without ribavirin in patients with chronic hepatitis C virus genotype 1 infection: analysis of phase III ION trials.Hepatology2015; 62:25–30.
- KlibanovOM,GaleSE,SantevecchiB.Ombitasvir/paritaprevir/ritonavir and dasabuvir tablets for hepatitis C virus genotype 1 infection.Ann Pharmacother2015; 49:566–581.
- DeeksED.Ombitasvir/paritaprevir/ritonavir plus dasabuvir: a review in chronic HCV genotype 1 infection.Drugs2015; 75:1027–1038.
- A 4-drug combination (Viekira Pak) for hepatitis C.JAMA.2015; 313:1857–1858.
- Canadian Agency for Drugs and Technologies in Health.Holkira (ombitasvir/paritaprevir/ritonavir with dasabuvir) and harvoni (ledipasvir/sofosbuvir) for chronic hepatitis C: a review of the clinical evidence. Ottawa: CADTH, 2015.Available at https://www.cadth.ca/sites/default/files/pdf/htis/jan-2015/RC0632%20Holkira%20and%20Harvoni%20Final.pdf (accessed 16 Jan 2015).
- KeatingGM.Ledipasvir/sofosbuvir: a review of its use in chronic hepatitis C.Drugs2015; 75:675–685.
- TemesgenZ,RizzaSA.Daclatasvir for the treatment of chronic hepatitis C virus infection.Drugs Today (Barc)2015; 51:277–288.
- KowdleyKV,LawitzE,CrespoIet al.Sofosbuvir with pegylated interferon alfa-2a and ribavirin for treatment-naive patients with hepatitis C genotype-1 infection (ATOMIC): an open-label, randomised, multicentre phase 2 trial.Lancet2013; 381:2100–2107.
- LawitzE,LalezariJP,HassaneinTet al.Sofosbuvir in combination with peginterferon alfa-2a and ribavirin for non-cirrhotic, treatment-naive patients with genotypes 1, 2, and 3 hepatitis C infection: a randomised, double-blind, phase 2 trial.Lancet Infect Dis2013; 13:401–408.
- SarrazinC,HezodeC,ZeuzemS,PawlotskyJM.Antiviral strategies in hepatitis C virus infection.J Hepatol2012; 56:S88–100.
- BarthH,SchaferC,AdahMIet al.Cellular binding of hepatitis C virus envelope glycoprotein E2 requires cell surface heparan sulfate.J Biol Chem2003; 278:41003–41012.
- BarthH,SchnoberEK,ZhangFet al.Viral and cellular determinants of the hepatitis C virus envelope-heparan sulfate interaction.J Virol2006; 80:10579–10590.
- JiangJ,CunW,WuX,ShiQ,TangH,LuoG.Hepatitis C virus attachment mediated by apolipoprotein E binding to cell surface heparan sulfate.J Virol2012; 86:7256–7267.
- MolinaS,CastetV,Fournier-WirthCet al.The low-density lipoprotein receptor plays a role in the infection of primary human hepatocytes by hepatitis C virus.J Hepatol2007; 46:411–419.
- AlbeckaA,BelouzardS,Op de BeeckAet al.Role of low-density lipoprotein receptor in the hepatitis C virus life cycle.Hepatology2012; 55:998–1007.
- SyedGH,TangH,KhanM,HassaneinT,LiuJ,SiddiquiA.Hepatitis C virus stimulates low-density lipoprotein receptor expression to facilitate viral propagation.J Virol2014; 88:2519–2529.
- HelleF,WychowskiC,Vu-DacN,GustafsonKR,VoissetC,DubuissonJ.Cyanovirin-N inhibits hepatitis C virus entry by binding to envelope protein glycans.J Biol Chem2006; 281:25177–25183.
- KhanalM,BarrasA,VausselinTet al.Boronic acid-modified lipid nanocapsules: a novel platform for the highly efficient inhibition of hepatitis C viral entry.Nanoscale2015; 7:1392–1402.
- HolmskovU,ThielS,JenseniusJC.Collections and ficolins: humoral lectins of the innate immune defense.Annu Rev Immunol2003; 21:547–578.
- HamedMR,BrownRJ,ZothnerCet al.Recombinant human L-ficolin directly neutralizes hepatitis C virus entry.J Innate Immun2014; 6:676–684.
- CiesekS,von HahnT,ColpittsCCet al.The green tea polyphenol, epigallocatechin-3-gallate, inhibits hepatitis C virus entry.Hepatology2011; 54:1947–1955.
- CallandN,AlbeckaA,BelouzardSet al.(-)-Epigallocatechin-3-gallate is a new inhibitor of hepatitis C virus entry.Hepatology2012; 55:720–729.
- CallandN,SahucME,BelouzardSet al.Polyphenols inhibit hepatitis C virus entry by a new mechanism of action.J Virol2015; 89:10053–10063.
- RedwanEM,UverskyVN,El-FakharanyEM,Al-MehdarH.Potential lactoferrin activity against pathogenic viruses.C R Biol2014; 337:581–595.
- El-FakharanyEM,SanchezL,Al-MehdarHA,RedwanEM.Effectiveness of human, camel, bovine and sheep lactoferrin on the hepatitis C virus cellular infectivity: comparison study.Virol J2013; 10:199.
- RedwanEM,El-FakharanyEM,UverskyVN,LinjawiMH.Screening the anti infectivity potentials of native N- and C-lobes derived from the camel lactoferrin against hepatitis C virus.BMC Complement Altern Med2014; 14:219.
- HongW,LangY,LiTet al.A p7 ion channel-derived peptide inhibits hepatitis C virus infection in vitro.J Biol Chem2015; 290:23254–23263.
- VanCompernolleSE,WiznyciaAV,RushJR,DhanasekaranM,BauresPW,ToddSC.Small molecule inhibition of hepatitis C virus E2 binding to CD81.Virology2003; 314:371–380.
- BrimacombeCL,GroveJ,MeredithLWet al.Neutralizing antibody-resistant hepatitis C virus cell-to-cell transmission.J Virol2011; 85:596–605.
- LavilletteD,TarrAW,VoissetCet al.Characterization of host-range and cell entry properties of the major genotypes and subtypes of hepatitis C virus.Hepatology2005; 41:265–274.
- GottweinJM,ScheelTK,JensenTBet al.Development and characterization of hepatitis C virus genotype 1–7 cell culture systems: role of CD81 and scavenger receptor class B type I and effect of antiviral drugs.Hepatology2009; 49:364–377.
- VanwolleghemT,BukhJ,MeulemanPet al.Polyclonal immunoglobulins from a chronic hepatitis C virus patient protect human liver-chimeric mice from infection with a homologous hepatitis C virus strain.Hepatology2008; 47:1846–1855.
- MeulemanP,HesselgesserJ,PaulsonMet al.Anti-CD81 antibodies can prevent a hepatitis C virus infection in vivo.Hepatology2008; 48:1761–1768.
- JiC,LiuY,PamulapatiCet al.Prevention of hepatitis C virus infection and spread in human liver chimeric mice by an anti-CD81 monoclonal antibody.Hepatology2015; 61:1136–1144.
- MolinaS,CastetV,Pichard-GarciaLet al.Serum-derived hepatitis C virus infection of primary human hepatocytes is tetraspanin CD81 dependent.J Virol2008; 82:569–574.
- HsuM,ZhangJ,FlintMet al.Hepatitis C virus glycoproteins mediate pH-dependent cell entry of pseudotyped retroviral particles.Proc Natl Acad Sci USA2003; 100:7271–7276.
- WakitaT,PietschmannT,KatoTet al.Production of infectious hepatitis C virus in tissue culture from a cloned viral genome.Nat Med2005; 11:791–796.
- ZhongJ,GastaminzaP,ChengGet al.Robust hepatitis C virus infection in vitro.Proc Natl Acad Sci USA2005; 102:9294–9299.
- KapadiaSB,BarthH,BaumertT,McKeatingJA,ChisariFV.Initiation of hepatitis C virus infection is dependent on cholesterol and cooperativity between CD81 and scavenger receptor B type I.J Virol2007; 81:374–383.
- CaiZ,CaiL,JiangJ,ChangKS,van der WesthuyzenDR,LuoG.Human serum amyloid A protein inhibits hepatitis C virus entry into cells.J Virol2007; 81:6128–6133.
- LavieM,VoissetC,Vu-DacNet al.Serum amyloid A has antiviral activity against hepatitis C virus by inhibiting virus entry in a cell culture system.Hepatology2006; 44:1626–1634.
- ZeiselMB,KoutsoudakisG,SchnoberEKet al.Scavenger receptor class B type I is a key host factor for hepatitis C virus infection required for an entry step closely linked to CD81.Hepatology2007; 46:1722–1731.
- BartoschB,VerneyG,DreuxMet al.An interplay between hypervariable region 1 of the hepatitis C virus E2 glycoprotein, the scavenger receptor BI, and high-density lipoprotein promotes both enhancement of infection and protection against neutralizing antibodies.J Virol2005; 79:8217–8229.
- CataneseMT,GrazianiR,von HahnTet al.High-avidity monoclonal antibodies against the human scavenger class B type I receptor efficiently block hepatitis C virus infection in the presence of high-density lipoprotein.J Virol2007; 81:8063–8071.
- LacekK,VercauterenK,GrzybKet al.Novel human SR-BI antibodies prevent infection and dissemination of HCV in vitro and in humanized mice.J Hepatol2012; 57:17–23.
- MassonD,KosekiM,IshibashiMet al.Increased HDL cholesterol and apoA-I in humans and mice treated with a novel SR-BI inhibitor.Arterioscler Thromb Vasc Biol2009; 29:2054–2060.
- SyderAJ,LeeH,ZeiselMBet al.Small molecule scavenger receptor BI antagonists are potent HCV entry inhibitors.J Hepatol2011; 54:48–55.
- ZhuH,Wong-StaalF,LeeHet al.Evaluation of ITX 5061, a scavenger receptor B1 antagonist: resistance selection and activity in combination with other hepatitis C virus antivirals.J Infect Dis2012; 205:656–662.
- SulkowskiMS,KangM,MatiningRet al.Safety and antiviral activity of the HCV entry inhibitor ITX5061 in treatment-naive HCV-infected adults: a randomized, double-blind, phase 1b study.J Infect Dis2014; 209:658–667.
- SiY,LiuS,LiuXet al.A human claudin-1-derived peptide inhibits hepatitis C virus entry.Hepatology2012; 56:507–515.
- KriegerSE,ZeiselMB,DavisCet al.Inhibition of hepatitis C virus infection by anti-claudin-1 antibodies is mediated by neutralization of E2-CD81-claudin-1 associations.Hepatology2010; 51:1144–1157.
- FofanaI,KriegerSE,GrunertFet al.Monoclonal anti-claudin 1 antibodies prevent hepatitis C virus infection of primary human hepatocytes.Gastroenterology2010; 139:953–964 ,964 e951–954.
- MaillyL,XiaoF,LupbergerJet al.Clearance of persistent hepatitis C virus infection in humanized mice using a claudin-1-targeting monoclonal antibody.Nat Biotechnol2015; 5:549–54.
- LupbergerJ,ZeiselMB,XiaoFet al.EGFR and EphA2 are host factors for hepatitis C virus entry and possible targets for antiviral therapy.Nat Med2011; 17:589–595.
- MartinDN,UprichardSL.Identification of transferrin receptor 1 as a hepatitis C virus entry factor.Proc Natl Acad Sci USA2013; 110:10777–10782.
- Del CampoJA,RojasA,Romero-GomezM.Entry of hepatitis C virus into the cell: a therapeutic target.World J Gastroenterol2012; 18:4481–4485.
- SainzBJr,BarrettoN,MartinDNet al.Identification of the Niemann-Pick C1-like 1 cholesterol absorption receptor as a new hepatitis C virus entry factor.Nat Med2012; 18:281–285.
- BlanchardAA,WatsonPH,ShiuRPet al.Differential expression of claudin 1, 3, and 4 during normal mammary gland development in the mouse.DNA Cell Biol2006; 25:79–86.
- BlaisingJ,LevyPL,PolyakSJ,StaniferM,BoulantS,PecheurEI.Arbidol inhibits viral entry by interfering with clathrin-dependent trafficking.Antiviral Res2013; 100:215–219.
- MeertensL,BertauxC,DragicT.Hepatitis C virus entry requires a critical postinternalization step and delivery to early endosomes via clathrin-coated vesicles.J Virol2006; 80:11571–11578.
- BlanchardE,BelouzardS,GoueslainLet al.Hepatitis C virus entry depends on clathrin-mediated endocytosis.J Virol2006; 80:6964–6972.
- TscherneDM,JonesCT,EvansMJ,LindenbachBD,McKeatingJA,RiceCM.Time- and temperature-dependent activation of hepatitis C virus for low-pH-triggered entry.J Virol2006; 80:1734–1741.
- BoriskinYS,LenevaIA,PecheurEI,PolyakSJ.Arbidol: a broad-spectrum antiviral compound that blocks viral fusion.Curr Med Chem2008; 15:997–1005.
- Chamoun-EmanuelliAM,PecheurEI,SimeonRL,HuangD,CremerPS,ChenZ.Phenothiazines inhibit hepatitis C virus entry, likely by increasing the fluidity of cholesterol-rich membranes.Antimicrob Agents Chemother2013; 57:2571–2581.
- St VincentMR,ColpittsCC,UstinovAVet al.Rigid amphipathic fusion inhibitors, small molecule antiviral compounds against enveloped viruses.Proc Natl Acad Sci USA2010; 107:17339–17344.
- ColpittsCC,UstinovAV,EpandRF,EpandRM,KorshunVA,SchangLM.5-(Perylen-3-yl)ethynyl-arabino-uridine (aUY11), an arabino-based rigid amphipathic fusion inhibitor, targets virion envelope lipids to inhibit fusion of influenza virus, hepatitis C virus, and other enveloped viruses.J Virol2013; 87:3640–3654.
- WolfMC,FreibergAN,ZhangTet al.A broad-spectrum antiviral targeting entry of enveloped viruses.Proc Natl Acad Sci USA2010; 107:3157–3162.
- PolyakSJ,MorishimaC,ShuhartMC,WangCC,LiuY,LeeDY.Inhibition of T-cell inflammatory cytokines, hepatocyte NF-κB signaling, and HCV infection by standardized Silymarin.Gastroenterology2007; 132:1925–1936.
- WagonerJ,NegashA,KaneOJet al.Multiple effects of silymarin on the hepatitis C virus lifecycle.Hepatology2010; 51:1912–1921.
- VausselinT,CallandN,BelouzardSet al.The antimalarial ferroquine is an inhibitor of hepatitis C virus.Hepatology2013; 58:86–97.
- MatsumuraT,HuZ,KatoTet al.Amphipathic DNA polymers inhibit hepatitis C virus infection by blocking viral entry.Gastroenterology2009; 137:673–681.
- HaidS,NovodomskaA,GentzschJet al.A plant-derived flavonoid inhibits entry of all HCV genotypes into human hepatocytes.Gastroenterology2012; 143:213–222 e215.
- LinLT,ChungCY,HsuWCet al.Saikosaponin b2 is a naturally occurring terpenoid that efficiently inhibits hepatitis C virus entry.J Hepatol2015; 62:541–548.
- YuF,WangQ,ZhangZet al.Development of oleanane-type triterpenes as a new class of HCV entry inhibitors.J Med Chem2013; 56:4300–4319.
- BlanchetM,SureauC,GuevinC,SeidahNG,LabonteP.SKI-1/S1P inhibitor PF-429242 impairs the onset of HCV infection.Antiviral Res2015; 115:94–104.
- LiuS,ChenR,HagedornCH.Tannic acid inhibits hepatitis C virus entry into Huh7.5 Cells.PloS One2015; 10:e0131358.
- HsuWC,ChangSP,LinLCet al.Limonium sinense and gallic acid suppress hepatitis C virus infection by blocking early viral entry.Antiviral Res2015; 118:139–147.
- HeS,LinB,ChuVet al.Repurposing of the antihistamine chlorcyclizine and related compounds for treatment of hepatitis C virus infection.Sci Transl Med2015; 7:282–249.
- DescampsV,HelleF,LouandreCet al.The kinase-inhibitor sorafenib inhibits multiple steps of the hepatitis C virus infectious cycle in vitro.Antiviral Res2015; 118:93–102.
- YinP,ZhangL.Aspirin inhibits hepatitis C virus entry by downregulating claudin-1.J Viral Hepat2015Aug 20. doi: 10.1111/jvh.12446.
- KoutsoudakisG,KaulA,SteinmannEet al.Characterization of the early steps of hepatitis C virus infection by using luciferase reporter viruses.J Virol2006; 80:5308–5320.
- KhanN,MukhtarH.Tea polyphenols for health promotion.Life Sci2007; 81:519–533.
- Halegoua-De MarzioD,KraftWK,DaskalakisC,YingX,HawkeRL,NavarroVJ.Limited sampling estimates of epigallocatechin gallate exposures in cirrhotic and noncirrhotic patients with hepatitis C after single oral doses of green tea extract.Clin Ther2012; 34:2279–2285 e2271.
- Garcia-MontoyaIA,CendonTS,Arevalo-GallegosS,Rascon-CruzQ.Lactoferrin a multiple bioactive protein: an overview.Biochim Biophys Acta2012; 1820:226–236.
- GentzschJ,BrohmC,SteinmannEet al.Hepatitis C virus p7 is critical for capsid assembly and envelopment.PLoS Pathog2013; 9:e1003355.
- SteinmannE,PietschmannT.Hepatitis C virus p7-a viroporin crucial for virus assembly and an emerging target for antiviral therapy.Viruses2010; 2:2078–2095.
- OrenR,TakahashiS,DossC,LevyR,LevyS.TAPA-1, the target of an antiproliferative antibody, defines a new family of transmembrane proteins.Mol Cell Biol1990; 10:4007–4015.
- LevyS,ShohamT.The tetraspanin web modulates immune-signalling complexes.Nat Rev Immunol2005; 5:136–148.
- BoucheixC,RubinsteinE.Tetraspanins.Cell Mol Life Sci2001; 58:1189–1205.
- BaughJM,Garcia-RiveraJA,GallayPA.Host-targeting agents in the treatment of hepatitis C: a beginning and an end?Antiviral Res2013; 100:555–561.
- PerraultM,PecheurEI.The hepatitis C virus and its hepatic environment: a toxic but finely tuned partnership.Biochem J2009; 423:303–314.
- CataneseMT,AnsuiniH,GrazianiRet al.Role of scavenger receptor class B type I in hepatitis C virus entry: kinetics and molecular determinants.J Virol2010; 84:34–43.
- BaranovaIN,VishnyakovaTG,BocharovAVet al.Serum amyloid A binding to CLA-1 (CD36 and LIMPII analogous-1) mediates serum amyloid A protein-induced activation of ERK1/2 and p38 mitogen-activated protein kinases.J Biol Chem2005; 280:8031–8040.
- CaiL,de BeerMC,de BeerFC,van der WesthuyzenDR.Serum amyloid A is a ligand for scavenger receptor class B type I and inhibits high density lipoprotein binding and selective lipid uptake.J Biol Chem2005; 280:2954–2961.
- FarquharMJ,HuK,HarrisHJet al.Hepatitis C virus induces CD81 and claudin-1 endocytosis.J Virol2012; 86:4305–4316.
- HarrisHJ,FarquharMJ,MeeCJet al.CD81 and claudin 1 coreceptor association: role in hepatitis C virus entry.J Virol2008; 82:5007–5020.
- HarrisHJ,DavisC,MullinsJGet al.Claudin association with CD81 defines hepatitis C virus entry.J Biol Chem2010; 285:21092–21102.
- EvansMJ,von HahnT,TscherneDMet al.Claudin-1 is a hepatitis C virus co-receptor required for a late step in entry.Nature2007; 446:801–805.
- FuruseM,FujitaK,HiiragiT,FujimotoK,TsukitaS.Claudin-1 and -2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin.J Cell Biol1998; 141:1539–1550.
- HaidS,GretheC,DillMT,HeimM,KaderaliL,PietschmannT.Isolate-dependent use of claudins for cell entry by hepatitis C virus.Hepatology2014; 59:24–34.
- PlossA,EvansMJ,GaysinskayaVAet al.Human occludin is a hepatitis C virus entry factor required for infection of mouse cells.Nature2009; 457:882–886.
- DornerM,HorwitzJA,RobbinsJBet al.A genetically humanized mouse model for hepatitis C virus infection.Nature2011; 474:208–211.
- DornerM,HorwitzJA,DonovanBMet al.Completion of the entire hepatitis C virus life cycle in genetically humanized mice.Nature2013; 501:237–241.
- SendiH,Mehrab-MohseniM,FoureauDMet al.MiR-122 decreases HCV entry into hepatocytes through binding to the 3’ UTR of OCLN mRNA.Liver Int2015; 35:1315–1323.
- DiaoJ,PantuaH,NguHet al.Hepatitis C virus induces epidermal growth factor receptor activation via CD81 binding for viral internalization and entry.J Virol2012; 86:10935–10949.
- FujitaN,HoriikeS,SugimotoRet al.Hepatic oxidative DNA damage correlates with iron overload in chronic hepatitis C patients.Free Radic Biol Med2007; 42:353–362.
- FujitaN,SugimotoR,UrawaNet al.Hepatic iron accumulation is associated with disease progression and resistance to interferon/ribavirin combination therapy in chronic hepatitis C.J Gastroenterol Hepatol2007; 22:1886–1893.
- KaitoM.Molecular mechanism of iron metabolism and overload in chronic hepatitis C.J Gastroenterol2007; 42:96–99.
- YuL.The structure and function of Niemann-Pick C1-like 1 protein.Curr Opin Lipidol2008; 19:263–269.
- AltmannSW,DavisHRJr,ZhuLJet al.Niemann-Pick C1 like 1 protein is critical for intestinal cholesterol absorption.Science2004; 303:1201–1204.
- JiaL,BettersJL,YuL.Niemann-pick C1-like 1 (NPC1L1) protein in intestinal and hepatic cholesterol transport.Annu Rev Physiol2011; 73:239–259.
- BrooksMJ,BurtsevaEI,ElleryPJet al.Antiviral activity of arbidol, a broad-spectrum drug for use against respiratory viruses, varies according to test conditions.J Med Virol2012; 84:170–181.
- DeloguI,PastorinoB,BarontiC,NougairedeA,BonnetE,de LamballerieX.In vitro antiviral activity of arbidol against Chikungunya virus and characteristics of a selected resistant mutant.Antiviral Res2011; 90:99–107.
- HarrisonSC.Viral membrane fusion.Nat Struct Mol Biol2008; 15:690–698.
- SharmaNR,MateuG,DreuxM,GrakouiA,CossetFL,MelikyanGB.Hepatitis C virus is primed by CD81 protein for low pH-dependent fusion.J Biol Chem2011; 286:30361–30376.
- PecheurEI,DiazO,MolleJet al.Morphological characterization and fusion properties of triglyceride-rich lipoproteins obtained from cells transduced with hepatitis C virus glycoproteins.J Biol Chem2010; 285:25802–25811.
- HaidS,PietschmannT,PecheurEI.Low pH-dependent hepatitis C virus membrane fusion depends on E2 integrity, target lipid composition, and density of virus particles.J Biol Chem2009; 284:17657–17667.
- BlaisingJ,PecheurEI.Lipids: a key for hepatitis C virus entry and a potential target for antiviral strategies.Biochimie2013; 95:96–102.
- WilhelmS,CarterC,LynchMet al.Discovery and development of sorafenib: a multikinase inhibitor for treating cancer.Nat Rev Drug Discov2006; 5:835–844.
- FofanaI,JilgN,ChungRT,BaumertTF.Entry inhibitors and future treatment of hepatitis C.Antiviral Res2014; 104:136–142.
- ZeiselMB,FofanaI,Fafi-KremerS,BaumertTF.Hepatitis C virus entry into hepatocytes: molecular mechanisms and targets for antiviral therapies. J Hepatol 2011; 54:566–576.
