ABSTRACT
The genus Cheiromycina is one of the few genera of lichenized hyphomycetes for which no sexual reproductive stages have been observed. The genus includes species from boreal to temperate regions of the Northern Hemisphere where it is found growing on bark or wood. Congeners in Cheiromycina are characterized by a noncorticate thallus, nearly immersed in the substrate and presenting powdery unpigmented sporodochia, and containing chlorococcoid photobionts. The relationships of members of Cheiromycina with other fungi are not known. Here we inferred the phylogenetic placement of Cheiromycina using three loci (nuSSU, nuLSU, and mtSSU) representing C. flabelliformis, the type species for the genus, C. petri, and C. reimeri. Our results revealed that the genus Cheiromycina is found within the family Malmideaceae (Lecanorales) where members formed a monophyletic clade sister to the genera Savoronala and Malmidea. This phylogenetic placement and the relationships of Cheiromycina with other lichenized hyphomycetous taxa are here discussed.
INTRODUCTION
Hyphomycetes comprises a polyphyletic assemblage generally recognized by mycologists as one of the major group of anamorphic fungi, including the majority of what are commonly called molds. Fungi generally characterizes as members of Hyphomycetes include parasitic and saprotrophic species growing on diverse natural substrates, such as wood, plant tissues, insects, and other arthropods, and other fungi, including lichens (Seifert et al. Citation2011). Hyphomycetes represent fungi in asexual stages, and, in many cases, members have subsequently been recognized as anamorphs of several ascomycete and basidiomycete lineages. However, taxa for which no teleomorph has been identified have been hypothesized to have permanently lost the ability of developing sexual structures and are regarded as anamorphic holomorphs (Seifert et al. Citation2011).
Historically, two successful sorting systems for anamorphic fungi were developed: the sporological system (Saccardo Citation1886), which focused on conidiomata morphology, and the ontogenetic system (Hughes Citation1953), which used the conidium ontogeny as a primary diagnostic character. Because Saccardo’s sporological system based on conidiomata morphology has been recognized as artificial, and the conidium ontogeny still groups unrelated taxa, Berbee and Taylor (Citation1993) suggested integrating all anamorphic fungi into a unique fungal systematic scheme, which was further expanded by Reynolds and Taylor (Citation1993). More recently, anamorphic fungi are distinguished into three major groups according to the presence and the structures in which conidiospores are produced: (i) blastomycetes, such as the asexually reproducing yeasts; (ii) coelomycetes, fungi producing pycnidia and acervuli; (iii) hyphomycetes, fungi lacking pycnidia or acervular fruting bodies and with conidia developed “out in the air” (Seifert et al. Citation2011).
In spite of these attempts for a natural classification, the debate for the classification of many anamorphic genera remains unsolved. Recent application of molecular- and culture-based analyses have helped resolving many anamorph-teleomorph connections (e.g., Crous et al. Citation2001, Citation2004, Citation2006; Lizel et al. Citation2003; Réblová and Seifert Citation2004, Citation2011; Huhndorf and Fernández Citation2005; Shenoy et al. Citation2007; Ertz et al. Citation2011, Citation2013, Citation2014; Pérez-Ortega et al. Citation2011; Muggia et al. 2015).
Lichenicolous hyphomycetes are recognized within both coelomycetes and hyphomycetes and comprise multiple genera that are known parasites of lichen thalli (Diederich Citation2011). After Hawksworth’s (Citation1979) initial treatment, ongoing research continues to contribute to understanding diversity of lichenicolous hyphomycetes (e.g., Hawksworth and Poelt Citation1986; Diederich and Scheidegger Citation1996; Earland-Bennett and Hawksworth Citation2005; Heuchert and Braun Citation2006; Zhurbenko et al. Citation2015). This research has included the description of new species and, to a lesser extent, discovering anamorph-teleomorph relationships (Ertz et al. Citation2013, Citation2014; Frisch et al. Citation2014; Muggia et al. 2015).
The diversity of lichenized and algicolous hyphomycetes has received considerably less attention. The degree to which these anamorphic fungi associate with algae is still argued to be at the edge of lichenization and is further blurred by the saprotrophic and parasitic lifestyles that can be observed in congeneric species.
Vobis and Hawksworth (Citation1981) reported about 41 genera of conidial fungi found to form stable association with algae from temperate and tropical regions. Three genera of lichenized, anamorphic coelomycetes, i.e., Lichingoldia, Woessia, and Hastifera (Hawksworth and Poelt Citation1986), were later found representing pycnidial stage of species currently placed in Bacidina and Micarea, respectively (Ekman Citation1996; Fryday Citation2001). Some other anamorphic lichenized species were originally described as lichenicolous fungi on sterile lichens, such as Reichlingia leopoldii (Diederich and Scheidegger Citation1996), which was later recognized as a sporodochiate lichen (e.g., Diederich and Coppins Citation2009). Conversely, other hyphomycetous species found in loose association with algae are now considered lichen parasites. For example, Nigropuncta rugulosa was originally described as a lichenized hyphomycete (Hawksworth and Poelt Citation1986) but subsequently considered to be a lichenicolous fungus specialized on Bellemerea cinereorufescens, on which its infection strongly suppresses the formation of the host apothecia (Hafellner Citation2012). Alternatively, species believed to be saprotrophic on wood are now recognized to be lichenized, such as Dictyocatenulata alba (Diederich et al. Citation2008). At the time of its original description (Morris and Finley Citation1967), D. alba was poorly understood due to the unclear illustrations and was generally considered to be a bark-inhabiting hyphomycete (Seifert et al. Citation1987). However, more recently it has been recognized to be a lichen-forming species (Lendemer and Harris Citation2004).
The nutritional status of some sporodochiate fungi is not easily interpreted, and nutritional modes of various taxa are interpreted differently depending on the author, who may categorized them as either a lichenicolous species or as lichen-forming fungi. Sclerococcum griseisporodochium was described as a lichenicolous, parasymbiotic fungus associated with a calcicolous species of Opegrapha, but optional lichenization of this taxon was recognized (Etayo Citation1995). For this reason, S. griseisporodochium is sometimes treated as a lichen (e.g., Smith Citation2009; Ertz et al. Citation2013), but it is more commonly listed among the lichenicolous fungi in fungal surveys. Similarly, Milospium graphideorum is reported as an example of a facultative, lichenized sporodochiate fungus (Diederich and Coppins Citation2009; Diederich Citation2013); however, it is usually recorded as a lichenicolous fungus of several epiphytic, crustose lichens associated with trentepohlioid photobionts.
Further, anamorph-teleomorph connections have been described only for few genera to date, and the majority of lichenized hyphomycetes are still known as anamorphic holomorphs. Both Blarneya hibernica (Hawksworth et al. Citation1979) and the type species of the genus Sporodochiolichen, S. lecanorinus (Aptroot and Sipman Citation2011), have been recognized as one anamorph of Tylophoron species (Ertz et al. Citation2013). Sporodochia of the taxon Cheiromycina ananas (Aptroot and Schiefelbein Citation2003) were reinterpreted as representing sessile synnemata corresponding to the widely distributed and polymorphic species Dictyocatenulata alba and therefore synonymized with it (Diederich et al. Citation2008). The generic concept of Reichlingia was further enlarged by Frisch et al. (Citation2014) incorporating three additional species with ascocarps and with concordant chemistry (perlatolic acid derivatives).
Lichenized hyphomycetes are still poorly represented in molecular phylogenies. However, molecular data have been generated for a limited number of genera. Based on these data, Tylophoron and Reichlingia have been placed in the order Arthoniales (Ertz et al. Citation2011; Ertz and Tehler Citation2011), and Dictyocatenulata alba was recovered within Ostropales but leaving the genus as incerta saedis within this order (An et al. Citation2012; Lücking et al. Citation2017). A single specimen of the sporodochiate lichen Chirleja buckii was sequenced, and the taxon was suggested to be a member of Icmadophilaceae (Pertusariales; Lendemer and Hodkinson Citation2012). Further DNA evidence even included Chirleja buckii in the genus Endocena, and the new combination of Endocena buckii was proposed by Fryday et al. (Citation2017). Recently, the description of Savoronala madagascariensis from arid regions in southeast Madagascar was coupled with phylogenetic data (Ertz et al. Citation2013). These latter supported the introduction of a new family, Malmideaceae, in which both S. madagascariensis and Malmidea were placed within Lecanorales (Ertz et al. Citation2013). Presently, additional molecular data are unavailable for other genera of lichenized hyphomycetes.
The genus Cheiromycina is unique because of its peculiar, cheiroid (finger/hand forming), multiseptate conidia, which are easily recognizable by microscopy. Cheiroid conidia are also described for other hyphomycetous taxa (Sutton Citation1985), such as the predominantly heterotrophic genus Psammina, including two lichenized species (Earland-Bennett and Hawksworth Citation2005; Cáceres and Aptroot Citation2016), and the nonlichenized genera Cheirospora (Hughes Citation1958), Cheiromyceopsis (Mercado Sierra and Mena Portales Citation1988), Ramoconidiifera (Sutton et al. Citation1996), Digitomyces (Mercado Sierra et al. Citation2003), and Cheirosporium (Cai et al. Citation2008). In contrast to Cheiromycina, Psammina palmata presents branched, nonseptate conidia (Earland-Bennett and Hawksworth Citation2005), whereas the recently described taxon P. tropica produces septate, only basally branched conidia, and further differentiates by associating with a trentepohlioid photobiont (Cáceres and Aptroot Citation2016). The other nonlichenized genera are melanized fungi within the class Dothideomycetes (Cai et al. Citation2008).
Cheiromycina is characterized by a thallus nearly immersed in bark or wood with hemispherical, whitish to pale gray or brownish, powdery sporodochia. The conidia are multicellular, hyaline, rarely pale brownish, smooth, flabelliform to palmate, usually consisting of a basal, subglobose (strongly inflated) conidiogenous cell from which distoseptate dichotomous branches derive. Ecologically, the genus seems to be confined to acidic substrates in humid forests of the boreal zone. The genus was described with Cheiromycina flabelliformis B. Sutton as the type species based on material from Picea wood from northern Sweden (Sutton and Muhr Citation1986). It was initially described as a hyphomycete with eustromatic sporodochial conidiomata, resembling the genus Cheiromyces due to the presence of distoseptate conidia. A stable association with green algae was confirmed by Hawksworth and Poelt (Citation1986) in the holotype and in further specimens reported from Austria. Later the same authors described an additional species from Austria, Cheiromycina petri (Hawksworth and Poelt Citation1990). According to their description, C. petri differs from C. flabelliformis by the melanized (noneroding) sporodochia, the branching pattern, and the size of the conidia. Printzen (Citation2007), studying further the type material, emphasized that an indistinctly enlarged conidiogenous cell is the main diagnostic character for C. petri. In the previous decade, three other species of Cheiromycina have been described: C. globosa from Germany (Aptroot and Schiefelbein Citation2003), C. ananas from USA (Aptroot and Schiefelbein Citation2003), later synonymized under Dictyocatenulata alba (Diederich et al. Citation2008), and C. reimeri from Turkey and Russian Far East (Printzen Citation2007). However, the most rarely recorded taxon, C. globosa, does not fit the description of the genus and most likely does not belong to Cheiromycina s. str. Cheiromycina globosa forms single-celled, globose conidia and differs from other congeners ecologically, being found in nitrophilous lichen communities.
The phylogenetic placement of the genus Cheiromycina has not investigated to date, although preliminary sequencing results showed similarity with multiple taxa in Lecanoromycetes. Therefore, in this study, we aimed to infer the phylogenetic placement of Cheiromycina within lichen-forming fungi using nuclear and mitochondrial ribosomal genetic markers. Below we discuss the relationship of Cheiromycina within Lecanoromycetes and with other lichenized hyphomycetous taxa.
MATERIALS AND METHODS
Sampling and morphological analyses
Fresh samples of Cheiromycina were collected in different localities in six countries including two U.S. states (Czech Republic, Norway, Ukraine, Russia, Poland, and USA [Alaska and Washington]; ). Five specimens representing C. flabelliformis, seven of C. petri, and two of C. reimeri were included (). Lichen specimens were sorted under a dissecting microscope, and species identification was performed by analyzing anatomical and squash sections of the sporodochia. Identifications followed Sutton and Muhr (Citation1986), Hawksworth and Poelt (Citation1990), and Printzen (Citation2007). Sections were prepared in water and examined with light microscopy. Digital images of growth habit and anatomical section ( and ) were acquired with a ZeissAxioCam MRc5 digital camera (Goettingen, Germany) fitted to the stereo- and light microscopes and were digitally optimized using the image processing software Combine ZM (www.hadleyweb.pwp.blueyonder.co.uk/CZM/).
Table 1. Information about Cheiromycina samples considered in this study.
Figure 1. Habit of Cheiromycina spp. thalli; samples are reported with their collector and DNA extraction numbers. A, C, D, F. C. flabelliformis: A. Z. Palice 21313, L2347; C. Z. Palice 18257, L2253; D. F. Jonsson LK46, L2325; F. Z. Palice 21313, L2344. B, E, G, H. C. petri: B. Z. Palice 17855, L2222; E. Z. Palice 19030, L2327; G, H. T. Tønsberg 42848, L2223 (G is a detail of H). Arrows point to the sporodochia. Bars: A–F = 0.5 mm; H = 1 mm.

Figure 2. Conidia and sporodochia sections of Cheiromycina spp.; samples are reported with their collector and DNA extraction numbers. C, E, F, J, K. Conidia of C. flabelliformis in various developmental stages: C, E, F. Z. Palice 18257, L2253. A, B, D, G. Pale brown conidia of C. petri: A, B. Z. Palice 17855, L2222; D. Z. Palice 19030, L2327; G. T. Tønsberg 43060, L2224. H, I, L. Squash preparation of vertical section of sporodochia: H. C. flabelliformis, Z. Palice 18257, L2253; L. C. flabelliformis, Z. Palice 21313, L2344; and I. C. petri, Z. Palice 19030, L2327. Arrows point to the algal cells scattered among the hyphae in the basal part of the conidioma. Bars: A–E = 10 μm; F–K = 20 μm; L= 50 μm.
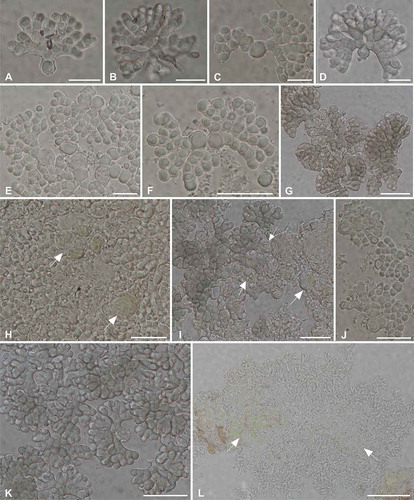
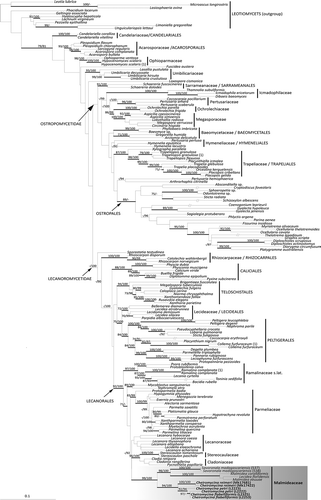
Figure 3. Multigene phylogenetic hypothesis of Cheiromycina spp. inferred from the combined data set of nucLSU, nucSSU, and mtSSU markers. Bayesian PP >95% and ML bootstrap support values >70% are reported above branches. Clades are named according to Miadlikowska et al. (Citation2014) and represent a broad selection of taxa within the Lecanoromycetes. The newly sequenced samples (as in ) are reported in bold.
DNA extraction, amplification, and sequencing
All samples were checked for contamination, and the sporodochia were carefully dissected under the stereomicroscope and taken for DNA extraction. The material was first frozen in liquid nitrogen and then pulverized with polypropylene pestles. The DNA was extracted according to the protocol of Cubero et al. (Citation1999). Molecular sequence data from Cheiromycina samples were generated for the nuclear large and partial nuclear small ribosomal subunits (28S and 18S, respectively) and for the mitochondrial small ribosomal subunit (12S). The nuclear 28S locus was amplified using primers LR3R and LR7 (Vilgalys and Hester Citation1990); the nuclear 18S region was amplified using primers NS1 (White et al. Citation1990) and nuSSU0852 (Gargas and Taylor Citation1992); and a fragment of the mitochondrial 12S locus was amplified with primers mtSSU1KL (Zoller et al. Citation1999) and MSU7 (Zhou and Stanosz Citation2001). Polymerase chain reaction (PCR) amplifications were carried out using Illustra Ready-To-Go GenomiPhi V3 DNA amplification kit (GE Healthcare Bio-Sciences, Pittsburgh, Pennsylvania, USA), and the temperature profile followed the “touch-down” PCR conditions reported in Muggia et al. (2015). Complementary strands were sequenced at Microsynth (Vienna, Austria). The sequences were manually assembled and edited in BioEdit (Hall Citation1999).
Phylogenetic analyses
The identity of the new generated sequences was checked with sequences available in the GenBank database using the BLAST search algorithm (Altschul et al. Citation1990); the taxa with the highest sequences similarity, as inferred from the BLAST search, were selected and included in subsequent phylogenetic analyses. In addition, an exploratory analysis was performed, including fungi from a wide phylogenetic spectrum (not shown), comprising the classes Dothideomycetes, Eurotiomycetes, Lecanoromycetes, Leotiomycets, and Sordariomycetes. As the National Center for Biotechnology Information (NCBI) BLAST results always output as closest matches representatives of Lecanoromycetes, and our first single-locus analyses also placed the sequences within Lecanoromycetes, we reduced the data set of the final phylogenetic analysis to Lecanoromycetes only and selected 10 species of Leotiomycetes as outgroups (TABLE S1). The selection of the outgroups and of the Lecanoromycetes ingroups was based on the recent phylogenetic analyses of Ertz et al. (Citation2013) and Miadlikowska et al. (Citation2014). For a number of specimens, we were unable to generate sequences for all the selected loci (18S, 28S, and mitochondrial 12S), and a complete set of all three sampled loci were not available in GenBank. The sequence alignments were prepared manually in BioEdit and individually for the three loci. Introns and ambiguous single-nucleotide polymorphisms (SNPs) were removed from the alignments. Alignments are deposited in TreeBASE under the reference number TB21301 (http://purl.org/phylo/treebase/phylows/study/TB2:S21301).
Combined data of different loci, whether fully or partially congruent, should be considered when inferring phylogenetic relationships (Dettman et al. Citation2003), and here we analyzed both single-locus alignments and a combined, three-marker data set. We analyzed the single-locus data sets within a maximum likelihood (ML) framework (Mason-Gamer and Kellogg Citation1996; Reeb et al. Citation2004) and the combined data set using both ML and Bayesian approaches. The combined data set was partitioned by individual locus, nuclear 28S and 18S and mitochondrial 16S, in both ML and Bayesian analyses. The program RAxML 7.0.4 (Stamatakis et al. Citation2005) was used for ML analyses and estimation of bootstrap support. As only a single model of molecular evolution can be used across the partitions, the ML analysis was performed with the GTRMIX model and 1000 bootstrap replicates were run. The model of molecular evolution applied to each gene partition in the Bayesian analysis, GTR+I+G, was estimated in jModeltest 2.1.4 (Darriba et al. Citation2012) using the Akaike information criterion (Posada and Crandall Citation1998). The Bayesian Markov chain Monte Carlo (B/MCMC) analyses were run in MrBayes 3.1.2 (Huelsenbeck and Ronquist 2003; Ronquist et al. Citation2005) with six chains simultaneously, each initiated with a random tree, for 10 million generations; trees were sampled every 100th generation for a total sample of 100 000 trees. Log-likelihood scores against generation time were plotted using Tracer 1.4 (Rambaut and Drummond Citation2007) to determine when the stationarity of likelihood values had been reached as a guide for where to set the burn-in stage (Ronquist et al. Citation2005). Burn-in was set at 3 million generations (the first 30 000 sampled trees), and a majority rule consensus tree was calculated from the posterior sample of 70 001 trees. The convergence of the chains was confirmed by the convergent diagnostic of the potential scale reduction factor (PSRF), which approached 1 (Ronquist et al. Citation2005). The phylogenetic trees were visualized in TreeView (Page Citation1996).
RESULTS
We obtained a total of four sequences for C. flabelliformis (one nuLSU, two nuSSU, and one mtSSU sequences), five for C. petri (one nuLSU, two nuSSU, and two mtSSU sequences), and four for C. reimeri (two nuSSU and two mtSSU sequences; ). We were unable to generate sequences for the selected loci for nine specimens. Six samples in total were included in the final phylogenetic analysis (). All the retrieved sequences found their closest matches with representatives of Lecanoromycetes.
Our phylogenetic inference () is topologically congruent with the previous analysis of Miadlikowska et al. (Citation2014). Although this topology was limited in terms of taxon sampling relative to previous studies, the relationships among families and orders within Lecanoromycetes in our phylogenetic inference were largely consistent with previous reconstructions and relationships were generally recovered with high support. All newly sequenced samples, both in the single-locus (not shown) and in the multilocus analyses, form a monophyletic, fully supported clade. This clade was recovered with high support as the sister group of the recently described, monophyletic family Malmideaceae (Ertz et al. Citation2013), which include “Lecidea” floridensis (just recently recombined into Malmidea floridensis in Cáceres et al. [2017]), Malmidea coralliformis, M. eeuuae, and the hyphomycetous species Savoronala madagascariensis.
DISCUSSION
This study places the hyphomycetous genus Cheiromycina in Lecanorales and within the family Malmideaceae (Kalb et al. Citation2011). Our phylogeny, inferred from a three-marker data set, recovered Cheiromycina as sister lineage to Savoronala, another lichenized hyphomycetous genus (Ertz et al. Citation2013). Apart from sharing the anamorphic state, the two genera occur in different habitats and significantly differ in sporodochia and conidia morphology, as reported in the original description of the two genera by Sutton and Muhr (Citation1986) and Ertz et al. (Citation2013), respectively. In Savoronala, sporodochia form at the apex of erected stipes that develop at the center of the thallus and are not branched. The sporodochia are convex, grayish blue in color, and their surface become uneven when covered by the conidial agglomerations (Ertz et al. Citation2013). In Cheiromycina, sporodochia are alternatively eustromatic, usually isolated, white to gray in color, dry, pulverulent, and consist of conidiophores, conidiogenous cells, and conidia intermixed (Sutton and Muhr Citation1986). Furthermore, thalli of Savoronala are well distinguishable, whereas those of Cheiromycina are hardly detectable on the substrate surface.
Interestingly, both these hyphomycetous genera are included in a family of lichen-forming fungi for which multiple forms of dispersion are known. In fact, only recently the genera Crustospathula, Kalbionora, and Sprucidea have been recognized to be part of Malmideaceae (Cáceres et al. Citation2017; Sodamuk et al. Citation2017), in addition to the originally included species of Malmidea, Savonorala, and two taxa of Lecidea s. lat. (Kalb et al. Citation2011; Ertz et al. Citation2013). In light of these recent studies, the circumscription of Malmideaceae is in need of a more comprehensive assessment, both in terms of taxon sampling and molecular phylogenetic studies. Future molecular studies in this group of lichen-forming fungi should also consider including the genus Xyleborus R.C. Harris & Ladd (Harris and Ladd Citation2007; Lendemer and Harris Citation2015) as a potential member of the family Malmideaceae. This unique epixylic genus confined to the Appalachian region of eastern North America is usually fertile, forming dark lecideoid apothecia but simultaneously often produces delimited pale sporodochia with superficial resemblance to the sporodochia of Cheiromycina. Harris and Ladd (Citation2007) attributed Xyleborus to Stereocaulaceae, which was accepted also in the new classification of Ascomycetes (Lücking et al. Citation2017). However, this assignment should be understood as tentative due to the lack of molecular data. The potential placement of Xyleborus in Malmideaceae would further corroborate the similarity observed with “Lecidea” plebeja (nowadays known to be a member of Malmideaceae; Ertz et al. Citation2013; Cáceres et al. Citation2017) and the presence of sporodochia as known for many members of the family.
Sequence data available for Dictyocatenulata alba (An et al. Citation2012) were purposely not included here for multiple reasons, although Cheiromycina ananas was synonymized with D. alba (Diederich et al. Citation2008). First, D. alba does not produce cheiroid conidia and, together with C. globosa, have a conidiogenesis very distinct from that of C. flabelliformis and C. petri. Dictyocatenulata alba also produces variably tall, long-stiped to sessile synnemata that were misinterpreted to be sporodochia due to the extremely low number of samples bearing sporodochia-like synnemata (which were compared at the time of its description). D. alba is a subcosmopolitan lichen reaching also the subtropics and definitely cannot be considered closely related to Cheiromycina s. str. Second, our new sequences matched neither with any of the D. alba in the GenBank BLAST searches nor with any representatives of Ostropomycetes, the class in which D. alba was placed with incertae sedis (An et al. Citation2012).
Interestingly, An et al. (Citation2012) reported on the relatively fast and successful growth of D. alba in culture, but our multiple attempts to isolate Cheiromycina spp. failed and the fungi grown in cultures were identified as Sordariomycetes. It is well known that lecanoralean lichen mycobionts grow rather slowly in axenic cultures, whereas Ostropalean fungi, which include also several nonlichenized and optionally lichenized taxa (Wedin et al. Citation2004, Citation2006), seem to be easier to culture (Muggia et al. Citation2011, Citation2016). This anecdotal observation further supports the likely different phylogenetic placement for the two fungi.
A green, coccoid alga has been observed to be the photobiont in Cheiromycina species (Hawksworth and Poelt Citation1990). However, the identity Cheiromycina-associated photobionts has not been studied yet. In this case, all attempts to amplify and sequence and/or culture the algae failed, likely due to the insufficient amount of photobiont cells in our preparations. In fact, algae were found localized only at the base of the sporodochia (, I, L) and were never observed to be tightly associated with the conidiogenous hyphae or conidia. Co-dispersion of the fungus with the algae might not happen in Cheiromycina, although this strategy has been observed for the sister genus Savoronala, where the conidia wrap around a single algal cell already at the early stage of the diaspore formation and before the fungal cells become melanized (Ertz et al. Citation2013).
In general, hyphomycetous lichenicolous and lichenized fungi are less frequently collected than other fungal taxa that develop conspicuous thalli and sexual reproductive structures (apothecia or perithecia). The often subtle morphological characters create unique challenges in effectively circumscribing species boundaries using morphological data alone. Further confounding our understanding of diversity in this enigmatic group is the fact that most specimens of anamorphic fungi usually hold too little material to allow multiple analyses. For example, in the genus Cheiromycina, the published descriptions of the three species—C. ananas, C. globosa, and C. petri—were based on single collections, which definitely have not encompassed the range of intraspecific morphological and anatomical variability that a species comprises (Muggia et al. Citation2014). External morphological characters are also of little help when trying to distinguish Cheiromycina species, as the habit of the sporodochia is rather variable and “not eroding” or “eroding white” sporodochia (Hawksworth and Poelt Citation1986) are recovered in both species (see also Printzen Citation2007). More efficient diagnostic, key characters were suggested to be represented by the conidiogenous cells, size, and branching patterns of the conidia (Printzen Citation2007). Cheiromycina flabelliformis, C. petri, and C. reimeri are distinguished by their multicellular, palmately branched conidia and have been segregated from each other based on differences in the size of conidiogenous cells and shape and septation of conidial branches. In addition, conidia in C. flabelliformis have been reported as displaying a secondary three-dimensional structure. We observed, however, that conidia can be three-dimensional also in specimens otherwise referring to C. petri (, ). The enlarged conidiogenous cells are easily recognized in the majority of the conidia in C. flabelliformis, but squash preparation may destroy or break them from the rest of the conidium, thus biasing the species identification. In light of our results, we propose that the genus Cheiromycina is in need of taxonomic revision. The current circumscription of the genus may in fact not reflect the phylogenetic position of the other, here not sequenced taxon C. globosa, which significantly differs by the lack of cheiroid conidia. However, a taxonomic treatment of the genus is beyond the scope of the present study.
Cheiromycina was described about 30 years ago as “an unusual deuteromycete with the appearance of a sorediate endoxylic lichen” (Sutton and Muhr Citation1986, p. 831), and no teleomorphic state is known for it so far. In this study, we could detect tiny, pale brown apothecia on the thallus bearing the sporodochia in a single specimen of C. petri (L2224, Tønsberg 43060) (SUPPLEMENTARY FIG. 1), although the apothecia contained immature asci without ascospores. Unfortunately, PCR amplifications from the apothecia and those from the sporodochia preparations were unsuccessful for this sample; thus, comparison with the other Cheiromycina samples was impossible. Recovering Cheiromycina and other lichenized hyphomycetous genera within Lecanorales further encourages future investigations of additional anamorphic lichenized fungi for which both no sequence data are available so far and no teleomorphic state is known (Ertz et al. Citation2011, Citation2013; Diederich et al. Citation2013).
In lichen-forming fungi, sexual reproduction is by far the most dominant reproductive mode (Seymour et al. Citation2005). However, some species-rich genera are exceptionally represented by only asexual species, such as Lepraria, which have also evolved strikingly diverse array of secondary metabolites (Ekman and Tønsberg Citation2002; Elix and Tønsberg Citation2004; Kukwa Citation2006; Nelsen and Gargas Citation2008; Flakus et al. Citation2011; Lendemer Citation2013). Whether speciation in Lepraria has occurred in the absence of recombination is still a debated question (Fehrer et al. Citation2008). Although parasexual processes may play a crucial role in lineages of lichen-forming fungi, it is very difficult to demonstrate and it might be speculated that they act in the successful distribution of the species and their genetic diversity. Due to the limited number of Cheiromycina samples available, obtaining detailed, robust insight into intraspecific diversity of these lichenized hyphomycetes will be challenging into the foreseeable future.
Suppl_Material_Table_S1_Cheiromycina.pdf
Download PDF (73.3 KB)ACKNOWLEDGMENTS
L.M. and T.T. are grateful to Bruce McCune (Oregon State University) for the invitation to do lichenological field work in Katmai National Park in 2013. Amy Miller and James Walton (both National Park Service) are thanked for project coordination and for organizing and executing field logistics. L.M. thanks Claudio G. Ametrano and Theodora Kopun for their support in the laboratory work, and Martin Grube and Josef Hafellner for constructive discussions. M.K. thanks Anna Łubek (Jan Kochanowski University in Kielce) for organizing field studies and great help during collecting. Timothy Wheeler and Steven Leavitt are thanked for their support in revising the English text.
SUPPLEMENTAL DATA
Supplemental data for this article can be accessed on the publisher’s Web site.
FUNDING
L.M. was financially supported by the Austrian Science Fund (FWF project P24114-B16). M.K. has received financial support by the Polish-Norwegian Research Programme operated by the National Centre for Research and Development under the Norwegian Financial Mechanism 2009–2014 in the frame of project contract no. Pol-Nor/196829/87/2013. L.M. and T.T. are grateful to the National Park Service (NPS) and to the Southwest Alaska Network Anchorage for funding. Z.P. acknowledges the development project no. RVO67985939 from the Czech Academy of Sciences and institutional resources of the Ministry of Education, Youth and Sports of the Czech Republic for the support of science and research.
Additional information
Funding
LITERATURE CITED
- Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. 1990. Basic local alignment search tool. The Journal of Molecular Biology 215:403–410.
- An K-D, Degawa Y, Fujihara E, Mikawa T, Ohkuma M, Okada G. 2012. Molecular phylogenetic analyses based on the nuclear rRNA genes and the intron-exon structures of the nuSSU rRNA gene in Dictyocatenulata alba (anamorphic Ascomycota). Fungal Biology 116:1134–1145.
- Aptroot A, Schiefelbein U. 2003. Additional species of Cheiromycina (lichenized hyphomycetes), with a key to the known species. Mycological Research 107:104–107.
- Aptroot A, Sipman HJM. 2011. Sporodochiolichen a new genus of tropical hyphomicetous lichens. The Lichenologist 43:357–362.
- Berbee ML, Taylor JW. 1993. Ascomycete relationships: dating the origin of asexual lineages with 18S ribosomal RNA gene sequence data. In: Reynolds DR, Taylor JW, eds. The fungal holomorph: mitotic, meiotic, and pleomorphic speciation in fungal systematics. Wallingford, UK: CAB International. p. 68–77.
- Cáceres MES, Aptroot A. 2016. First inventory of lichens from the Brazilian Amazon in Amapá State. The Bryologist 119:250–265.
- Cáceres MES, Aptroot A, Oliveira Mendoca C, dos Santos LA, Lücking R. 2017. Sprucidea, a further new genus of rain forest lichens in the family Malmideaceae (Ascomycota). The Bryologist 120:202–211.
- Cai L, Guo XY, Hyde KD. 2008. Morphological and molecular characterization of a new anamorpjhic genus Cheirosporium, from freshwater in China. Persoonia 20:53–58.
- Crous PW, Groenewald JZ, Mansilla JP, Hunter GC, Wingfield MJ. 2004. Phylogenetic reassessment of Mycosphaerella spp. and their anamorphs occurring on Eucalyptus. Studies in Mycology 50:195–214.
- Crous PW, Kang JC, Braun U. 2001. A phylogenetic redefinition of anamorph genera in Mycosphaerella based on ITS rDNA sequence and morphology. Mycologia 93:1081–101.
- Crous PW, Wingfield MJ, Mansilla JP, Alfenas AC, Groenewald JZ. 2006. Phylogenetic reassessment of Mycosphaerella spp. and their anamorphs occurring on Eucalyptus. II. Studies in Mycology 55:99–131.
- Cubero OF, Crespo A, Fatehi J, Bridge PD. 1999. DNA extraction and PCR amplification method suitable for fresh, herbarium stored and lichenized fungi. Plant Systematics and Evolution 217:243–249.
- Darriba D, Taboada GL, Doallo R, Posada D. 2012. jModelTest 2: more models, new heuristics and parallel computing. Natural Methods 9: article 772.
- Dettman JR, Jacobson DJ, Taylor JW. 2003. A multilocus genealogical approach to phylogenetic species recognition in the model eukaryote Neurospora. Evolution 57:2703–2720.
- Diederich P. 2011. Lichenicolous hyphomycete genera. In: Seifert K, Morgan-Jones G, Gams W, Kendrick B, eds. The genera of Hyphomycetes. CBS Biodiversity Series 9. Utrecht, The Netherlands: CBS-KNAW Fungal Biodiversity Center. p. 917–920.
- Diederich P. 2013. A redescription of Sporodochiolichen flavus (lichenized sporodochial Ascomycetes). Bulletin de la Société des Naturalistes Luxembourgeois 114:55–58.
- Diederich P, Coppins BJ. 2009. Reichlingia Diederich & Scheid. (1996). In: Smith CW, Aptroot A Coppins BJ, Fletcher A, Gilbert OL, James PW, Wolseley PA, eds. The lichens of Great Britain and Ireland. London: British Lichen Society. p. 790–791.
- Diederich P, Ertz D, Lawrey JD, Sikaroodi M, Untereiner WA. 2013. Molecular data place the hyphomycetous lichenicolous genus Sclerococcum close to Dactylospora (Eurotiomycetes) and S. parmeliae in Cladophialophora (Chaetothyriales). Fungal Diversity 58:61–72.
- Diederich P, Palice Z, Ertz D. 2008. Cheiromycina ananas is a synonym of Dictyocatenulata alba, a widespread, lichenized, synnematous hyphomycete herewith reported as new for Europe. Sauteria 15:205–214.
- Diederich P, Scheidegger C. 1996. Reichlingia leopoldii gen. et sp. nov., a new lichenicolous hyphomycete from Central Europe. Bulletin de la Société des Naturalistes Luxembourgeois 97:3–8.
- Earland-Bennett PM, Hawksworth DL. 2005. The first lichen-forming species of Psammina, P. palmata sp. nov., with notes on the status of Cheiromycina and Pycnopsammina. The Lichenologist 37:191–197.
- Ekman S. 1996. The corticolous and lignicolous species of Bacidia and Bacidina in North America. Opera Botanica 127:1–148.
- Ekman S, Tønsberg T. 2002. Most species of Lepraria and Leproloma form a monophyletic group closely related to Stereocaulon. Mycological Research 106:1262–1276.
- Elix JA, Tønsberg T. 2004. Notes on the chemistry of some lichens, including four species of Lepraria. Graphis Scripta 16:43–45.
- Ertz D, Bungartz F, Diederich P, Tibell L. 2011. Molecular and morphological data place Blarneya in Tylophoron (Arthoniaceae). The Lichenologist 43:345–356.
- Ertz D, Fischer E, Killmann D, Razafindrahaja T, Sérusiaux E. 2013. Savoronala, a new genus of Malmideaceae (Lecanorales) from Madagascar with stipes producing sporodochia. Mycological Progress 12:645–656.
- Ertz D, Lawrey JD, Common RS, Diederich P. 2014. Molecular data resolve a new order of Arthoniomycetes sister to the primarily lichenized Arthoniales and composed of black yeasts, lichenicolous and rock-inhabiting species. Fungal Diversity 66:113–137.
- Ertz D, Tehler A. 2011. The phylogeny of Arthoniales (Pezizomycotina) inferred from nucLSU and RPB2 sequences. Fungal Diversity 49:47–71.
- Etayo J. 1995. Two new species of lichenicolous fungi from the Pyrenees. Nova Hedwigia 61:189–197.
- Fehrer J, Bayerová Š, Orange A. 2008. Large genetic divergence of new, morphologically similar species of sterile lichens from Europe (Lepraria, Stereocaulaceae, Ascomycota): concordance of DNA sequence data with secondary metabolites. Cladistics 24:443–458.
- Flakus A, Elix JA, Rodriguez P, Kukwa M. 2011. New species and records of Lepraria (Stereocaulaceae, lichenized Ascomycota) from South America. The Lichenologist 43:57–66.
- Frisch A, Thor G, Sheil D. 2014. Four new Arthoniomycetes from Bwindi Impenetrable National Park, Uganda—supported by molecular data. Nova Hedwigia 98:295–312.
- Fryday AM. 2001. The lichen vegetation associated with areas of late snow-lie in the Scottish Highlands. The Lichenologist 33:121–150.
- Fryday AM, Schmitt I, Perez-Ortega S. 2017. The genus Endocena (Icmadophilaceae): DNA evidence suggests the same fungus forms different morphologies. The Lichenologist 49:347–363.
- Gargas A, Taylor JW. 1992. Polymerase chain reaction (PCR) primers for amplifying, sequencing nuclear 18S rDNA from lichenized fungi. Mycologia 84:589–592.
- Hafellner J. 2012. Lichenicolous Biota (Nos 101–120). Fritschiana 74:1–17.
- Hall TA. 1999. BioEdit: a user friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acid Symposia Series 41:95–98.
- Harris RC, Ladd D. 2007. New taxa of lichens and lichenicolous fungi from the Ozark Ecoregion. Opuscula Philolichenum 4:57–68.
- Hawksworth DL. 1979. The lichenicolous hyphomycetes. Bulletin of the British Museum (Natural History), Botany 6:183–300.
- Hawksworth DL, Coppins BJ, James PW. 1979. Blarneya, a lichenized hyphomycete from southern Ireland. The Botanical Journal of the Linnean Society 79:357–367.
- Hawksworth DL, Poelt J. 1986. Five additional genera of conidial lichen-forming fungi from Europe. Plant Systematics and Evolution 154:195–211.
- Hawksworth DL, Poelt J. 1990. A second lichen-forming species of Cheiromycina from Austria. The Lichenologist 22:219–224.
- Heuchert B, Braun U. 2006. On some dematiaceous lichenicolous hyphomycetes. Herzogia 19:11–21.Huelsenbeck JP, Ronquist F. 2003. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572e1574.
- Hughes SJ. 1953. Conidiophores, conidia and classification. Canadian Journal of Botany 31:577–657.
- Hughes SJ. 1958. Revisiones Hyphomycetum aliquot cum appendice de nominibus rejiciendis. Canadian Journal of Botany 36:727–836.
- Huhndorf SM, Fernández FA. 2005. Teleomorph-anamorph connections: Chaetosphaeria raciborskii and related species, and their Craspedodidymum-like anamorphs. Fungal Diversity 19:23–49.
- Kalb K, Rivas Plata E, Lücking R, Lumbsch HT. 2011. The phylogenetic position of Malmidea, a new genus for the Lecidea piperis- and Lecanora granifera-groups (Lecanorales, Malmideaceae), inferred from nuclear and mitochondrial ribosomal DNA sequences, with special reference to Thai species. Bibliotheca Lichenologica 106:143–168
- Kukwa M. 2006. The lichen genus Lepraria in Poland. The Lichenologist 38:293–305.
- Lendemer JC. 2013. A monograph of the crustose members of the genus Lepraria Ach. s.str. (Stereocaulaceae, Lichenized Ascomycetes) in North America north of Mexico. Opuscula Philolichenum 11:27–141.
- Lendemer JC, Harris RC. 2004. A checklist of the lichens collected on the 28th A. Leroy Andrews Foray. Evansia 21:88–100.
- Lendemer JC, Harris RC. 2015. Xyleborus nigricans, a second species for the previously monospecific genus newly found in the Mid-Atlantic Coastal Plain of North America. Bryologist 118:284–292.
- Lendemer JC, Hodkinson BP. 2012. Chirleja buckii, a new genus and species of lichenized-fungi from Tierra del Fuego, southern South America. New Zealand Journal of Botany 50:449–456.
- Liu ZY, Yao YJ, Liang ZQ, Liu AY, Pegler DN, Chase MW. 2001. Molecular evidence for the anamorph—teleomorph connection in Cordyceps sinensis. Mycological Research 105:827–832.
- Lizel M, Crous PW, Groenewald JZE, Gams W, Summerbell C. 2003. Togninia (Calosphaeriales) is confirmed as teleomorph of Phaeoacremonium by means of morphology, sexual compatibility and DNA phylogeny. Mycologia 95:646–659.
- Lücking R, Hodkinson BP, Leavitt SD. 2017. The 2016 classification of lichenized fungi in the Ascomycota and Basidiomycota—approaching one thousand genera. The Bryologist 119:361–416.
- Mason-Gamer RJ, Kellogg EA. 1996. Testing for phylogenetic conflict among molecular data sets in the tribe Triticeae (Gramineae). Systematic Biology 45:524–545.
- Mercado Sierra A, Calduch M, Gené J, Guarro J, Delgado G. 2003. Digitomyces, a new genus of hyphomycetes with cheiroid conidia. Mycologia 95:860–864.
- Mercado Sierra A, Mena Portales J. 1988. Nuevos o raros hifomicetes de Cuba. B. Un nuevo género sobre Roystonea regia. Acta Botanica Cubana 53:1–5.
- Miadlikowska J, Kauff F, Högnabba F, Oliver JC, Molnár K, Fraker E, Otálora MA. 2014. A multigene phylogenetic synthesis for the class Lecanoromycetes (Ascomycota): 1307 fungi representing 1139 infrageneric taxa, 317 genera and 66 families. Molecular Phylogenetics and Evolution 79:132–168.
- Morris EF, Finley DE. 1967. Two new genera of stilbellaceous fungi. The American Midland Naturalist 77:200–204.
- Muggia L, Baloch E, Stabentheiner E, Grube M, Wedin M. 2011. Photobiont association and genetic diversity of the optionally lichenized fungus Schizoxylon albescens. FEMS Microbiology Ecology 75:255–272.
- Muggia L, Fernandez Brime S, Grube M, Wedin M. 2016. Schizoxylon as an experimental model for studying interkingdom symbiosis. FEMS Microbiology Ecology 92:fiw165.
- Muggia L, Pérez-Ortega S, Fryday A, Spribille T, Grube M. 2014. Global assessment of genetic variation and phenotypic plasticity in the lichen-forming species Tephromela atra. Fungal Diversity 64:233–251.
- Nelsen MP, Gargas A. 2008. Phylogenetic distribution and evolution of secondary metabolites in the lichenized fungal genus Lepraria (Lecanorales: Stereocaulaceae). Nova Hedwigia 86:115–131.
- Page RDM. 1996. TREEVIEW: an application to display phylogenetic trees on personal computers. Computational Application in Bioscience 12:357–358.
- Pérez-Ortega S, Suija A, de Los Rios A. 2011. The connection between Abrothallus and its anamorph state Vouauxiomyces established by Denaturing Gradient Gel Electrophoresis (DGGE). The Lichenologist 43:277–279.
- Posada D, Crandall KA. 1998. Modeltest—testing the model of DNA substitution. Bioinformatics 14:817–818.
- Printzen C. 2007. New records of Cheiromycina species, a genus of lichenized hyphomycetes, with C. reimeri sp. nov. and a revised key to the species. Nova Hedwigia 84:261–267.
- Rambaut A, Drummond A. 2007. Tracer. [cited 2007 Oct 11]. Available from: beast.bio.ed.ac.uk/Tracer
- Réblová M, Seifert KA. 2004. Conioscyphascus, a new ascomycetous genus for holomorphs with Conioscypha anamorphs. Studies in Mycology 50:95–108.
- Réblová M, Seifert KA. 2011. Discovery of the teleomorph of the hyphomycete, Sterigmatobotrys macrocarpa, and epitypification of the genus to holomorphic status. Studies in Mycology 68:193–202.
- Reeb V, Lutzoni F, Roux C. 2004. Contribution of RPB2 to multilocus phylogenetic studies of the euascomycetes (Pezizomycotina, Fungi) with special emphasis on the lichen-forming Acarosporaceae and evolution of polyspory. Molecular Phylogenetics and Evolution 32:1036–1060.
- Reynolds DR, Taylor JW. 1993. The fungal holomorph: mitotic, meiotic and pleomorphic speciation in fungal systematics. Oxon, UK: CAB International.
- Ronquist F, Huelsenbeck JP, Van der Mark P. 2005. MrBayes 3.1 manual. [cited 2005 May 26]. Available from: http://mrbayes.csit.fsu.edu/mb3.1_manual.pdf
- Saccardo PA. 1886. Sylloge fungorum 4. Patavia (Passau): Saccardo.
- Seifert KA, Kendrick B, Morgan-Jones G, Gams W. 2011. The genera of Hyphomycetes. Utrecht, The Netherlands: CBS-KNAW Fungal Biodiversity Centre. 997 p.
- Seifert KA, Okada G, Rao V. 1987. The synnematous hyphomycete Dictyocatenulata alba. Mycologia 79:459–462.
- Seymour F, Crittenden P, Dyer P. 2005. Sex in the extremes: lichen-forming fungi. Mycologist 19:51–58.
- Shenoy BD, Jeewon R, Hyde KD. 2007. Impact of DNA sequence-data on the taxonomy of anamorphic fungi. Fungal Diversity 26:1–54.
- Smith CW. 2009. Sclerococcum Fr. (1825). In: Smith CW, Aptroot A, Coppins BJ, Fletcher A, Gilbert OL, James PW, Wolseley PA, eds. The lichens of Great Britain and Ireland. London: British Lichen Society. p. 837.
- Sodamuk M, Boonpragob K, Mongkolsuk P, Tehler A, Leavitt SD, Lumbsch HT. 2017. Kalbionora paleotropica, a new genus and species from coastal forest in South Asia and Australia (Malmideaceae, Ascomycota). MycoKeys 22:15–25.
- Stamatakis A, Ludwig T, Meier H. 2005. RAxML-iii: a fast program for maximum likelihood-based inference of large phylogenetic trees. Bioinformatics 21:456–463.
- Sutton BC. 1985. Notes on some deuteromycete genera with cheiroid or digitate brown conidia. Proceeding of the Indian Academy of Science (Plant Science) 94:229–244.
- Sutton BC, Carmaran CC, Romero AI. 1996. Ramoconidiifera, a new genus of hyphomycetes with cheiroid conidia from Argentina. Mycological Research 100:1337–1340.
- Sutton BC, Muhr L-E. 1986. Cheiromycina flabelliformis gen. et sp. nov. on Picea from Sweden. Nordic Journal of Botany 6:831–836.
- Vilgalys R, Hester M. 1990. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. Journal of Bacteriology 172:4238–4246.
- Vobis G, Hawksworth DL. 1981. Conidial lichen-forming fungi. In: Cole GT, Kandrick WB, eds. The biology of conidial fungi 1. New York: Academic Press. p. 245–273.
- Wedin M, Döring H, Gilenstam G. 2004. Saprotrophy and lichenization as options for the same fungal species on different substrata: environmental plasticity and fungal lifestyles in the Stictis-Conotrema complex. New Phytologist 164:459–465.
- Wedin M, Döring H, Gilenstam G. 2006. Stictis s. lat. (Ostropales, Ascomycotina) in northern Scandinavia, with a key and notes on morphological variation in relation to lifestyle. Mycological Research 110:773–789.
- White TJ, Burns TD, Lee S, Taylor J. 1990. Amplification and direct sequencing of fungal ribosomal DNA genes for phylogenies. In: Innis MA, Gelfand DH, Snisky JJ, White TJ, eds. PCR protocols, a guide to methods and applications. San Diego, California: Academic Press. p. 315–322.
- Zhou S, Stanosz GR. 2001. Primers for amplification of mtSSU rDNA, and a phylogenetic study of Botryosphaeria and associated anamorphic fungi. Mycological Research 105:1033–1044.
- Zhurbenko MP, Braun U, Heuchert B, Kobzeva AA. 2015. New lichenicolous hyphomycetes from Eurasia. Herzogia 28:584–598.
- Zoller S, Scheidegger C, Sperisen C. 1999. PCR primers for the amplification of mitochondrial small subunit ribosomal DNA of lichen-forming ascomycetes. The Lichenologist 31:511–516.
