?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.ABSTRACT
1. The hypothesis that air-classified faba bean protein fraction (FBP) can replace soybean meal (SBM) in pelleted or extruded broiler diets without adverse effect on performance or nutrient digestibility was tested.
2. At 17 d of age, male broilers were randomly distriibuted among four dietary treatments consisting of either SBM or FBP (main dietary protein source) and pelleting or extrusion as processing methods. Treatments had 10 replicate pens containing five birds each.
3. Compared to SBM, birds fed FBP had significantly lower feed intake, less weight gain and had poorer feed conversion.
4. Pellet durability was high (above 92%) for all diets. In pelleted diets, FBP was harder than SBM whereas extruded diets had similar hardness. Pelleting increased water stability compared to extrusion. FBP diets were more water stable than the SBM diets.
5. Gizzard content weight was 2.2-fold higher (P = 0.002) for birds given FBP compared to those fed SBM. The weight of the jejunum and ileum with contents was 1.4-fold lower for the FBP diets, and this effect was larger (P < 0.05) for the extruded FBP diet.
6. Birds fed FBP diets had significantly higher nitrogen digestibility in the lower jejunum and ileum compared to those given SBM diets, while the starch digestibility coefficient was above 0.980 in all treatments.
7. The high nutrient digestibility of FBP diets indicates that the poor performance of the FBP group was due to lower feed intake which was not explained by the differences in pellet durability or hardness. The reduced palatability of the FBP and the longer retention of the FBP diets in the upper gut are more likely to have depressed feed intake.
Introduction
Due to the high protein and energy content and adaptability to various climatic areas of Europe (Crépon et al. Citation2010), faba beans have recently received more attention for use in poultry nutrition (Nalle et al. Citation2011; O’Neill et al. Citation2012; Koivunen et al. Citation2016). Yet, its use in broiler diets as a feed ingredient is limited due to its lower protein content compared to soybean meal (SBM) and to the presence of anti-nutritive factors (ANFs) such as tannins (in the hulls), convicine and vicine (in the cotyledons). These have been associated with reduced protein and amino acids digestibility and poor performance (Gatel Citation1994; Helsper et al. Citation1996; Alonso et al. Citation2000; Vilariño et al. Citation2009). Significant progress in plant breeding combined with dehulling have proven successful strategies to reduce or eliminate some of the ANFs in faba beans, including a large portion of the non-starch polysaccharides (Nalle et al. Citation2010c; Vilariño et al. Citation2009) which are known to have adverse effect on nutrient digestibility (Smits and Annison Citation1996). Other hydrothermal processing techniques, like pelleting or extrusion, have been reported to enhance the nutritional value of faba beans due to heat-induced changes in several nutrient components. For instance, heat processing can increase the availability of protein and starch, through denaturation and gelatinisation, respectively in faba beans, and to deactivate some of heat labile ANFs (Ginste and de Schrijver Citation1998; Alonso et al. Citation2000; Crépon et al. Citation2010). Despite these issues, the use of faba beans is still generally limited, with maximum recommended inclusion level of 200 g/kg diet (Farrell et al. Citation1999; Nalle et al. Citation2010a, Citation2010b; Koivunen et al. Citation2014).
Air classification is a technique for the dry separation of particles of different shapes and densities using a stream of air, for example from finely ground dehulled faba beans, into a protein concentrate (FBP; light fraction) and a denser starchy flour (Vose et al. Citation1976). Air classification has a low physical impact on the particles and allows retention of the native functionality of both protein and starch (Hansen et al. Citation2017).
These locally produced fractions have the potential to be used as novel ingredients for different classes of animals and, at the same time, help reduce the negative environmental impact of SBM imports (Taelman et al. Citation2015). For instance, FBP may partially replace conventional protein sources like SBM, soy protein concentrate, corn gluten and fish meal, without negatively affecting performance or nutrient digestibility when included at 16% of diets given to weaned pigs (Gunawardena et al. Citation2010) or 21% in salmon diets (de Santis et al. Citation2015). However, the latter diet usually contains twice as much protein as that of pigs or broilers. According to current literature, no studies have assessed the effect of complete replacement of SBM with FBP in broiler diets. Thus, the hypothesis that FBP can replace SBM in pelleted or extruded broiler diets without adverse effect on performance or nutrient digestibility was tested.
Materials and methods
According to Polish law and EU directive 2010/63/EU, the experiment conducted within the study did not require approval of the Local Ethical Committee for Experiments on Animals in Poznań.
Processing of main ingredients and experimental diets
The FBP was obtained following air classification of pin-milled dehulled faba beans (white flowering, low-tannin variety) as described by Itani et al. (Citation2020). The SBM was ground to pass through a 1 mm sieve in a hammer mill (Münch-Edelstahl, Wuppertal, Germany licenced by Bliss, USA, 18.5 kW, 3000 RPM) before being mixed with other ingredients. The wheat was pin-milled similarly to the faba beans, but without further processing. The chemical and amino acid composition of the FBP and SBM are shown in and . Experimental SBM- and FBP-based diets were processed at the Centre for Feed Technology, Norwegian University of Life Sciences, Aas, Norway.
Table 1. Analysed chemical composition (g/kg) of dehulled faba beans (FB), faba bean protein fraction (FBP) and soybean meal (SBM)
Table 2. Analysed amino acid* composition (g/kg DM)
Diets were formulated to be isonitrogenous and isoenergetic and to meet or exceed Ross 308 strain average recommendations (Aviagen Citation2019) for the starter and grower periods for major nutrients ( and ). The SBM-based diet has been presented elsewhere (Itani et al. Citation2020) as part of another experiment.
Table 3. Experimental diet composition, analysed and calculated nutrient content
Table 4. Analysed amino acid*composition (g/kg DM) of the diets
The particle size of the diets (mash) is presented in . The diets contained titanium dioxide (TiO2) as a digestibility marker. The mash was steam-conditioned in a double pass pellet-press conditioner (Münch-Edelstahl, Wuppertal, Germany) at 81°C and then pelleted using a pellet press (Münch-Edelstahl, Wuppertal, Germany, 1.2 t/h, 2 × 17 kW, RMP 350) equipped with a 3 mm die (42 mm thickness), at a production rate of 400 for the SBM diet and 200 kg/h for the FBP diet due to reduced flowability of the FBP. Specific energy consumption values (kWh/t) were 38 and 72 for the SBM and FBP-based diets, respectively. Post-pelleting temperatures were 89 and 92°C for the SBM and FBP-based diet, respectively, and were measured by collecting a sample of hot pellets from immediately below the pellet press into an insulated box fitted with a thermometer. The extruded diet was steam heated at 89°C in an extruder pre-conditioner (Bühler BCTC 10, Uzwil, Switzerland) prior to processing in a co-rotating twin-screw extruder (Bühler BCTG 62/20 D with five sections, 72 kW DC, Uzwil, Switzerland) fitted with 12 dies x 3 mm and with a feeder rate of 145 kg/h. The temperatures in the five sections of the extruder were 92, 112, 95, 90 and 64°C for the SBM diet and 95, 110, 100, 95 and 67°C for the FBP diet. Specific mechanical energy values (kWh/t) were around 64 for both diets and die temperatures were 91 and 92°C for the SBM- and FBP- based diets, respectively. Moisture content during extrusion was kept at around 290 g/kg by addition of steam and water (ambient temperature) in amounts of 60 g/kg and 100 g/kg in the conditioner. During pelleting, around 43 g/kg of steam were added in the conditioner to achieve an average total moisture of 150 g/kg. The physical characteristics of the diets are presented in .
Table 5. Physical quality of the experimental diets
Birds, housing and management
One-day-old male broilers (Ross 308) with mean initial body weight of 45.0 g ± 0.55 SD, were allocated to 40 floor pens (1 x 1 m) bedded with chopped wheat straw (7–15 cm length). They were kept in an environmentally-controlled broiler house (PIAST PASZE Sp. z o.o., Experimental Unit no. 0616, Olszowa, Poland) that contained 9000 birds of the same age and origin as those in the experiment. From 1 to 17 d of age, all birds were fed a commercial diet based on wheat, maize and SBM, with crude protein content and AMEn of 237 g/kg and 14.3 MJ/kg, respectively, and calculated available phosphorus and digestible lysine of 5.5 and 13 g/kg DM, respectively. Mean body weight and average feed intake at d 17 were not different between pens assigned to each treatment (data not shown), with values of 740 g ± 20 g SD and 860 g ± 20 g SD, respectively. Further details of bird housing and management are given elsewhere (Itani et al. Citation2020). At 17 d of age, the birds were randomly assigned to one of four dietary treatments, giving 10 replicate pens per treatment with five birds per pen. Due to the low amount of available raw material, the experimental diet would have been insufficient if more birds were to be used per pen. Treatments consisted of SBM- or FBP-based diets in either pelleted or extruded form, constituting a 2 × 2 factorial experiment.
Performance and sample collection
The birds and the feed were weighed on a per pen basis on d 17 and 29 of age. At d 30, 20 randomly selected birds per treatment (two birds/pen) were weighed, killed by cervical dislocation and the gizzard removed, freed from surrounding fat and then weighed both full and empty. Next, using forceps, the jejunum and ileum were clamped at the end of the duodenal loop, at Meckel’s diverticulum and at the ileo-caecal junction to prevent the passage of contents along the intestine, then weighed. Each of the two segments was then divided into two parts of equal length (upper and lower jejunum/ ileum) and the contents of each segment were expressed by gentle manipulation into a pre-weighed plastic container and stored at −20°C until analysis. As described below, samples for enzyme activity and RNA analysis were taken from one bird and the rest of the digesta from both birds were collected and pooled for digestibility analysis. Around 200 mg of representative samples of digesta from the lower jejunum were transferred into a 2 ml tube (Sarstedt AG & Co. KG, Nümbrecht, Germany), frozen on dry ice, and then stored at −80°C until enzyme activity analysis. A cross-section (2 cm in length) was taken from the midpoint of the lower jejunum, rinsed with ice-cold phosphate-buffered saline (PBS) and cut into three sections of less than 4 mm in thickness. These sections were transferred to a corresponding 2 ml Sarstedt tube containing 1.6 ml RNAlater solution (Merck, Germany) and kept at 4°C for 48 hours. The tubes were then stored at −80°C until RNA extraction. The rest of the intestinal contents were freeze-dried, weighed and pooled for the analysis of nutrient contents and TiO2.
RNA extraction, cDNA synthesis, real-time qPCR, primers and gene expression calculation
The RNA extraction, cDNA synthesis and qPCR were carried out as described by Itani et al. (Citation2020). The primers used in the current experiment were sourced from several studies and are shown with the selected genes in . The change in gene expression was calculated using the relative quantification (2−ΔΔCT) method (Livak and Schmittgen Citation2001). Cycle threshold (CT) values from each group were normalised against HMBS (hydroxymethylbilane synthase) as an optimised housekeeping gene (normally distributed and lowest coefficient of variation), and the average ΔCT of the control group (SBM-pelleted) served as the calibrator for each target gene in the treatment groups.
Table 6. Sequences of primers used for quantitative real-time PCR
Chemical and physical analyses
Representative feed samples were ground in a cutting mill (Pulverisette 19, Fritsch Industriestr. 8, 55743 Idar-Oberstein, Germany) through a 0.5 mm sieve. Gross energy (GE) was determined using an adiabatic bomb calorimeter (Parr 6400, Moline, USA) standardised with benzoic acid. Dry matter (DM) and ash content of the feed were determined after drying overnight at 105°C and after 6 h ashing at 550°C, respectively. Nitrogen content was determined by the Dumas method using a Vario El Cube (Elementar Analysensysteme GmbH, Hanau, Germany). Amino acids concentration in the protein ingredients and diets were determined using a Biochrom 30 amino acid analyser (Biochrom Ltd., Cambridge, UK). Ether extract was determined after extraction with 80% petroleum ether and 20% acetone in an accelerated solvent extractor from Dionex (ASE200; Sunnyvale, CA, USA). Fibre content was determined using a fibre analyser system (Ankom200; ANKOM Technologies, Fairport, NY, USA) with filter bags (Ankom F58; ANKOM Technologies, Fairport, NY, USA). Starch content was analysed enzymatically with use of thermostable α-amylase and amylo-glucosidase (Mccleary et al. Citation1994) and TiO2 content was determined as described by Short et al. (Citation1996). Phosphorus was analysed spectrophotometrically according to the method described in European Commission Regulation (EC) No 152/2009 and ISO 6491, and calcium was measured using atomic emission spectroscopy (MP-AES 4200, Agilent Technologies, USA) following microwave digestion. Freeze-dried jejunal and ileal contents were pulverised using a mortar and pestle and analysed in duplicates for nitrogen and TiO2 as described above. Digesta samples from the lower jejunum were prepared as described by Pérez de Nanclares et al. (Citation2017) for the analysis of protein concentration (Bradford Citation1976) and trypsin activity. Trypsin activity in the jejunal chyme was assayed colorimetrically using trypsin commercial assay kits (Abcam, Cambridge, UK) according to manufacturer’s instructions, and expressed as U/mg protein. The particle size distribution of the diets (mash) was determined by the laser diffraction method using a Malvern Mastersizer 2000 (Malvern Instruments Ltd., Worcestershire, UK) as described by Hetland et al. (Citation2002). Pellet durability (PDI) was measured using a Holmen pellet tester (Holmen Chemical Ltd., Borregaard Group, Norfolk, UK), as described by Zimonja and Svihus (Citation2009). Pellet water stability was analysed as described by Baeverfjord et al. (Citation2006) but with minor modifications, whereby the samples were incubated for 30 min at 40°C in a shaking water bath at 30 rpm. Pellet hardness was determined using a Kahl device (AMANDUS KAHL GmbH and Co. KG, Reinbek, Germany) with 20 pellets of equal length per treatment (Thomas and van der Poel Citation1996). Digesta viscosity was measured after centrifugation of upper ileal digesta samples at 6000 × g for 15 min at 10°C. Around 0.25 ml of supernatant was added to a MCR301 rheometer (Paar Physica) with a PP25 measuring cell. The shear rate of 150 1/s and temperature of 40°C were used. Due to the small amount of available supernatant and to minimise evaporation, an evaporation blocker was used in addition to few drops of water around the measuring plate. Readings were recorded every 6 s, and the average of 10 readings/sample was taken.
Calculations
The apparent digestibility coefficients of nutrients were calculated using the following formula: Apparent digestibility coefficient =
Where was the ratio of the nutrient to TiO2 in the diet or in the digesta.
Statistical analysis
Statistical analyses were performed using the general linear models in R using the Rcmdr package version 2.3.2. All data sets were tested for normality using the Shapiro–Wilk test. A two-way analysis of variance (ANOVA) was performed to determine the main effects and interactions of protein sources and processing methods (as independent variables) on growth parameters, feed physical quality, digestive characteristics, nutrient digestibility, gene expression and enzyme activities. Means were separated using the Tukey post-hoc test and the differences between means were considered significant at P < 0.05 and tendencies if P values were between 0.05 and 0.10. Pen was used as the experimental unit for all data.
Results
Growth performance
Compared to SBM diets, birds fed FBP diets consumed 23% less feed (P < 0.001), gained significantly less weight and had poorer feed conversion ratio (FCR) (P < 0.001) ().
Table 7. The effect of dietary protein source1 and processing method on the growth performance2 of male broilers from 17 to 29 d
Physical characteristics of the diets
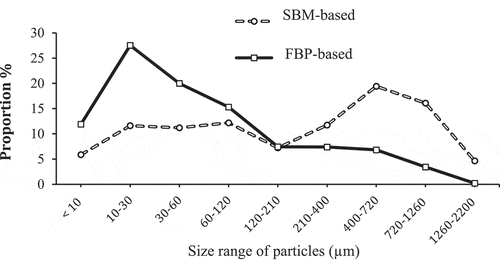
Particle size analysis showed that the FBP diet was finer than the SBM diet, with almost 60% of the particles being smaller than 60 microns. In pelleted diets, FBP had a lower durability than the SBM, however the opposite was true for extruded diets, resulting in a significant interaction (P < 0.001) between protein source and processing method (). Compared to SBM, FBP increased pellet hardness only in pelleted diets, resulting in a significant interaction (P < 0.001) between protein source and processing method. The FBP diets were more (P = 0.003) water stable than the SBM diets, and pelleting resulted in more (P < 0.001) water-stable pellets compared to extrusion.
Gizzard and small intestine characteristics
As shown in , birds fed FBP diets had significantly lower body weight than those fed the SBM diets. Gizzard content weight was 2.2-fold higher for birds given FBP or extruded diets (P < 0.01 and P < 0.05, respectively). Feeding extruded diets increased the relative empty weight of the gizzard, particularly in birds given FBP, resulting in a significant interaction between protein source and processing method. Feeding FBP reduced the weight of the jejunum and ileum with contents by 1.4-fold, and this effect was larger for extruded diets, resulting in an interaction (P < 0.05) between protein source and processing method.
Table 8. The effect of dietary protein source1 and processing method on body weight (g), gizzard characteristics and weight (g) of jejunum and ileum with content of 30-d-old male broilers2.
Apparent intestinal nutrient digestibility, trypsin activity and digesta viscosity
Compared to SBM, FBP had higher (P < 0.001) nitrogen digestibility in the lower jejunum and in the ileum (). Processing methods did not affect (P > 0.05) ileal nitrogen digestibility. Ileal starch digestibility was high (above 0.980) for all diets (data not shown). There was a tendency for higher (P = 0.061) trypsin activity in the jejunal digesta of birds given extruded diets compared to those fed pelleted diets. No difference (P > 0.05) in digesta viscosity was detected between the treatments.
Table 9. The effect of dietary protein source1 and processing method on nitrogen digestion along the intestinal tract, jejunal trypsin activity (U/mg protein) and ileal digesta viscosity (cp) of 30-d-old male broilers2.
Gene expression in the lower jejunum
There was a significant interaction between protein source and processing method on the expression of alanine, serine, cysteine and threonine transporter (ASCT1) (). Thus, feeding SBM resulted in an increase in the expression of ASCT1, but only in pelleted diet. No interaction effects were observed for L type amino acid transporter-1 (LAT1) or peptide transporter-1 (PEPT1) expression. However, feeding SBM significantly upregulated these two transporters by 1.7- and 1.9-fold, respectively compared to FBP.
Table 10. The effect of dietary protein source1 and processing method on the gene expression of nutrient transporters2 in the lower jejunum of 30-d-old male broilers3.
Discussion
The significantly lower feed intake in birds fed FBP diets was accompanied by a marked reduction in BWG and poorer FCR. The lack of mortality during the experimental period and the high nutrient digestibility of the FBP indicated that the poor growth rate in the FBP group was not related to a health problem or to a low nutritive value of the FBP per se, but rather to the lower feed intake compared to the SBM group. Studies have shown that pellet durability and hardness (Abdollahi et al. Citation2018), dietary ANFs (Mawson et al. Citation1993) and feed passage (Dänicke et al. Citation1997; Rochell et al. Citation2012) are some of the major diet-related factors affecting feed intake in broilers.
In the current experiment, pellet durability was high for all diets, with values ranging from 92 to 95%. Therefore, the lower feed intake in the FBP group was not related to a poor pellet durability, as reported by others (Lilly et al. Citation2011). Singh et al. (Citation2014) detected a linear reduction in feed intake with increasing pellet hardness. In this experiment, extruded diets had similar hardness, yet feed intake was still lower for the FBP diet. Thus, the effect of pellet hardness was not consistent and did not explain the lower feed intake.
As mentioned earlier, the highest inclusion rate of faba beans in broiler diets is generally 200 g/kg. Higher inclusion levels caused a reduction in feed intake and BWG (Koivunen et al. Citation2014). The amount of FBP used was equivalent to more than double the recommended level of faba beans, thus, the magnitude of the depression in feed intake appeared logical. Reasons for this may have been related in part to some ANFs which are known to affect feed intake (Iyayi et al. Citation2006) and to concentrate in the FBP fraction (Pitz et al. Citation1980). The concentrated ANFs can result in a significant decline in in-vitro protein digestibility in the FBP fraction (Coda et al. Citation2015). If this was the case, then the negative effects of the ANFs would have been more apparent in the pelleted compared to extruded diets. For instance, if some heat-labile ANFs were not eliminated after pelleting, the more intense conditions during extrusion would have been sufficient to deactivate them (Alonso et al. Citation2000; Hejdysz et al. Citation2016). Although no ANFs were analysed in the current study, feed processing did not affect the apparent nitrogen digestibility (ANiD) of either diet. The FBP diets had higher ANiD compared to SBM, although the trypsin activity was not different between protein sources. This suggested that some undetermined thermostable compound(s) in the FBP fraction may have decreased diet palatability and, thus, feed intake, without reducing nutrient digestibility or affecting enzyme activities.
Berhow et al. (Citation2020) reported that the average total saponin content of 2240 samples of ground whole soybean was 6.5 mg/g, whereas it was found to be higher in white flowering faba beans cultivars, averaging 18.7 mg/g (Revilla Citation2015). Saponin, another heat-stable ANF, is located mainly in the protein bodies of the cotyledon (Frikha et al. Citation2013) and, compared to pulse flour, its concentration can be four-fold higher in the protein-rich fraction (Price et al. Citation1985). The saponin content in faba beans decreased by just 40% following soaking, dehulling and autoclaving for 15 minutes (SHI Et al. Citation2004). In typical commercial feed production, the retention time of feed, when subjected to heat and frictional energy, does not exceed two minutes (Gilpin et al. Citation2002). Thus, it can be hypothesised that saponin was not eliminated in such ‘milder’ processing conditions. Saponin has a very bitter, astringent and metallic taste (Price et al. Citation1985), hence why, high dietary levels depressed feed intake in rats (Cheeke et al. Citation1978), fish (Chen et al. Citation2011), pigs (Thacker and Haq Citation2008) and chickens (Jenkins and Atwal Citation1994; Atuahene et al. Citation1998). Despite this, dietary saponin did not lower the digestibility of amino acids or protein in broilers (Frikha et al. Citation2013) which was in agreement with the current ANiD results. Ivarsson and Wall (Citation2017) reported a 27% reduction in feed intake at an inclusion level of 300 compared to 200 g/kg white-flowered faba beans in a wheat-SBM-based pelleted broiler diets. It appears that the maximum tolerable concentration of saponin is roughly 4 g/kg diet which is equivalent to that found in 200 g/kg faba beans or 50 g/kg FBP, assuming a saponin concentration of 16 g/kg in this fraction.
It was observed that birds fed FBP diets retained considerably more digesta in the gizzard and had lower contents in the jejunum and ileum combined, compared to those given SBM. Although crop weights were not recorded, it appears that the passage rate of FBP diets was slower than SBM, which in turn, limited feed intake (Shires et al. Citation1987). AAS et al. (Citation2011) showed that high pellet water stability reduced feed intake by slowing down gastric evacuation in rainbow trout. Interestingly, FBP diets exhibited higher water stability than the SBM diets, with values being higher in the pelleted diets. In fact, Fernández-Quintela et al. (Citation1997) found that a faba bean protein isolate had higher water and oil absorption capacities and better gelling properties, compared to a soy protein isolate. In addition, the larger surface area of the FBP may have increased this water absorption capacity (Hasjim et al. Citation2013) and, potentially, the water stability of the FBP diets (Obaldo et al. Citation1999). Although extrusion resulted in lower water stability than pelleting, the consumption of the extruded FBP diet was still significantly lower. This indicated that other factors were responsible for the reduced feed passage, despite the relatively lower water stability of the extruded FBP diet. Matsuda et al. (Citation1999b) found that dietary saponin isolated from medicinal herbs inhibited gastric emptying in mice, resulting in significant increases in the weight of the stomach. In his review, Cheeke (Citation1971) reported that the intra-ruminal or intra-venous administration of alfalfa saponin resulted in a pronounced reduction in rumen motility in sheep. Abu Hafsa et al. (Citation2015) observed a 19% reduction in feed intake in broilers given diets containing on average 8.3 g/kg saponin, after partial replacement of SBM with guar korma meal, and attributed the low feed intake to the inhibitory effect of saponin on gastric emptying. In agreement with the above, Ueda et al. (Citation1996) reported that 10 g/kg alfalfa saponin in chicks diet delayed feed passage time by 50% (measured by chromic oxide excretion), and noted that the crops of saponin-fed birds contained more ingesta. The mechanism by which saponin inhibits gastric emptying is not clear, although it may be related to the ability of saponin to stimulate the release of secretin and cholecystokinin, which are known to inhibit gastric emptying (Matsuda et al. Citation1999a).
Jejunal digesta viscosity values were low and in line with other studies (Gao et al. Citation2008), and were not affected by either protein source or processing method. The reasons for this may have been related to the inclusion of NSP-degrading enzymes and/or to the content of soluble NSP that was not high enough to affect digesta viscosity or feed intake.
The FBP diets had a higher ANiD compared to SBM possibly due to the finer particle size of the FBP fraction. Corroborating this, broilers fed finely milled pea seeds exhibited higher ANiD compared to those fed coarsely ground peas (Crévieu et al. Citation1997). In addition, micronisation of faba beans was found to improve nitrogen retention compared to coarser grinding (Mcnab and Wilson Citation1974). However, Valencia et al. (Citation2009) and Pacheco et al. (Citation2013) did not detect any improvement in protein digestibility by decreasing SBM particle size. Moreover, intestinal nutrient digestibility may be affected by the amount of substrate flowing into the small intestine, which is dependent on feed intake (Péron et al. Citation2005; Svihus et al. Citation2010). Accordingly, a significant increase in ileal ANiD coincided with a lower feed intake in broilers as a result of feeding a mash compared to pelleted diets (Abdollahi et al. Citation2011, Citation2013; Naderinejad et al. Citation2016).
The higher expression of nutrient transporters in the SBM group was counterintuitive, because FBP had higher ANiD and an upregulation of nutrient transporters would be expected with higher amount of available substrates (Cowieson et al. Citation2019). However, the higher volume of digesta in the small intestine of birds fed SBM may have provided more luminal stimulation, thereby causing this upregulation of nutrient transporters (Chen et al. Citation2005; Mott et al. Citation2008; Gilbert et al. Citation2010).
In conclusion, substituting SBM for FBP in pelleted or extruded broiler diets resulted in reduced feed consumption, lower weight gain and poorer FCR. The high nutrient digestibility of FBP diets indicated that the slower growth rate was due to the lower feed intake in the FBP group. Differences in pellet durability or hardness did not explain the lower feed intake, however, reduced palatability of the FBP and the longer retention of the FBP diets in the upper gut were likely to have caused such effect.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Aas, T., B. Terjesen, T. Sigholt, M. Hillestad, J. Holm, S. Refstie, G. Baeverfjord, K. A. Rørvik, M. Sørensen, and M. Oehme. 2011. “Nutritional Responses in Rainbow Trout (Oncorhynchus Mykiss) Fed Diets with Different Physical Qualities at Stable or Variable Environmental Conditions.” Aquaculture Nutrition 17: 657–670. doi:https://doi.org/10.1111/j.1365-2095.2011.00868.x.
- Abdollahi, M., V. Ravindran, and B. Svihus. 2013. “Influence of Grain Type and Feed Form on Performance, Apparent Metabolisable Energy and Ileal Digestibility of Nitrogen, Starch, Fat, Calcium and Phosphorus in Broiler Starters.” Animal Feed Science and Technology 186: 193–203. doi:https://doi.org/10.1016/j.anifeedsci.2013.10.015.
- Abdollahi, M., V. Ravindran, T. Wester, G. Ravindran, and D. Thomas. 2011. “Influence of Feed Form and Conditioning Temperature on Performance, Apparent Metabolisable Energy and Ileal Digestibility of Starch and Nitrogen in Broiler Starters Fed Wheat-based Diet.” Animal Feed Science and Technology 168: 88–99. doi:https://doi.org/10.1016/j.anifeedsci.2011.03.014.
- Abdollahi, M., F. Zaefarian, and V. Ravindran. 2018. “Feed Intake Response of Broilers: Impact of Feed Processing.” Animal Feed Science and Technology 237: 154–165. doi:https://doi.org/10.1016/j.anifeedsci.2018.01.013.
- Abu Hafsa, S., M. Basyony, and A. Hassan. 2015. “Effect of Partial Replacement of Soybean Meal with Different Levels of Guar Korma Meal on Growth Performance, Carcass Traits and Blood Metabolites of Broiler Chickens.” Asian Journal of Poultry Science 9 (3): 112–122. doi:https://doi.org/10.3923/ajpsaj.2015.112.122.
- Alonso, R., A. Aguirre, and F. Marzo. 2000. “Effects of Extrusion and Traditional Processing Methods on Antinutrients and in Vitro Digestibility of Protein and Starch in Faba and Kidney Beans.” Food Chemistry 68: 159–165. doi:https://doi.org/10.1016/S0308-8146(99)00169-7.
- Atuahene, C., A. Donkoh, and F. Asante. 1998. “Value of Sheanut Cake as a Dietary Ingredient for Broiler Chickens.” Animal Feed Science and Technology 72: 133–142. doi:https://doi.org/10.1016/S0377-8401(97)00172-7.
- Aviagen. 2019. ROSS Nutrition Specifications. Newbridge, UK: Aviagen. https://en.aviagen.com/assets/Tech_Center/Ross_Broiler/RossBroilerNutritionSpecs2019-EN.pdf
- Baeverfjord, G., S. Refstie, P. Krogedal, and T. Åsgård. 2006. “Low Feed Pellet Water Stability and Fluctuating Water Salinity Cause Separation and Accumulation of Dietary Oil in the Stomach of Rainbow Trout (Oncorhynchus Mykiss).” Aquaculture 261: 1335–1345. doi:https://doi.org/10.1016/j.aquaculture.2006.08.033.
- Berhow, M. A., M. Singh, M. J. Bowman, N. P. Price, S. F. Vaughn, and S. X. Liu. 2020. “Quantitative NIR Determination of Isoflavone and Saponin Content of Ground Soybeans.” Food Chemistry 317: 126373. doi:https://doi.org/10.1016/j.foodchem.2020.126373.
- Bradford, M. M. 1976. “A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-dye Binding.” Analytical Biochemistry 72: 248–254. doi:https://doi.org/10.1016/0003-2697(76)90527-3.
- Cheeke, P. 1971. “Nutritional and Physiological Implications of Saponins: A Review.” Canadian Journal of Animal Science 51: 621–632. https://cdnsciencepub.com/doi/pdf/10.4141/cjas71-082
- Cheeke, P., D. England, and M. Pedersen. 1978. “Responses of Rats and Swine to Alfalfa Saponins.” Canadian Journal of Animal Science 58: 783–789. doi:https://doi.org/10.4141/cjas78-097.
- Chen, H., Y. Pan, E. A. Wong, and K. E. Webb JR. 2005. “Dietary Protein Level and Stage of Development Affect Expression of an Intestinal Peptide Transporter (Cpept1) in Chickens.” The Journal of Nutrition 135: 193–198. doi:https://doi.org/10.1093/jn/135.2.193.
- Chen, W., Q. Ai, K. Mai, W. Xu, Z. Liufu, W. Zhang, and Y. Cai. 2011. “Effects of Dietary Soybean Saponins on Feed Intake, Growth Performance, Digestibility and Intestinal Structure in Juvenile Japanese Flounder (Paralichthys Olivaceus).” Aquaculture 318: 95–100. doi:https://doi.org/10.1016/j.aquaculture.2011.04.050.
- Coda, R., L. Melama, C. G. Rizzello, J. A. Curiel, J. Sibakov, U. Holopainen, M. Pulkkinen, and N. Sozer. 2015. “Effect of Air Classification and Fermentation by Lactobacillus Plantarum VTT E-133328 on Faba Bean (Vicia Faba L.) Flour Nutritional Properties.” International Journal of Food Microbiology 193: 34–42. doi:https://doi.org/10.1016/j.ijfoodmicro.2014.10.012.
- Cowieson, A., M. Toghyani, S. Kheravii, S.-B. Wu, L. Romero, and M. Choct. 2019. “A Mono-component Microbial Protease Improves Performance, Net Energy, and Digestibility of Amino Acids and Starch, and Upregulates Jejunal Expression of Genes Responsible for Peptide Transport in Broilers Fed Corn/wheat-based Diets Supplemented with Xylanase and Phytase.” Poultry Science 98: 1321–1332. doi:https://doi.org/10.3382/ps/pey456.
- Crépon, K., P. Marget, C. Peyronnet, B. Carrouee, P. Arese, and G. Duc. 2010. “Nutritional Value of Faba Bean (Vicia Faba L.) Seeds for Feed and Food.” Field Crops Research 115: 329–339. doi:https://doi.org/10.1016/j.fcr.2009.09.016.
- Crévieu, I., B. Carré, A. M. Chagneau, J. Guéguen, and J. P. Melcion. 1997. “Effect of Particle Size of Pea (Pisum sativumL) Flours on the Digestion of Their Proteins in the Digestive Tract of Broilers.” Journal of the Science of Food and Agriculture 75: 217–226. doi:https://doi.org/10.1002/(SICI)1097-0010(199710)75:2<217::AID-JSFA867>3.0.CO;2-O.
- Dänicke, S., O. Simon, H. Jeroch, and M. Bedford. 1997. “Interactions between Dietary Fat Type and Xylanase Supplementation When Rye‐based Diets are Fed to Broiler Chickens. 1. Physicochemical Chyme Features.” British Poultry Science 38: 537–545. doi:https://doi.org/10.1080/00071669708418034.
- de Santis, C., K. Ruohonen, D. R. Tocher, S. A. Martin, E. Krol, C. J. Secombes, J. G. Bell, A. El-mowafi, and V. Crampton. 2015. “Atlantic Salmon (Salmo Salar) Parr as a Model to Predict the Optimum Inclusion of Air Classified Faba Bean Protein Concentrate in Feeds for Seawater Salmon.” Aquaculture 444: 70–78. doi:https://doi.org/10.1016/j.aquaculture.2015.03.024.
- Farrell, D., R. Perez-maldonado, and P. Mannion. 1999. “Optimum Inclusion of Field Peas, Faba Beans, Chick Peas and Sweet Lupins in Poultry Diets. II. Broiler Experiments.” British Poultry Science 40: 674–680. doi:https://doi.org/10.1080/00071669987070.
- Fernández-Quintela, A., M. Macarulla, A. Del Barrio, and J. Martínez. 1997. “Composition and Functional Properties of Protein Isolates Obtained from Commercial Legumes Grown in Northern Spain.” Plant Foods for Human Nutrition 51: 331–341. doi:https://doi.org/10.1023/A:1007936930354.
- Frikha, M., D. Valencia, A. de Coca-Sinova, R. Lázaro, and G. Mateos. 2013. “Ileal Digestibility of Amino Acids of Unheated and Autoclaved Pea Protein Concentrate in Broilers.” Poultry Science 92: 1848–1857. doi:https://doi.org/10.3382/ps.2013-03007.
- Gao, F., Y. Jiang, G. Zhou, and Z. Han. 2008. “The Effects of Xylanase Supplementation on Performance, Characteristics of the Gastrointestinal Tract, Blood Parameters and Gut Microflora in Broilers Fed on Wheat-based Diets.” Animal Feed Science and Technology 142: 173–184. doi:https://doi.org/10.1016/j.anifeedsci.2007.07.008.
- Gatel, F. 1994. “Protein Quality of Legume Seeds for Non-ruminant Animals: A Literature Review.” Animal Feed Science and Technology 45: 317–348. doi:https://doi.org/10.1016/0377-8401(94)90036-1.
- Gilbert, E., H. Li, D. Emmerson, K. Webb JR, and E. Wong. 2010. “Dietary Protein Composition Influences Abundance of Peptide and Amino Acid Transporter Messenger Ribonucleic Acid in the Small Intestine of 2 Lines of Broiler Chicks.” Poultry Science 89: 1663–1676. doi:https://doi.org/10.3382/ps.2010-00801.
- Gilpin, A., T. Herrman, K. Behnke, and F. Fairchild. 2002. “Feed Moisture, Retention Time, and Steam as Quality and Energy Utilization Determinants in the Pelleting Process.” Applied Engineering in Agriculture 18: 331–340. doi:https://doi.org/10.13031/2013.8585.
- Ginste, J. V., and R. de Schrijver. 1998. “Expansion and Pelleting of Starter, Grower and Finisher Diets for Pigs: Effects on Nitrogen Retention, Ileal and Total Tract Digestibility of Protein, Phosphorus and Calcium and in Vitro Protein Quality.” Animal Feed Science and Technology 72: 303–314. doi:https://doi.org/10.1016/S0377-8401(97)00192-2.
- Gunawardena, C., R. Zijlstra, L. Goonewardene, and E. Beltranena. 2010. “Protein and Starch Concentrates of Air-classified Field Pea and Zero-tannin Faba Bean for Weaned Pigs.” Journal of Animal Science 88: 2627–2636. doi:https://doi.org/10.2527/jas.2009-2291.
- Hansen, J. Ø., A. Skrede, L. T. Mydland, and M. Øverland. 2017. “Fractionation of Rapeseed Meal by Milling, Sieving and Air classification—Effect on Crude Protein, Amino Acids and Fiber Content and Digestibility.” Animal Feed Science and Technology 230: 143–153. doi:https://doi.org/10.1016/j.anifeedsci.2017.05.007.
- Hasjim, J., E. Li, and S. Dhital. 2013. “Milling of Rice Grains: Effects of Starch/flour Structures on Gelatinization and Pasting Properties.” Carbohydrate Polymers 92: 682–690. doi:https://doi.org/10.1016/j.carbpol.2012.09.023.
- Hejdysz, M., S. Kaczmarek, and A. Rutkowski. 2016. “Extrusion Cooking Improves the Metabolisable Energy of Faba Beans and the Amino Acid Digestibility in Broilers.” Animal Feed Science and Technology 212: 100–111. doi:https://doi.org/10.1016/j.anifeedsci.2015.12.008.
- Helsper, J. P., Y. P. van Loon, R. P. Kwakkel, A. van N#, and A. F. van der Poel. 1996. “Growth of Broiler Chicks Fed Diets Containing Tannin-free and Tannin-containing Near-isogenic Lines of Faba Bean (Vicia Faba L.).” Journal of Agricultural and Food Chemistry 44: 1070–1075. doi:https://doi.org/10.1021/jf950484w.
- Hetland, H., B. Svihus, and V. Olaisen. 2002. “Effect of Feeding Whole Cereals on Performance, Starch Digestibility and Duodenal Particle Size Distribution in Broiler Chickens.” British Poultry Science 43: 416–423. doi:https://doi.org/10.1080/00071660120103693.
- Itani, K., J. Ø. Hansen, B. Kieronczyk, A. Benzertiha, P. P. Kurk, R. M. Ånestad, F. Sundby, L. T. Mydland, M. Øverland, and B. Svihus. 2020. “Interactions between Starch Source and Gelatinisation Degree on Performance and Small Intestinal Digestion in Broiler Chickens.” Submitted to British Poultry Science 62 (3): 424–434. doi:https://doi.org/10.1080/00071668.2020.1868406.
- Ivarsson, E., and H. Wall. 2017. “Effects of Toasting, Inclusion Levels and Different Enzyme Supplementations of Faba Beans on Growth Performance of Broiler Chickens.” Journal of Applied Poultry Research 26: 467–475. doi:https://doi.org/10.3382/japr/pfx016.
- Iyayi, E. A., H. Kluth, and M. Rodehutscord. 2006. “Chemical Composition, Antinutritional Constituents, Precaecal Crude Protein and Amino Acid Digestibility in Three Unconventional Tropical Legumes in Broilers.” Journal of the Science of Food and Agriculture 86: 2166–2171. doi:https://doi.org/10.1002/jsfa.2592.
- Jenkins, K. J., and A. ATWAL. 1994. “Effects of Dietary Saponins on Fecal Bile Acids and Neutral Sterols, and Availability of Vitamins A and E in the Chick.” The Journal of Nutritional Biochemistry 5: 134–137. doi:https://doi.org/10.1016/0955-2863(94)90084-1.
- Kaminski, N., and E. Wong. 2018. “Differential mRNA Expression of Nutrient Transporters in Male and Female Chickens.” Poultry Science 97: 313–318. doi:https://doi.org/10.3382/ps/pex262.
- Koivunen, E., K. Partanen, S. Perttilä, S. Palander, P. Tuunainen, and J. Valaja. 2016. “Digestibility and Energy Value of Pea (Pisum Sativum L.), Faba Bean (Vicia Faba L.) And Blue Lupin (Narrow-leaf)(lupinus Angustifolius) Seeds in Broilers.” Animal Feed Science and Technology 218: 120–127. doi:https://doi.org/10.1016/j.anifeedsci.2016.05.007.
- Koivunen, E., P. Tuunainen, L. Rossow, and J. Valaja. 2014. “Digestibility and Utilization of Faba Bean (Vicia Faba L.) Diets in Broilers.” Acta Agriculturae Scandinavica, Section A—Animal Science 64: 217–225. doi:https://doi.org/10.1080/09064702.2015.1020857.
- Lilly, K., C. Gehring, K. Beaman, P. Turk, M. Sperow, and J. Moritz. 2011. “Examining the Relationships between Pellet Quality, Broiler Performance, and Bird Sex.” Journal of Applied Poultry Research 20: 231–239. doi:https://doi.org/10.3382/japr.2009-00138.
- Livak, K. J., and T. D. Schmittgen. 2001. “Analysis of Relative Gene Expression Data Using Real-time Quantitative PCR and the 2− ΔΔCT Method.” methods 25: 402–408. doi:https://doi.org/10.1006/meth.2001.1262.
- Matsuda, H., Y. Li, T. Murakami, J. Yamahara, and M. Yoshikawa. 1999b. “Structure-related Inhibitory Activity of Oleanolic Acid Glycosides on Gastric Emptying in Mice.” Bioorganic & Medicinal Chemistry 7: 323–327. doi:https://doi.org/10.1016/S0968-0896(98)00207-7.
- Matsuda, H., Y. Li, J. Yamahara, and M. Yoshikawa. 1999a. “Inhibition of Gastric Emptying by Triterpene Saponin, Momordin Ic, in Mice: Roles of Blood Glucose, Capsaicin-sensitive Sensory Nerves, and Central Nervous System.” Journal of Pharmacology and Experimental Therapeutics 289: 729–734. https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.1064.9766&rep=rep1&type=pdf
- Mawson, R., R. Heaney, Z. Zdunczyk, and H. J. F. N. Kozlowska. 1993. “Rapeseed Meal‐glucosinolates and Their Antinutritional Effects. Part II.” Flavour and Palatability 37: 336–344. doi:https://doi.org/10.1002/food.19930370405.
- Mccleary, B., V. SOLAH, and T. Gibson. 1994. “Quantitative Measurement of Total Starch in Cereal Flours and Products.” Journal of Cereal Science 20: 51–58. doi:https://doi.org/10.1006/jcrs.1994.1044.
- Mcnab, J. M., and B. J. Wilson. 1974. “Effects of Micronising on the Utilisation of Field Beans (Vicia Faba L.) By the Young Chick.” Journal of the Science of Food and Agriculture 25: 395–400. doi:https://doi.org/10.1002/jsfa.2740250407.
- Mott, C., P. Siegel, K. Webb JR, and E. Wong. 2008. “Gene Expression of Nutrient Transporters in the Small Intestine of Chickens from Lines Divergently Selected for High or Low Juvenile Body Weight.” Poultry Science 87: 2215–2224. doi:https://doi.org/10.3382/ps.2008-00101.
- Naderinejad, S., F. Zaefarian, M. Abdollahi, A. Hassanabadi, H. Kermanshahi, and V. Ravindran. 2016. “Influence of Feed Form and Particle Size on Performance, Nutrient Utilisation, and Gastrointestinal Tract Development and Morphometry in Broiler Starters Fed Maize-based Diets.” Animal Feed Science and Technology 215: 92–104. doi:https://doi.org/10.1016/j.anifeedsci.2016.02.012.
- Nalle, C., V. Ravindran, and G. Ravindran. 2010a. “Nutritional Value of Faba Beans (Vicia Faba L.) For Broilers: Apparent Metabolisable Energy, Ileal Amino Acid Digestibility and Production Performance.” Animal Feed Science and Technology 156: 104–111. doi:https://doi.org/10.1016/j.anifeedsci.2010.01.010.
- Nalle, C., V. Ravindran, and G. Ravindran. 2010b. “Evaluation of Faba Beans, White Lupins and Peas as Protein Sources in Broiler Diets.” International Journal of Poultry Science 9: 567–573. doi:https://doi.org/10.3923/ijps.2010.567.573.
- Nalle, C., V. Ravindran, and G. Ravindran. 2011. “Nutritional Value of Peas (Pisum Sativum L.) For Broilers: Apparent Metabolisable Energy, Apparent Ileal Amino Acid Digestibility and Production Performance.” Animal Production Science 51: 150–155. doi:https://doi.org/10.1071/AN10100.
- Nalle, C. L., G. Ravindran, and V. Ravindran. 2010c. “Influence of Dehulling on the Apparent Metabolisable Energy and Ileal Amino Acid Digestibility of Grain Legumes for Broilers.” Journal of the Science of Food and Agriculture 90: 1227–1231. doi:https://doi.org/10.1002/jsfa.3953.
- O’Neill, H. M., M. Rademacher, I. Mueller-harvey, E. Stringano, S. Kightley, and J. Wiseman. 2012. “Standardised Ileal Digestibility of Crude Protein and Amino Acids of UK-grown Peas and Faba Beans by Broilers.” Animal Feed Science and Technology 175: 158–167. doi:https://doi.org/10.1016/j.anifeedsci.2012.05.004.
- Obaldo, L. G., W. G. Dominy, J. H. Terpstra, J. J. Cody, and K. C. Behnke. 1999. “The Impact of Ingredient Particle Size on Shrimp Feed.” Journal of Applied Aquaculture 8: 55–67. doi:https://doi.org/10.1300/J028v08n04_07.
- Pacheco, W., C. Stark, P. Ferket, and J. Brake. 2013. “Evaluation of Soybean Meal Source and Particle Size on Broiler Performance, Nutrient Digestibility, and Gizzard Development.” Poultry Science 92: 2914–2922. doi:https://doi.org/10.3382/ps.2013-03186.
- Paris, N., and E. Wong. 2013. “Expression of Digestive Enzymes and Nutrient Transporters in the Intestine of Eimeria Maxima-infected Chickens.” Poultry Science 92: 1331–1335. doi:https://doi.org/10.3382/ps.2012-02966.
- Pérez de Nanclares, M., M. Trudeau, J. Hansen, L. Mydland, P. Urriola, G. Shurson, C. Åkesson, N. Kjos, M. Arntzen, and M. Øverland. 2017. “High-fiber Rapeseed Co-product Diet for Norwegian Landrace Pigs: Effect on Digestibility.” Livestock Science 203: 1–9. doi:https://doi.org/10.1016/j.livsci.2017.06.008.
- Péron, A., D. Bastianelli, F.-X. Oury, J. Gomez, and B. Carré. 2005. “Effects of Food Deprivation and Particle Size of Ground Wheat on Digestibility of Food Components in Broilers Fed on a Pelleted Diet.” British Poultry Science 46: 223–230. doi:https://doi.org/10.1080/00071660500066142.
- Pitz, W., F. Sosulski, and L. Hogge. 1980. “Occurrence of Vicine and Convicine in Seeds of Some Vicia Species and Other Pulses.” Canadian Institute of Food Science and Technology Journal 13: 35–39. doi:https://doi.org/10.1016/S0315-5463(80)73300-X.
- Price, K., N. Griffiths, C. Curl, and G. Fenwick. 1985. “Undesirable Sensory Properties of the Dried Pea (Pisum Sativum). The Role of Saponins.” Food Chemistry 17: 105–115. doi:https://doi.org/10.1016/0308-8146(85)90079-2.
- Revilla, I. 2015. “Impact of Thermal Processing on Faba Bean (Vicia Faba) Composition.” In Processing and Impact on Active Components in Food, edited by V. Preedy, 337–343. UK, London: Elsevier.
- Rochell, S., T. Applegate, E. Kim, and W. Dozier III. 2012. “Effects of Diet Type and Ingredient Composition on Rate of Passage and Apparent Ileal Amino Acid Digestibility in Broiler Chicks.” Poultry Science 91: 1647–1653. doi:https://doi.org/10.3382/ps.2012-02173.
- Sauvant, D., J. M. Perez, and G. Tran. 2004. Tables of Composition and Nutritional Value of Feed Materials: Pigs, Poultry, Cattle, Sheep, Goats, Rabbits, Horses and Fish. The Netherlands, Wageningen: Wageningen Academic Publishers.
- Shi, J., K. Arunasalam, D. Yeung, Y. Kakuda, G. Mittal, and Y. Jiang. 2004. “Saponins from Edible Legumes: Chemistry, Processing, and Health Benefits.” Journal of Medicinal Food 7: 67–78. doi:https://doi.org/10.1089/109662004322984734.
- Shires, A., J. Thompson, B. Turner, P. Kennedy, and Y. Goh. 1987. “Rate of Passage of Corn-canola Meal and Corn-soybean Meal Diets through the Gastrointestinal Tract of Broiler and White Leghorn Chickens.” Poultry Science 66: 289–298. doi:https://doi.org/10.3382/ps.0660289.
- Short, F., P. Gorton, J. Wiseman, and K. Boorman. 1996. “Determination of Titanium Dioxide Added as an Inert Marker in Chicken Digestibility Studies.” Animal Feed Science and Technology 59: 215–221. doi:https://doi.org/10.1016/0377-8401(95)00916-7.
- Singh, Y., V. Ravindran, T. Wester, A. Molan, and G. Ravindran. 2014. “Influence of Prepelleting Inclusion of Whole Corn on Performance, Nutrient Utilization, Digestive Tract Measurements, and Cecal Microbiota of Young Broilers.” Poultry Science 93: 3073–3082. doi:https://doi.org/10.3382/ps.2014-04110.
- Smits, C. H., and G. Annison. 1996. “Non-starch Plant Polysaccharides in Broiler Nutrition–towards a Physiologically Valid Approach to Their Determination.” World’s Poultry Science Journal 52: 203–221. doi:https://doi.org/10.1079/WPS19960016.
- Su, S., K. Miska, R. Fetterer, M. Jenkins, and E. Wong. 2014. “Expression of Digestive Enzymes and Nutrient Transporters in Eimeria Acervulina-challenged Layers and Broilers.” Poultry Science 93: 1217–1226. doi:https://doi.org/10.3382/ps.2013-03807.
- Svihus, B., A. Sacranie, V. Denstadli, and M. Choct. 2010. “Nutrient Utilization and Functionality of the Anterior Digestive Tract Caused by Intermittent Feeding and Inclusion of Whole Wheat in Diets for Broiler Chickens.” Poultry Science 89: 2617–2625. doi:https://doi.org/10.3382/ps.2010-00743.
- Taelman, S. E., S. de Meester, W. van Dijk, V. da Silva, and J. Dewulf. 2015. “Environmental Sustainability Analysis of a Protein-rich Livestock Feed Ingredient in the Netherlands: Microalgae Production versus Soybean Import.” Resources, Conservation and Recycling 101: 61–72. doi:https://doi.org/10.1016/j.resconrec.2015.05.013.
- Teng, P.-Y., S. Yadav, F. L. de Souza Castro, Y. H. Tompkins, A. L. Fuller, and W. K. Kim. 2020. “Graded Eimeria Challenge Linearly Regulated Growth Performance, Dynamic Change of Gastrointestinal Permeability, Apparent Ileal Digestibility, Intestinal Morphology, and Tight Junctions of Broiler Chickens.” Poultry Science 99: 4203–4216. doi:https://doi.org/10.1016/j.psj.2020.04.031.
- Thacker, P. A., and I. HAQ. 2008. “Nutrient Digestibility, Performance and Carcass Traits of Growing–finishing Pigs Fed Diets Containing Graded Levels of Dehydrated Lucerne Meal.” Journal of the Science of Food and Agriculture 88: 2019–2025. doi:https://doi.org/10.1002/jsfa.3314.
- Thomas, M., and A. Van Der Poel. 1996. “Physical Quality of Pelleted Animal Feed 1. Criteria for Pellet Quality.” Animal Feed Science and Technology 61: 89–112. doi:https://doi.org/10.1016/0377-8401(96)00949-2.
- Ueda, H., Y. Kakutou, and M. Ohshima. 1996. “Growth-depressing Effect of Alfalfa Saponin in Chicks.” Animal Science and Technology (Japan). doi:https://doi.org/10.2508/chikusan.67.772.
- Valencia, D., M. Serrano, R. Lázaro, E. Jiménez-Moreno, and G. Mateos. 2009. “Influence of Micronization (Fine Grinding) of Soya Bean Meal and Full-fat Soya Bean on the Ileal Digestibility of Amino Acids for Broilers.” Animal Feed Science and Technology 150: 238–248. doi:https://doi.org/10.1016/j.anifeedsci.2008.08.010.
- Vilariño, M., J. Métayer, K. Crépon, and G. Duc. 2009. “Effects of Varying Vicine, Convicine and Tannin Contents of Faba Bean Seeds (Vicia Faba L.) On Nutritional Values for Broiler Chicken.” Animal Feed Science and Technology 150: 114–121. doi:https://doi.org/10.1016/j.anifeedsci.2008.08.001.
- Vose, J., M. Basterrechea, P. Gorin, A. Finlayson, and C. Youngs. 1976. “Air Classification of Field Peas and Horsebean Flours: Chemical Studies of Starch and Protein Fractions.” Cereal Chem 53: 928–936. http://www.aaccnet.org/publications/cc/backissues/1976/Documents/chem53_928.pdf
- Zimonja, O., and B. Svihus. 2009. “Effects of Processing of Wheat or Oats Starch on Physical Pellet Quality and Nutritional Value for Broilers.” Animal Feed Science and Technology 149: 287–297. doi:https://doi.org/10.1016/j.anifeedsci.2008.06.010.