Abstract
Subtype H6 influenza A viruses (IAVs) are commonly detected in wild birds and domestic poultry and can infect humans. In 2010, a H6N6 virus emerged in southern China, and since then, it has caused sporadic infections among swine. We show that this virus binds to α2,6-linked and α2,3-linked sialic acids. Mutations at residues 222 (alanine to valine) and 228 (glycine to serine) of the virus hemagglutinin (HA) affected its receptor-binding properties. Experiments showed that the virus has limited transmissibility between ferrets through direct contact or through inhalation of infectious aerosolized droplets. The internal genes of the influenza A(H1N1)pdm09 virus, which is prevalent in swine worldwide, increases the replication efficiency of H6N6 IAV in the lower respiratory tract of ferrets but not its transmissibility between ferrets. These findings suggest H6N6 swine IAV (SIV) currently poses a moderate risk to public health, but its evolution and spread should be closely monitored.
Emerging Microbes & Infections (2017) 6, e17; doi:10.1038/emi.2017.3; published online 12 April 2017
Introduction
Influenza A virus (IAV) is an enveloped, segmented, single- and negative-stranded RNA virus belonging to the family Orthomyxoviridae. Migratory waterfowl are the natural reservoirs for IAVs, but these viruses also infect humans, domestic poultry, wild birds, pigs, dogs, cats, horses, mink and marine mammals, including seals and whalesCitation1 Human IAVs bind preferentially to N-acetylneuraminic acid-α2, 6-linked galactose (Neu5Acα2,6-Gal) receptors, whereas avian influenza viruses (AIVs) prefer N-acetylneuraminic acid-α2,3-linked galactose (Neu5Acα2,3-Gal) receptors.Citation2, Citation3, Citation4 Swine are considered a ‘mixing vessel’ of IAVs because they have both Neu5Acα2, 3-Gal and Neu5Acα2,6-Gal receptors throughout their respiratory tract. With these receptors, swine facilitate the generation of novel influenza reassortants and enable avian-like IAVs to obtain the ability to bind to human receptors,Citation5 as has been hypothesized to have occurred during the genesis of viruses that caused the 1957 H2N2 and 1968 H3N2 influenza pandemics.Citation6
H6-subtype IAVs have been detected in various migratory waterfowl and domestic poultry in Eurasia and North America.Citation7 Most H6 viruses introduced from waterfowl into domestic poultry have gained only limited spread. However, during 2000–2005, subtype H6N2 IAVs caused illness outbreaks among domestic poultry in CA, USA.Citation8, Citation9, Citation10 In addition, H6 IAVs have been shown to replicate well in mice without pre-adaptation, indicating that these viruses could cause cross-species infection in mammals.Citation11, Citation12 Laboratory experiments showed that humans can be infected with H6 IAVs through experimental inoculation.Citation13 Furthermore, findings from serologic surveillance suggested that veterinarians exposed to H6 IAV-infected domestic birds can become infected with the virus,Citation14 and in 2013, an avian-origin H6N1 IAV was reported to cause human infection, but there has been no evidence of subsequent human-to-human transmission.Citation15
Since 2002, H6 IAVs have been one of the predominant IAV subtypes circulating in live bird markets in southern China,Citation16, Citation17, Citation18 and some of these H6 viruses recognized human receptors.Citation19 In 2010, after an avian-origin H6N6 swine influenza A virus (SIV) was isolated from sick pigs in southern China, it was found that the virus had been transmitted to and was circulating among the swine population; seroprevalence rates ranged from 1.8% to 3.4%.Citation20, Citation21 The hemagglutinin (HA) protein of the currently circulating H6N6 SIV has amino acids 222V and 228S, compared with amino acids 222A and 228G in its potential AIV precursors. In other IAV subtypes, HA amino acids 222V and 228S have been reported to affect virus replication in mammals.Citation7, Citation22
The virus that caused the 2009 H1N1 pandemic, influenza A(H1N1)pdm09, was a swine-origin IAV.Citation23 After its discovery in humans, this virus quickly moved to swine and other animal populations worldwide.Citation24, Citation25, Citation26, Citation27, Citation28 During the past few years in southern China, A(H1N1)pdm09 virus has become one of the predominant viruses among domestic swine.Citation29, Citation30, Citation31 Frequent reassortments between influenza A(H1N1)pdm09 and other endemic SIVs have been observed.Citation32, Citation33
The aims of our study were to understand the impacts of two acquired mutations in HA of H6N6 virus on its receptor-binding properties and to assess the transmission potential of the H6N6 virus. We also aimed to assess the potential risks posed by reassortants of H6N6 virus with A(H1N1)pdm09 virus because such reassortants would be expected to emerge if both viruses continue to circulate in swine.
MATERIALS AND METHODS
Virus and RNA extraction
In 2010, an avian-like H6N6 SIV, A/swine/Guangdong/k6/2010(H6N6), was isolated from swine in southern China.Citation20 To study the virus, we used an RNeasy Mini Kit (Qiagen, Germantown, MD, USA) to extract RNA from the isolate; extraction was performed in a Biosafety Level 3 (BSL-3) laboratory.
Cells
Madin-Darby canine kidney (MDCK) cells and human embryonic kidney 293 T cells (both from American Type Culture Collection, Manassas, VA, USA) were used for virus propagation; the cells were maintained in Dulbecco’s Modified Eagle Medium (DMEM; Gibco/BRL, Grand island, NY, USA). A549 cells (American Type Culture Collection) used in assays were maintained in Advanced DMEM/F-12 (Gibco/BRL). The medium for each of the three cell lines was supplemented with 10% fetal bovine serum (Atlanta Biologicals, Lawrenceville, GA, USA), penicillin–streptomycin, and amphotericin B (Gibco/BRL), and the cells were held at 37 °C in 5% CO2.
Molecular cloning, mutagenesis and reverse genetics
The full-length cDNA for eight genes of A/swine/Guangdong/k6/2010(H6N6) virus were amplified by using the SuperScript One-Step RT-PCR system (Invitrogen, Carlsbad, CA, USA) and then cloned into a pHW2000 vector.Citation34 The site-directed mutagenesis on residues 222 and 228 of HA was performed using a QuikChange II Site-Directed Mutagenesis Kit (Stratagene, La Jolla, CA, USA). Primers are available upon request.
Eleven recombinant viruses were generated (Table ) by reverse genetics as previously described.Citation35 The recombinant viruses were confirmed by Sanger sequencing at the Life Sciences Core Laboratories Center at Cornell University (Ithaca, NY, USA).
Table 1 Recombinant influenza A(H6N6) viruses generated in this study
Hemagglutination and hemagglutination inhibition assays
Hemagglutination and hemagglutination inhibition (HI) assays were carried out by using 0.5% turkey erythrocytes as previously described.Citation35
Glycans
The biotinlyated α2,3- and α2,6-sialic acid receptors (3′SLN and 6′SLN, respectively) were purchased from GlycoTech (Gaithersburg, MD, USA). N-linked glycans, Manα1,6-(Neu5Acα2,3-Galβ1,4-GlcNAcβ1,2-Manα1,3-)Manβ1,4-GlcNAcβ1,4-GlcNAc (N32) and Neu5Acα2,3-Galβ1,4-GlcNAcβ1, 2-Manα1,6-(Manα1,3-)Manβ1,4-GlcNAcβ1,4-GlcNAc (N52) were synthesized to represent as N-acetylneuraminic acid-α2,3-linked galactose (Neu5Acα2,3-Gal) and Manα1,6-(Neu5Acα2,6-Galβ1,4-GlcNAcβ1,2-Manα1,3-)Manβ1,4-GlcNAcβ1,4-GlcNAc (N33) and Neu5Acα2,6-Galβ1,4-GlcNAcβ1,2-Manα1,6-(Manα1,3-)Manβ1,4-GlcNAcβ1,4-GlcNAc (N53) were synthesized to represent as N-acetylneuraminic acid-α2,6-linked galactose (Neu5Acα-2,6-Gal). The N-linked glycans were first labeled by 2-amino-N-(2-amino-ethyl)-benzamide (AEAB) as described previouslyCitation36 and then biotinlyated by using EZ-Link NHS-LC-LC-Biotin (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s instruction. Glycan quantities were measured by high-performance liquid chromatography (Shimadzu, Columbia, MD, USA).
Virus glycan receptor-binding assay
The glycan stock solution (1 mg/mL) was prepared in 50% glycerol in 1 × phosphate-buffered saline (PBS) (v/v), according to the manufacturer’s instructions. The protein concentration in viruses was determined using a Pierce BCA Protein Assay Kit (Thermo Fisher Scientific) according to the manufacturer’s instructions. For the binding analysis, we further diluted the sialic acid receptors (N-linked glycans, 3′SLN and 6′SLN) and the viruses in a PBS solution (pH=7.4) containing 0.01% bovine serum albumin and 0.002% Tween-20 (1 × Kinetics Buffer 10 × ; FortéBIO Inc., Menlo Park, CA, USA) with 10 μM neuraminidase inhibitor (zanamivir hydrate; Moravek Inc., Brea, CA, USA) and 10 μM oseltamivir phosphate (American Radiolabeled Chemicals Inc., St Louis, MO, USA). The binding assay was performed by using a FortéBIO Octet K2 interferometer equipped with streptavidin biosensor tips (FortéBIO Inc.). In summary, the biotinylated receptors were first coated onto the biosensor tips for 300 s, after which the tips were dipped into a 1 mg/mL protein concentration of virus for 1200 s (association step) and then into the kinetics buffer with neuraminidase inhibitors for 1000 s (dissociation step). The entire measurement cycle was maintained at 30 °C with orbital shaking at 1000 × rpm.
Growth kinetics and plaque assays
To determine the growth kinetics, we inoculated the MDCK and A549 cells with rgH6N6-222V/228S, rgH6N6 × pdm09-222V/228S, rgH6N6-222A/228G, rgH6N6 × pdm09-222A/228G, rgH3N2 × pdm09 (Table ), wild-type A/California/04/2009(H1N1), or wild-type A/swine/Ohio/11SW226/2011(H3N2) at a multiplicity of infection of 0.001 (for MDCK cells) or 0.1 (for A549 cells). After the cells were incubated at 37 °C for 1 h, the inocula were removed. Cells were then washed twice with PBS and incubated for 96 h at 37 °C in 5% CO2 with 1.5 μg/mL Opti-MEM I (Life Technologies, Carlsbad, CA, USA) or Opti-MEM I containing TPCK-treated trypsin. At 12, 24, 48 and 72 h after inoculation, supernatant (200 μL) was collected from the cells and titrated, by 50% tissue culture infectious dose (TCID50), in MDCK cells.
Plaque assays were performed on MDCK cells in six-well tissue culture plates. Serial dilutions were prepared from the virus stock, and 800 μL of each dilution was incubated in MDCK cells at 37 °C with 5% CO2 for 1 h. The inocula were then aspirated, and the cells were overlaid with 2 mL of 1% agarose containing TPCK-treated trypsin (1.5 μg/mL). Cultures were incubated for 3 days at 37 °C and then fixed with methanol and stained with 1% crystal violet to reveal plaques.
Glycan microarray and data analyses
The viruses were purified using 25% sucrose as previously described.Citation37 The virus labeling, glycan microarray hybridization and data analyses were performed as previously described.Citation38
Animal experiments
To test the transmissibility of the two testing viruses (rgH6N6-222V/228S and rgH6N6 × pdm09-222V/228S), we designed six experiment groups for each virus: three aerosol transmission and three direct contact transmission groups. Two 4-month-old female ferrets (Triple F Farms) were included in each of the 12 groups: one as a virus-inoculated ferret to be inoculated intranasally with a testing virus (106 TCID50 viral load in a 1-mL volume) and the other as an exposure ferret to be exposed to the virus through indirect (that is, aerosol) or direct contact with the virus-inoculated ferret. Before the experiments were conducted, all 24 ferrets tested negative for antibodies to rgH6N6-222V/228S (wild-type-like), A/California/04/2009(H1N1), A/Perth/16/2009(H3N2), A/Victoria/361/2011(H3N2) and A/Minnesota/307875/2012(H3N2) influenza viruses. In the aerosol transmission groups, the virus-inoculated and exposure ferrets were housed in the same cage on different sides of a 1-cm-thick, double-layered, steel partition with 5-mm perforations (Allentown Inc., Allentown, NJ, USA). The airflow in the cage went from the exposure ferret to the virus-inoculated ferret. In the direct-contact transmission groups, the virus-inoculated and exposure ferrets were housed together in the same cage without a partition. In all cages, the exposure ferret was put into the cage 1 day after the virus-inoculated ferret was inoculated with a testing virus.
Nasal wash fluids were collected from virus-inoculated ferrets at one and two days post inoculation (DPI) and from exposure ferrets at 1 DPI; thereafter, nasal wash fluids were collected every other day until 10 DPI for both groups of ferrets. Body temperature and weight were measured before nasal wash fluids were collected.
Serum samples were collected from all ferrets at 14 DPI, immediately before they were killed. Virus titers in nasal wash fluids were determined by TCID50 in MDCK cells and confirmed by 50% egg infectious dose (EID50) in 9-day-old embryonated chicken eggs.
To test the replication efficiency of testing viruses in the ferret respiratory track, we killed two of the three virus-inoculated ferrets in each contact transmission group at 3 DPI. The turbinates, trachea, bronchi and lungs were collected, and virus titers were determined by TCID50 in MDCK cells.
Biosafety and animal handling
All laboratory and animal experiments were conducted under BSL-2 conditions, with investigators wearing appropriate protective equipment, and in compliance with protocols approved by the Institutional Animal Care and Use Committee (IACUC) of Mississippi State University.
Phylogenetic analyses
We conducted multiple sequence alignments by using the MUSCLE software package.Citation39 We used GARLI version 0.96Citation40 and maximum likelihood criteria to perform phylogenetic analyses, and we conducted bootstrap resampling analyses with 1000 runs by using PAUP* 4.0 BetaCitation41 with a neighbor-joining method as described elsewhere.Citation42
RESULTS
H6N6 SIV differs genetically from the H6N1 AIV that infected humans
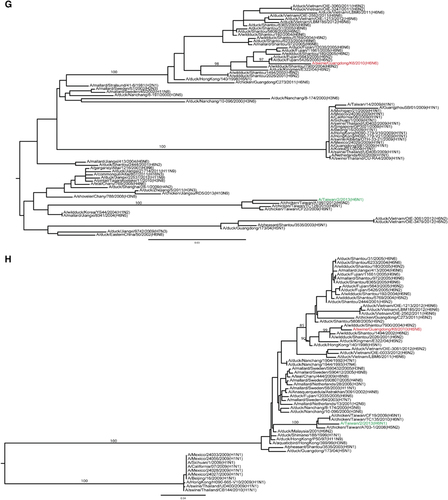
Phylogenetic analyses showed that the HA genes from H6N6 SIVs and H6 AIVs, including the strain that caused human infection, belong to different sublineages within a Eurasian lineage (Figure ). The PB2, NP and NS genes of H6N6 and H6N1 viruses belong to the same genetic lineages, but the PB1, PA and MP genes belong to different lineages (Figures ). None of these genes was genetically close to those of influenza A(H1N1)pdm09 virus or other circulating H3N2 and H1N1 SIVs in southern China (Figures ). The HA protein in the H6N6 SIV is 73.2% identical to that of the H6N1 AIV that was isolated from a human in southern China. The HA in the H6N6 SIV has amino acids 222V and 228S, whereas the HA in the human H6N1 AIV has amino acids 222A and 228S; the corresponding progenitors of these SIVs (that is, subtype H6 AIVs) have amino acids 222A and 228G.
Substitution G228S but not A222V increases binding affinity of H6N6 viruses to guinea pig and horse erythrocytes
To investigate whether substitutions A222V and G228S (avian to swine) affect virus-binding affinity to erythrocytes, we conducted hemagglutination assays with eight reassortant viruses generated by reverse genetics: rgH6N6 × PR8-222V/228S, rgH6N6 × PR8-222A/228S, rgH6N6 × PR8-222V/228G, rgH6N6 × PR8-222A/228G, rgH6N6 × pdm09-222V/228S, rgH6N6 × pdm09-222A/228S, rgH6N6 × pdm09-222V/228G, and rgH6N6 × pdm09-222A/228G (Table ). Erythrocytes from chicken, turkey, guinea pigs and horses were used. All eight viruses were normalized to a hemagglutination titer of 32 by using turkey erythrocytes (Table ). The rgH6N6 × PR8-222V/228S, rgH6N6 × pdm09-222V/228S, rgH6N6 × PR8-222A/228S and rgH6N6 × pdm09-222A/228S viruses had hemagglutination titers of 16 or 32 against chicken, guinea pig and horse erythrocytes. However, the rgH6N6 × PR8-222V/228G, rgH6N6 × pdm09-222V/228G, rgH6N6 × PR8-222A/228G and rgH6N6 × pdm09-222A/228G viruses had a hemagglutination titer of ≤2 to guinea pig and horse erythrocytes and a hemagglutination titer of ≥64 to chicken erythrocytes. Thus, substitution G228S increased the binding affinity of H6N6 virus to guinea pig and horse erythrocytes.
To further determine the effects of mutations A222V and G228S (avian to swine) on the receptor-binding properties of H6N6 virus, we also performed binding assays using a FortéBIO system with a set of Neu5Acα2,3-Gal and Neu5Acα2,6-Gal glycans: two glycan analogs (3′SLN and 6′SLN) and four synthetic N-linked glycans (N32, N33, N52 and N53). Results showed that the rgH6N6-222V/228S (wild-type-like) virus can bind to Neu5Acα2,3-Gal (3′SLN, N32 and N52) and Neu5Acα2,6-Gal (6′SLN, N33 and N53), but the affinities to Neu5Acα2,3-Gal (3′SLN, N32 and N52) were at least two-fold higher than those to Neu5Acα2,6-Gal (6′SLN, N33 and N53; Figure ). Binding affinities for the avian-like mutants rgH6N6-222V/228G and rgH6N6-222A/228G to the Neu5Acα2,6-Gal glycans (6′SLN, N33 and N53) were reduced by more than 80% compared with those for rgH6N6-222V/228S, but binding affinities to two synthetic Neu5Acα2,3-Gal glycans (3′SLN, N32 and N52) were not reduced to the same extent. The binding affinities of avian-like mutant rgH6N6-222A/228S to Neu5Acα2,3-Gal (3′SLN, N32 and N52) and Neu5Acα2,6-Gal (6′SLN, N33 and N53) were reduced by different extents (Figure ). In summary, mutation G228S (avian to swine) increased the binding affinity of H6N6 IAV to the testing Neu5Acα2,6-Gal glycans (6′SLN, N33 and N53) and Neu5Acα2,3-Gal (3′SLN) but not to Neu5Acα2,3-Gal (N32 and N52). Compared with mutation G228S, mutation A222V (avian to swine) had much less effect on binding affinities to Neu5Acα2,3-Gal and Neu5Acα2,6-Gal glycans.
H6N6 virus binds to α2,3- and α2,6-linked sialic acid receptors, and mutations A222V and G228S affect virus receptor affinity
The glycan array, which contained a total of 152 α2,3-linked and α2,6-linked glycans, was used to determine the receptor-binding profile of H6N6 SIV and the effect of mutations V222A and S228G on the glycan-binding profile of H6N6 IAV. Results showed that rgH6N6 × PR8-222V/228G and rgH6N6 × PR8-222A/228G had binding affinities below the detection threshold for the majority of the α2,3-linked and α2,6-linked glycans on the glycan array (Figure ). However, rgH6N6 × PR8-222V/228S showed high affinity for binding to 98 of the α2,3-linked and 54 of the α2,6-linked glycans; binding affinities were above the detection threshold of 2000 mean relative fluorescence units. Furthermore, the binding affinities to α2,3-linked and α2,6-linked glycans for rgH6N6 × PR8-222A/228S were weaker than those for rgH6N6 × PR8-222V/228S but higher than those for rgH6N6 × PR8-222V/228G and rgH6N6 × PR8-222A/228G. These results suggest double mutations A222V and G228S increased the binding affinities of H6N6 virus to the sialic acid glycans used in the glycan arrays.
Transmission of H6N6 wild-type SIV and rgH6N6 virus possible between ferrets
We used a ferret model to determine the transmissibility of rgH6N6-222V/228S virus by direct and indirect (aerosol) contact. In the direct-contact transmission experiment, the rgH6N6-222V/228S virus-inoculated ferrets did not show obvious clinical signs of illness. At 1 DPI, nasal wash fluids from these ferrets had virus titers ranging from 103.67 to 103.83 TCID50/mL, and at 2 DPI, titers peaked at 104.5 TCID50/mL; viral shedding continued until 5 DPI in these ferrets (Figure ). HI assay results showed that serum collected from these virus-inoculated ferrets at 14 DPI had virus titers ranging from 1:320 to 1:1280, indicating all ferrets seroconverted (Table ). Ferrets exposed to the virus-inoculated ferrets through direct contact had no detectable viral shedding when MDCK cells were used for detection; however, one of the three direct-contact ferrets showed seroconversion (Table ).
As in the direct-contact transmission experiment, rgH6N6-222V/228S virus-inoculated ferrets in the aerosol transmission study did not exhibit clinical signs of illness. At 1 DPI, nasal wash fluids from these virus-inoculated ferrets had median rgH6N6 virus titers ranging from 103.00 to 104.00 TCID50/mL, and virus titers peaked at 2 DPI at 104.50 TCID50/mL; these virus-inoculated ferrets continued to shed viruses until 6 DPI (Figure ). HI assay results showed that serum collected from these virus-inoculated ferrets at 14 DPI had virus titers ranging from 1:320 to 1:1280, indicating all ferrets had seroconverted (Table ). None of the three exposure ferrets in the aerosol transmission study had detectable viral shedding when MDCK cells were used as the detection method, and HI assay results showed that none of the ferrets had seroconverted before being killed at 14 DPE (Table ). However, when we used embryonated chicken eggs as the detection method, the nasal wash fluids collected from one of the three ferrets at 6 and 8 DPE had an EID50 of 10 (Table ).
Table 3 Hemagglutination inhibition titers in serum samples from ferrets inoculated with or exposed to animals inoculated with influenza A(H6N6) virus
To validate the aerosol transmissibility of this H6N6 virus, we repeated the experiment with the wild-type H6N6 isolate at an animal BSL-3 facility. The results from this independent experiment showed that the wild-type H6N6 isolate caused seroconversion in only one of the two direct-contact exposure ferrets and one of the two aerosol-exposure ferrets (data not shown), supporting that H6N6 virus has limited transmissibility between ferrets through direct contact or through inhalation of infectious aerosolized droplets.
Internal genes of influenza A(H1N1)pdm09 virus did not facilitate transmission of H6N6 virus among ferrets
To assess the risks posed by a potential reassortant rgH6N6 × pdm09 strain, which could result from co-circulating H6N6 SIV and influenza A(H1N1)pdm09 virus, we determined transmissibility of the reassortant virus by direct contact and aerosol contact in ferrets. The three rgH6N6 × pdm09-222S/228V virus-inoculated ferrets had weight loss and slightly elevated body temperatures at 3 DPI, but the ferrets showed clinical recovery from 4 DPI onward. Nasal wash fluids collected from the virus-inoculated ferrets at 1 DPI had virus titers of 104.67–106.00 TCID50/mL; at 2 DPI, virus titers peaked at 106.33 TCID50/mL and continued to shed until 6 DPI (Figure ). All three virus-inoculated ferrets seroconverted, with HI titers ranging from 1:640 to 1:1280, at 14 DPI (Table ).
In the direct-contact transmission experiment, two of the three exposure ferrets had no overt signs of illness. However, these ferrets had detectable virus loads (range, 104.00–104.50 TCID50/mL in nasal wash fluids at 4 DPE (Figure ), and viral shedding was sustained for at least 5 days. All three direct-contact exposure ferrets seroconverted, with HI titers of 1:640, at 14 DPE (Table ).
In the aerosol transmission experiment, the exposure ferrets had no detectable viral shedding when MDCK cells were used as the detection method. However, the nasal wash fluids collected from one of the three exposure ferrets at 2 DPE had a virus titer of 10 EID50/mL when embryonated chicken eggs were used for detection (Figure ; Table ), but none of these exposure ferrets seroconverted by 14 DPE (Table ).
Internal genes of influenza A(H1N1)pdm09 virus increased replication efficiency of H6N6 virus in ferret lower respiratory tract
To evaluate the effects of the internal genes on the pathogenesis of H6N6 virus, we inoculated ferrets with rgH6N6-222S/228G and rgH6N6 × pdm09-222S/228G viruses and compared the replication efficiencies of the viruses in ferret respiratory track tissues. Results showed rgH6N6-222S/228G replicated without prior adaptation in ferret nasal turbinate, trachea and lung but not in bronchi (Figure ). At 3 DPI, the turbinate, trachea and lung tissues of the ferrets inoculated with rgH6N6-222S/228G (wild-type) virus had virus titers of 105.53, 103.84 and 102.87 TCID50/g, respectively. In the ferrets inoculated with rgH6N6 × pdm09-222S/228G virus, the turbinate, trachea, bronchi and lung tissues at 3 DPI had virus titers of 106.00, 103.67, 104.26 and 103.59 TCID50/g, respectively. Thus, the virus titers in respiratory tract tissues, especially lower respiratory tract tissues, such as bronchi and lung, from the rgH6N6 × pdm09-222S/228G virus-inoculated ferrets was five-fold higher than those from rgH6N6-222S/228G virus-inoculated ferrets.
DISCUSSION
Together, the demonstrated promiscuous nature of H6 AIVs, their prevalence in southern China, and the case of H6N1 virus infection in a human in southern China Citation15 raise concerns that the H6N6 SIV emerging in that area’s swine population could become or contribute to an enzootic influenza strain causing human infections. Such a transition could occur through the commonplace evolutionary events in influenza viruses, such as acquisition of adapted mutations or entire gene segments from currently co-circulating SIVs. Phylogenetic analyses showed that the H6N6 virus is genetically different from the H6N1 virus that infected a human in Taiwan. These findings suggest that the H6 IAVs in southern China are genetically diverse,Citation16, Citation17, Citation18 and active evolutionary events that are still ongoing among the H6 AIVs have led to the emergence of an H6N1 virus in a humanCitation15, Citation43, Citation44 and to H6N6 viruses in swine.Citation20 Virus mutation and reassortment rates have been key measures in virologic risk assessments of influenza.Citation45 The presence of genetically diverse H6 IAVs and active evolutionary events increases the possibility for a virus of this subtype to develop pandemic potential and present a risk to public health.
Compared with its progenitor AIV, H6N6 SIV has mutations 222V and 228S in the 220-loop of its HA. Mutations in this 220-loop (for example, at residues 222, 225, 226 and 228) have been shown to affect receptor-binding specificity. For example, mutations Q226L and G228S enabled H3 viruses to bind sialic acid α2,3-Gal and sialic acid α2,6-Gal receptors.Citation46 The D222G substitution (corresponding to residue 225 in H3 viruses) enabled influenza A(H1N1)pdm09 viruses to acquire dual receptor specificity for complex α2,3-linked and α2,6-linked sialic acids; the substitution also increased the virulence of this virus.Citation47 We recently showed that the W222L substitution in HA could facilitate infection of H3N2 IAV in dogs, possibly by increasing the binding affinity of the virus to canine-specific receptors with Neu5Acα2,3-Galβ1,4-(Fucα-) or Neu5Acα2,3-Galβ1,3-(Fucα-)-like structures.Citation38 In addition, mutation W222R in HA can increase influenza virus infectivity in miceCitation48, Citation49 by introducing a hydrogen bond between the virus HA and the host glycan receptor.Citation50
Previous studies showed that turkey and chicken erythrocytes express α2,3- and α2,6-linked sialic acids that horse erythrocytes almost exclusively express α2,3-linked sialic acids,Citation51 and that guinea pig erythrocytes disproportionately express α2,3- and α2,6-linked sialic acids.Citation52 The receptor-binding profiles we obtained in this study with turkey, chicken, guinea pig, dog and horse erythrocytes suggest that mutations V222A and G228S, which are associated with a change in affinity from avian to swine, especially mutation G228S, changed receptor-binding specificity. This mutation G228S led to a minimum 16-fold increase of receptor-binding affinities for guinea pig and horse erythrocytes and a minimum two-fold decrease in binding affinities to chicken erythrocytes (Table ). Thus, mutation G228S could affect virus binding to both α2,3-linked and α2,6-linked sialic acids, but the effects on α2,6-linked sialic acids were significantly higher than that on α2,3-linked sialic acids. These results were confirmed by the virus glycan-binding assay results, which showed that wild-type H6N6 virus can bind to α2,3- and α2,6-linked sialic acids and that substitution G228S (avian to swine) significantly increased the binding affinities of H6N6 IAV to three α2,6-linked sialic acids that we tested (Figure ). In addition, glycan microarray data analysis confirmed that mutations A222V and G228S (avian to swine) can increase HA-binding affinities to glycans, including α2,6-linked sialic acid (Figure ). The results from virus glycan binding were consistent with those using recombinant HA proteins of H6N1 human influenza virus, which has amino acid 228S.Citation53 In summary, these findings indicated that mutations A222V and especially G228S, could facilitate the binding ability of H6N6 virus to α2,6-linked sialic acids. The conservation of S228 between the H6N1 human isolate and H6N6 SIV suggests that the G228S mutation likely facilitated transmission of H6 IAV to swine or humans.
Table 2 Effect of mutations A222V and G228S of influenza A(H6N6) virus hemagglutinin on binding affinity to erythrocytes from various animalsFootnotea
In H6N6 SIV-infected and rgH6N6 virus-infected ferrets, virus was shed at high titers until 6 DPI, similar to viral shedding by other H6 viruses.Citation11, Citation12 Previous studies in ferrets showed that H6N2 AIVs replication was more efficiently in lungs than nasal turbinates at 5 DPI.Citation11 However, our findings show that the minimum H6N6 SIV load in nasal turbinates was 10-fold higher than that in lungs at 3 DPI (Figure ). In our experiments, it is likely that H6N6 SIV replicated better in the upper than lower respiratory tract of ferrets; such a situation would lead to constant viral shedding and to transmissibility of this virus among ferrets. Additional experiments are needed to understand whether mutations A222V and G228S (avian to swine) alter viral tissue tropisms in ferrets.
A number of studies have shown that AIVs, such as highly pathogenic H5N1 and low pathogenicity H9N2 viruses, have increased aerosol transmissibility after acquiring the internal genes of influenza A(H1N1)pdm09 virus.Citation54, Citation55, Citation56 However, the results of our transmission studies showed that the internal genes of A(H1N1)pdm09 virus did not increase transmissibility of H6N6 SIVs through aerosol or direct contact. Nonetheless, it is possible that additional mutations acquired through adaptation or genetic reassortments between H6N6 SIV and influenza A(H1N1)pdm09 virus would increase the transmissibility of H6N6 SIV.
In the pathogenesis studies, virus replicated in lungs of rgH6N6 virus–infected ferrets, but the ferrets exhibited no obvious signs of illness. Virus titers in lungs of rgH6N6 × pdm09 virus-infected ferrets were ≥5-fold higher than those in rgH6N6 virus-infected ferrets. In addition, ferrets infected with rgH6N6 × pdm09 virus showed slightly elevated temperatures and weight loss at 1 and 2 DPI.
We showed that the method used for detecting virus loads in nasal wash fluids or tissues affects the accuracy of the results. For example, embryonated chicken eggs were more sensitive than MDCK cells in detecting H6N6 SIVs in the virus transmission group: viruses in aerosol-exposed ferrets were detectable in embryonated chicken eggs but not in MDCK cells (Table ).
Table 4 Virus titers in nasal wash fluids of ferrets in aerosol transmission experiment nasally inoculated with influenza A(H6N6) virus
In summary, our findings suggest that subtype H6N6 virus can bind to α2,6-linked sialic acids, indicating H6N6 virus as a virus with zoonotic potential. Although H6N6 SIV has limited transmissibility between ferrets and probably cannot yet be transmitted between ferrets through infectious aerosolized droplets, the virus could evolve into a more transmissible H6 virus through additional adaptation and reassortment. Thus, evolution of this H6N6 virus and other H6 AIVs should be closely monitored.
Supplementary Information
Download PDF (217.1 KB)Acknowledgments
We thank Dr Lucy Senter (College of Veterinary Medicine, Mississippi State University) and the Mississippi State University Office of Laboratory Animal Resources for assistance with the ferret experiments. We acknowledge the Consortium for Functional Glycomics, funded by the National Institute of General Medical Science (GM62116 and GM98791), for services provided by the Glycan Array Synthesis Core (The Scripps Research Institute, La Jolla, CA, USA), which produced the mammalian glycan microarray. We also acknowledge the Protein–Glycan Interaction Core (Emory University School of Medicine, Atlanta, GA, USA) for assistance with the analysis of samples on the array. This study was supported by the National Institutes of Health (grant P20GM103646).
Supplementary Information for this article can be found on the Emerging Microbes & Infections website (http://www.nature.com/emi)
References
- Webster RG, Bean WJ, Gorman OTet al.Evolution and ecology of influenza A viruses. Microbiol Rev 1992;56: 152–179.
- Connor RJ, Kawaoka Y, Webster RGet al.Receptor specificity in human, avian, and equine H2 and H3 influenza virus isolates. Virology 1994;205: 17–23.
- Rogers GN, Pritchett TJ, Lane JLet al.Differential sensitivity of human, avian, and equine influenza A viruses to a glycoprotein inhibitor of infection: selection of receptor specific variants. Virology 1983;131: 394–408.
- Rogers GN, D'Souza BL.Receptor binding properties of human and animal H1 influenza virus isolates. Virology 1989;173: 317–322.
- Ito T, Couceiro JN, Kelm Set al.Molecular basis for the generation in pigs of influenza A viruses with pandemic potential. J Virol 1998;72: 7367–7373.
- Cox NJ, Subbarao K.Global epidemiology of influenza: past and present. Annu Rev Med 2000;51: 407–421.
- Munster VJ, Baas C, Lexmond Pet al.Spatial, temporal, and species variation in prevalence of influenza A viruses in wild migratory birds. PLoS Pathog 2007;3: e61.
- Kinde H, Read DH, Daft BMet al.The occurrence of avian influenza A subtype H6N2 in commercial layer flocks in Southern California (2000-02): clinicopathologic findings. Avian Dis 2003;47: 1214–1218.
- Yee KS, Novick CA, Halvorson DAet al.Prevalence of low pathogenicity avian influenza virus during 2005 in two US live bird market systems. Avian Dis 2011;55: 236–242.
- Knobler SL, Mack A, Mahmoud Aet al. The Threat of Pandemic Influenza: Are We Ready? Workshop Summary.Washington (DC): National Academies Press, 2005.
- Gillim-Ross L, Santos C, Chen Zet al.Avian influenza h6 viruses productively infect and cause illness in mice and ferrets. J Virol 2008;82: 10854–10863.
- Lee MS, Chang PC, Shien JHet al.Genetic and pathogenic characterization of H6N1 avian influenza viruses isolated in Taiwan between 1972 and 2005. Avian Dis 2006;50: 561–571.
- Beare AS, Webster RG.Replication of avian influenza viruses in humans. Arch Virol 1991;119: 37–42.
- Myers KP, Setterquist SF, Capuano AWet al.Infection due to 3 avian influenza subtypes in United States veterinarians. Clin Infect Dis 2007;45: 4–9.
- Wei SH, Yang JR, Wu HSet al.Human infection with avian influenza A H6N1 virus: an epidemiological analysis. Lancet Respir Med 2013;1: 771–778.
- Zhao G, Lu X, Gu Xet al.Molecular evolution of the H6 subtype influenza A viruses from poultry in eastern China from 2002 to 2010. Virol J 2011;8: 470.
- Chin PS, Hoffmann E, Webby Ret al.Molecular evolution of H6 influenza viruses from poultry in Southeastern China: prevalence of H6N1 influenza viruses possessing seven A/Hong Kong/156/97 (H5N1)-like genes in poultry. J Virol 2002;76: 507–516.
- Huang K, Bahl J, Fan XHet al.Establishment of an H6N2 influenza virus lineage in domestic ducks in southern China. J Virol 2010;84: 6978–6986.
- Wang G, Deng G, Shi Jet al.H6 influenza viruses pose a potential threat to human health. J Virol 2014;88: 3953–3964.
- Zhang G, Kong W, Qi Wet al.Identification of an H6N6 swine influenza virus in southern China. Infect Genet Evol 2011;11: 1174–1177.
- Su S, Qi W, Chen Jet al.Seroepidemiological evidence of avian influenza A virus transmission to pigs in southern China. J Clin Microbiol 2013;51: 601–602.
- Gambaryan AS, Tuzikov AB, Pazynina GVet al.6-sulfo sialyl Lewis X is the common receptor determinant recognized by H5, H6, H7 and H9 influenza viruses of terrestrial poultry. Virol J 2008;5: 85.
- Dawood FS, Jain S, Finelli Let al.Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009;360: 2605–2615.
- Nagarajan K, Saikumar G, Arya RSet al.Influenza A H1N1 virus in Indian pigs & its genetic relatedness with pandemic human influenza A 2009 H1N1. Indian J Med Res 2010;132: 160–167.
- Pereda A, Cappuccio J, Quiroga MAet al.Pandemic (H1N1) 2009 outbreak on pig farm, Argentina. Emerg Infect Dis 2010;16: 304–307.
- Welsh MD, Baird PM, Guelbenzu-Gonzalo MPet al.Initial incursion of pandemic (H1N1) 2009 influenza A virus into European pigs. Vet Rec 2010;166: 642–645.
- Weingartl HM, Berhane Y, Hisanaga Tet al.Genetic and pathobiologic characterization of pandemic H1N1 2009 influenza viruses from a naturally infected swine herd. J Virol 2010;84: 2245–2256.
- Berhane Y, Ojkic D, Neufeld Jet al.Molecular characterization of pandemic H1N1 influenza viruses isolated from turkeys and pathogenicity of a human pH1N1 isolate in turkeys. Avian Dis 2010;54: 1275–1285.
- Zhou H, Wang C, Yang Yet al.Pandemic (H1N1) 2009 virus in swine herds, People's Republic of China. Emerg Infect Dis 2011;17: 1757–1759.
- Yan JH, Xiong Y, Yi CHet al.Pandemic (H1N1) 2009 virus circulating in pigs, Guangxi, China. Emerg Infect Dis 2012;18: 357–359.
- Chen Y, Zhang J, Qiao Cet al.Co-circulation of pandemic 2009 H1N1, classical swine H1N1 and avian-like swine H1N1 influenza viruses in pigs in China. Infect Genet Evol 2013;13: 331–338.
- Vijaykrishna D, Poon LL, Zhu HCet al.Reassortment of pandemic H1N1/2009 influenza A virus in swine. Science 2010;328: 1529.
- Moreno A, Di Trani L, Faccini Set al.Novel H1N2 swine influenza reassortant strain in pigs derived from the pandemic H1N1/2009 virus. Vet Microbiol 2011;149: 472–477.
- Hoffmann E, Neumann G, Kawaoka Yet al.DNA transfection system for generation of influenza A virus from eight plasmids. Proc Natl Acad Sci USA 2000;97: 6108–6113.
- Sun H, Yang J, Zhang Tet al.Using sequence data to infer the antigenicity of influenza virus. mBio 2013;4: e00230–13.
- Song X, Xia B, Stowell SRet al.Novel fluorescent glycan microarray strategy reveals ligands for galectins. Chem Biol 2009;16: 36–47.
- van Riel D, Munster VJ, de Wit Eet al.H5N1 virus attachment to lower respiratory tract. Science 2006;312: 399.
- Yang G, Li S, Blackmon Set al.Mutation tryptophan to leucine at the position 222 of hemagglutinin could facilitate H3N2 influenza A virus infection in dogs. J Gen Virol 2013;94: 2599–2608.
- Edgar RC.MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 2004;32: 1792–1797.
- Zwickl DJ. Genetic Algorithm Approaches for the Phylogenetic Analysis of Large Biological Sequence Datasets Under the Maximum Likelihood Criterion.Austin, TX: University of Texas at Austin.2006.
- Swofford DL. PAUP*: Phylogenic Analysis Using Parsimony.Sunderland, MA: Sinauer.1998.
- Wan XF, Nguyen T, Davis CTet al.Evolution of highly pathogenic H5N1 avian influenza viruses in Vietnam between 2001 and 2007. PLoS One 2008;3: e3462.
- Yuan J, Zhang L, Kan Xet al.Origin and molecular characteristics of a novel 2013 avian influenza A(H6N1) virus causing human infection in Taiwan. Clin Infect Dis 2013;57: 1367–1368.
- Shi W, Shi Y, Wu Yet al.Origin and molecular characterization of the human-infecting H6N1 influenza virus in Taiwan. Protein Cell 2013;4: 846–853.
- Trock SC, Burke SA, Cox NJ.Development of an influenza virologic risk assessment tool. Avian Dis 2012;56: 1058–1061.
- Kumari K, Gulati S, Smith DFet al.Receptor binding specificity of recent human H3N2 influenza viruses. Virol J 2007;4: 42.
- Chutinimitkul S, Herfst S, Steel Jet al.Virulence-associated substitution D222G in the hemagglutinin of 2009 pandemic influenza A(H1N1) virus affects receptor binding. J Virol 2010;84: 11802–11813.
- Bradley KC, Galloway SE, Lasanajak Yet al.Analysis of influenza virus hemagglutinin receptor binding mutants with limited receptor recognition properties and conditional replication characteristics. J Virol 2011;85: 12387–12398.
- Meisner J, Szretter KJ, Bradley KCet al.Infectivity studies of influenza virus hemagglutinin receptor binding site mutants in mice. J Virol 2008;82: 5079–5083.
- Gamblin SJ, Haire LF, Russell RJet al.The structure and receptor binding properties of the 1918 influenza hemagglutinin. Science 2004;303: 1838–1842.
- Ito T, Suzuki Y, Mitnaul Let al.Receptor specificity of influenza A viruses correlates with the agglutination of erythrocytes from different animal species. Virology 1997;227: 493–499.
- Ovsyannikova IG, White SJ, Albrecht RAet al.Turkey versus guinea pig red blood cells: hemagglutination differences alter hemagglutination inhibition responses against influenza A/H1N1. Viral Immunol 2014;27: 174–178.
- Ni F, Kondrashkina E, Wang Q.Structural and functional studies of influenza virus A/H6 hemagglutinin. PLoS One 2015;10: e0134576.
- Zhang Y, Zhang Q, Kong Het al.H5N1 hybrid viruses bearing 2009/H1N1 virus genes transmit in guinea pigs by respiratory droplet. Science 2013;340: 1459–1463.
- Sun Y, Qin K, Wang Jet al.High genetic compatibility and increased pathogenicity of reassortants derived from avian H9N2 and pandemic H1N1/2009 influenza viruses. Proc Natl Acad Sci USA 2011;108: 4164–4169.
- Imai M, Watanabe T, Hatta Met al.Experimental adaptation of an influenza H5 HA confers respiratory droplet transmission to a reassortant H5 HA/H1N1 virus in ferrets. Nature 2012;486: 420–428.