ABSTRACT
Process philosophy offers a metaphysical foundation for domestication studies. This grounding is especially important given the European colonialist origin of ‘domestication’ as a term and 19th century cultural project. We explore the potential of process archaeology for deep-time investigation of domestication relationships, drawing attention to the variable pace of domestication as an ongoing process within and across taxa; the nature of domestication ‘syndromes’ and ‘pathways’ as general hypotheses about process; the importance of cooperation as well as competition among humans and other organisms; the significance of non-human agency; and the ubiquity of hybrid communities that resist the simple wild/domestic dichotomy.
Introduction
Studies of ‘domestication’ in archaeology and beyond continue to harbour notions of human mastery that reflect the European colonialist origins of the term. Accounts of farming origins routinely posit a ‘step change’ in human cultural development because domestication is something humans ‘did’ to plant and animal species. The causality of domestication begins exclusively with human interests or human niches. As a verb, ‘to domesticate’ is transitive, with a subject (humans, the domesticator) and an object (domesticates) (cf. Stépanoff and Vigne Citation2018).
It is in some ways remarkable that this understanding of domestication persists. Darwin himself drew attention to the importance of ‘unconscious selection’ in domestic relationships (Darwin Citation1868) and his later work on earthworms (Darwin Citation1881) foreshadowed the growing 20th century recognition that organisms continually (re-)construct their own environmental niches. From the beginnings of evolutionary biology, therefore, it was implicit that human relationships with other species, including those bracketed off as ‘domestication’, fitted into a broader web of evolutionary interactions. Archaeologists have followed biologists (Lewontin Citation1978, Citation1983) by demonstrating wide empirical support for co-evolutionary relationships among humans and other organisms, and for mutualistic symbioses and niche construction activities that include farming and herding (Rindos et al. Citation1980; Rindos Citation1984; Smith Citation2007; Rowley-Conwy and Layton Citation2011). Zeder (Citation2016) has built a detailed empirical case to argue that early domestication relationships among humans, plants and animals offer a model case study in niche construction. Archaeological studies of domestication are thus relevant to wider acceptance of the ‘extended evolutionary synthesis’ (EES), in which niche construction theory (NCT) plays a central role (Zeder Citation2017).
From a philosophical standpoint, it is less surprising that human-centred notions of domestication – a sort of ‘one-sided’ niche construction – continue to persist. In both biology and archaeology, the acceptance of NCT occurred on the basis of empirical evidence, without requiring philosophical or metaphysical reflection. This is where process philosophy can make a useful contribution to the study of domestication, and to unlearning its tacit assumptions.
Early 20th century process philosophers such as Alfred North Whitehead took inspiration from biology, building cathedrals of ideas on the basis that all phenomena commonly regarded as ‘things’ are actually processes, and hence organism-like, culminating in a ‘philosophy of organism’ (Whitehead Citation1925, Citation1929). In this perspective, the onus of explanation is on stability rather than change, and those explanations must accommodate causalities that may be complex, diverse and extended in time. Philosophers of biology (Nicholson and Gawne Citation2015; Dupré and Nicholson Citation2018) have shown how Whitehead’s process philosophy influenced the ‘organicists’: pioneers of philosophical biology between the world wars who developed ideas of process in organisms that anticipated the niche construction perspective of the later 20th century. In fact, Whitehead’s teaching at Harvard in 1924–5 reveals his reciprocal interest in the emerging ‘organicist’ perspective on what has come to be called niche construction (P. A. Bogaard Citation2020).
One figure of archaeological interest in the ‘organicist’ movement was Ludwig von Bertalanffy (Von Bertalanffy Citation1941). By the 1950s, Bertalanffy’s focus had shifted from ‘organism’ to ‘system’ in his efforts to transform biology into a ‘nomothetic’ science based on general laws (Pouvreau and Drack Citation2007; Dupré and Nicholson Citation2018), and he is better known for General System(s) Theory (Von Bertalanffy Citation1949, Citation1950). GST was a generalising, formal mathematical framework of pattern-searching for laws applicable to the natural and social sciences. It was one of many influences on American ‘New Archaeologists’ of the 1960s, who developed what became known as a ‘processual’ approach (Kushner Citation1970; Trigger Citation2006, 418–22). Flannery (Citation1967, Citation1972) used GST to frame cultural change as an ecologically driven process, abstracted from its historical context, in order to generate ‘rules’ or principles of systems dynamics. Binford’s definition of ‘process’ as ‘the dynamic relationships (causes and effects) operative among the components of a system or between systemic components and the environment’ (Binford Citation1968, 269) similarly reflects GST. The ‘processualism’ of New Archaeology also referred to the comparative anthropological goal of identifying ‘a finite number of general cultural processes’ (Caldwell Citation1959, 306), to which Binford (Citation1968) added his distinctive emphasis on ‘middle range’ processes linking past dynamics with the static material archaeological record.
In sum, ‘processual’ New Archaeology never embraced process as a contingent, historical unfurling of particular circumstances, the explanatory priority of stability over change, or the prospect of complex, diverse causality. GST-influenced 1960s processualism gave rise to models of domestication that depended on simplified, human-centred causality and rationality. Classic examples include the Broad Spectrum Revolution model and related perspectives from Optimal Foraging Theory (Flannery Citation1969; Stiner Citation2001). These models are useful in a heuristic sense (Outram and Bogaard Citation2019, 29–49), but as literal predictions or adequate ‘explanations’ for the origins of farming, they have since been found wanting, giving way in particular to NCT perspectives (Zeder Citation2012b, Citation2016, Citation2017).
Here, we explore process philosophy as a foundation for current NCT-influenced perspectives on domestication relationships that are empirically based. We develop the argument that ‘domestication’ is more usefully understood as one set of processual relationships within a wider sea of processes, and that its isolation from these wider processes takes us farther away from, rather than closer to, understanding their causal background. We develop this argument by first considering how philosophers of biology think about organisms as processes, how this extends to non-living matter, and why this is relevant for studies of domestication. We then consider the emergence of ‘domestication’ as a term within European colonial history. Finally, we highlight current empirical evidence for domestication phenomena in different parts of the world, and on different temporal and spatial scales, in order to demonstrate how a process archaeology perspective accommodates the insights of NCT/EES, while expanding the possibilities for causal explanation.
Processes versus things
To observe that domestication, like all of evolution, is a process, or a cascade of processes, is banal. On a metaphysical level, however, prioritising processes over the ‘things’ we might abstract from them – that is, placing the onus of explanation on stability rather than change – remains a radical and challenging perspective. The philosopher of biology John Dupré has developed this perspective in a series of works and collaborations (Dupré Citation2012, Citation2017, Citation2020; Dupré and Nicholson Citation2018).
Building on work by Nicholson and Gawne (Citation2015), Dupré and Nicholson (Citation2018) identify the precursors of modern philosophy of biology in ‘organicism’, a movement that itself took inspiration from Whitehead’s (Citation1925) book, Science and the Modern World (Whitehead Citation1925). As he unpacks the rise of science over centuries, Whitehead’s concern in SMW was to show how Newtonian assumptions left physics unprepared for the ‘shock’ of relativity theory. He proposed a ‘philosophy of organism’ or ‘nature,’ and set the scene for biology to seek its metaphysical foundation not from physics, but from its own observations about organisms. Organicists took up this challenge and developed their own processual line of thought founded on the insistence that organisms were processes, and that regarding them as things was an obstacle to understanding. Thus, wrote Woodger (Citation1929, 219), with serendipitous reference to the first domesticate:
“Your pet dog to-day and your pet dog yesterday are two different temporal parts of the same dog, just as his head and his tail are two different spatial parts of the same dog. It is in virtue of the particular kind of continuity of the dog yesterday and the dog to-day that we call it the ‘same’, and this seems to be the proper sense of the term. But it can no more be taken for granted that to-day’s temporal part is the same as yesterday’s than it can be taken for granted that one spatial part, e.g. the head, is the same as another, e.g. the tail. We know, in fact, that they are not the same. Organisms are temporally as well as spatially differentiated.”
That these observations remain radical in their implications is borne out by the continued dominance of thing- and substance-based thinking in biology (Dupré and Nicholson Citation2018). If Woodger’s dog is regarded as a ‘thing’, or a combination of substances, rather than a dynamic ‘process’, its persistence through time is taken for granted, or ‘explained’ as subject to a lack of external forces causing change. If instead the dog is a dynamically stabilised process, this persistence demands explanation and leads to understanding of how and why the dog functions.
In addition, by thinking of the dog as a thing and not a process, we will tend to misunderstand or even deny its agency (Dupré Citation2017; Walsh Citation2018). This insight is at the heart of Lewontin’s critique of classical Darwinism and the modern synthesis, in which organisms are the object of evolution rather than its subject (Lewontin Citation1985). In archaeological narratives under the lingering influence of classical Darwinism, an implicit thing- or substance-based metaphysics colludes with the view that humans are an exclusive source of effective agency. Particularly in any study of niche construction involving humans, including ‘domestication’ relationships, denying other organisms agency automatically undermines the very insights that niche construction offers as a theory. As agents, organisms have goals, and means of attaining them, and they respond to ‘affordances’ (Gibson Citation1979), ‘the joint product of the organism’s own capacities and the features of its setting’ (Walsh Citation2018, 181).
If humans as organisms are processes, too, then process ontology brings in wider questions of political philosophy that also impinge on discussion of domestication. Individual self-interest has been a prominent ‘explanation’ for how domestication was initiated (Hayden Citation1990). A process perspective, however, draws attention to the cooperative nature of individual organisms as parts of ‘colonies’, lineages and so on. Human niche construction provides especially dramatic examples of this ‘super-organism’ level of agency (Dupré Citation2020) and is arguably more relevant to the initiation of early farming as a cooperative endeavour than perspectives that assume ‘hard-boundaried self-interested individualism’ (Dupré Citation2020) familiar from the contemporary political mainstream.
Though both dogs and humans are processes, or hierarchies of processes (metabolism, reproduction, etc.), they are clearly differentiated by their time scales (life cycles), spatial scales and species-specific trajectories (cf. Dupré Citation2020). This differentiation allows for potential independence as well as forms of co-dependence. A more dramatic example is microorganisms, functioning on vastly different temporal and spatial scales to their dog or human hosts. Together they can be thought of as symbiotic wholes or holobionts (Margulis Citation1991): universes of processes within processes, with fluid boundaries. Recognising the processual nature of organisms ushers in a far richer ecology and potential for understanding. An emerging challenge (and opportunity) for a process archaeology of domestication is therefore to consider not only the causal agencies of dog and human, but also their microbiomes.
A final consideration for this discussion of things versus processes is whether the process perspective should be limited to organisms. As argued by Malafouris et al. (this volume), what archaeologists call ‘material culture’ – the materials and objects entangled with humans – is deeply engaged in their mutual becoming. Dupré (Citation2020) suggests that the way to conceptualise both living and non-living matter in a process ontology is to think of their stabilisation as playing out on radically different timescales. A process archaeology of domestication thus accommodates the niche constructing agencies of all interacting organisms, as well as the agencies of their material culture.
The ‘invention’ of domestication
Stépanoff and Vigne (Citation2018) have drawn attention to 18–19th European century conceptions of ‘domestication as domination’. As pointed out by Digard (Citation1990), the power as well as the fallacy of this view are neatly illustrated by images of Lord Rothschild driving a zebra-drawn carriage in the late 19th century ().
Figure 1. Lord Walter Rothschild’s carriage drawn by zebras and a disguised horse, in front of the Royal Albert Hall (© The Trustees of the Natural History Museum, London. Licensed under the Open Government Licence).

From the Middle Ages to the 18th century, the notion of domestic animals did not necessarily imply human domination, but rather proximity to human habitation (Latin domus). In scientific taxonomy, the word domesticus still echoes this old conception: Passer domesticus (House sparrow), Acheta domesticus (House cricket) or Mus musculus domesticus (House mouse) are called domestic though they are ‘wild commensals’ in present zoological terminology.
From the 18th century, naturalists developed new concepts inspired by confidence in human power over nature. For Buffon, mirroring wild animals created by God, domestic animals are quasi ‘creations’ of human labour. They have been withdrawn from the rules of nature and made ‘unnatural’: ‘Man changes the natural state of animals by forcing them to obey and serve him for his use’ (De Buffon Citation1753). These concepts were stimulated by new breeding practices developed in the 17–18th centuries aiming to ‘improve’ numerous breeds of domestic animals including horses, pigs, dogs, and chickens through selection and hybridisation with Asian populations, just as horticulturists were simultaneously creating and marketing hundreds of new flower and tree cultivars.
During the 19th century, the word ‘domestication’ came into use, designating a specific scientific and economic field in the frame of the Societies of Acclimatisation (founded in Paris in 1854, and in London in 1859). The aim of these societies was to submit all animals that could be economically useful to human control by collecting them from different parts of the world, and acclimatizing them in European countries. The famous criterion of domestication, reproduction under human control, introduced by the founder of the French Society of Acclimatisation, Isidore Geoffroy Saint-Hilaire, is directly borrowed from the rules of international competitions organized for the domestication of exotic species. Submitting wild animals became a prestigious game for European elites, as illustrated by Lord Rothschild’s famous zebra carriage (). Thus, the cradle of ‘domestication’ is in a sense western European parks of acclimatisation of the 19th century, and European colonial conquest, submission and improvement. To study the associations between human and non-human societies in other areas and other periods, we need to enlarge our understanding of this notion.
Given its origins, the term ‘domestication’ tends to imply closely controlled management. There are rich ethnographic examples of alternative approaches in which people explicitly recognise the reciprocal contributions and agency of other species, and that their own agricultural practices serve the needs of these species (e.g. Gudeman and Rivera Citation1990; Ingold Citation1996). Stépanoff (Citation2017) has contrasted Siberian/reindeer and European/industrial perspectives on human-animal domestication relationships, raising also the prospect that the lasting biological effects of domestication on non-human species – a ‘domestication syndrome’ (see below) of traits – can range from ‘low’ to ‘high’, depending especially on the degree of reproductive control.
Here, we use the term ‘domestication’ in a broad sense, in reference to the relationships between humans, plants and animals that are not necessarily initiated or closely monitored by humans, but which persist for myriad reasons, normally presumed to be ‘mutual benefits’ to the species involved. In sympathy with NCT and EES (Müller Citation2017), we choose not to limit the definition using an arbitrary threshold of reproductive isolation or genetic change. It is only by allowing for the definition to expand beyond its standard confines that we can contextualise the peculiarity of recent European approaches.
Current perspectives on domestication in (bio)archaeology
Current work on domestication relationships in archaeology offers multiple avenues for exploring the potential of a process approach. We offer ‘vignettes’ of five such topics.
Domestication as a dynamic, ongoing process
A misconception embedded in archaeological discussion of domestication is to talk of ‘domestication events’ and to seek the ‘origins’ in time and space of a domesticate. Standard summaries state, for example, that barley was domesticated ~8000 BC in the Upper Euphrates, or rice was domesticated ~6500 BC in the Yangtze valley (e.g. Bellwood Citation2005). Archaeologists typically use these dates as markers in cultural-historical accounts of Neolithic ‘revolutions’, while geneticists model the age and population size of such ‘events’ with coalescence theory (e.g. Bollongino et al. Citation2012). But as a process, domestication is both extended and structured over time, whether the pace is fast, slow or variable. The structure and rate of this process is crucial for assessing the causal framework. The more extended in time a process unfolds, the greater the likelihood that the causal elements were diffuse and complex. To state that domestication traits became ‘fixed’ is itself shorthand for indicating a shift in the pace of change, though of course the evolution of the taxa did not cease. To comprehend domestication as an ongoing process, stability requires as much explanation as change.
The growth of data related to domestication processes has demanded a revision of how the process is conceptualised. A generation ago, the prospect of a rapid domestication process (20–100 years) emerged from experiments that modelled strong selection on a single key recessive gene of interest in cereals like wheat, emphasising a shift in human harvesting technology (sickle use) at the forefront of cause (Hillman and Davies Citation1990). In animal domestication, the Russian Fox-Farm experiment suggested that numerous morphological and behavioural changes occurred within a few decades of selection for tame behaviour (Trut Citation1999; Lord et al. Citation2020a).
The bioarchaeological evidence of plants and animals, however, has resisted expectations of rapid change. Instead, diverse cereals including wheat, barley, rice and pearl millet (Tanno and Willcox Citation2006; Fuller et al. Citation2014, Citation2021) exhibit both protracted periods of morphological change over millennia and variable rates of change (Allaby et al. Citation2017). West Asian livestock and Chinese pigs alike, whether considered in terms of size and shape of individual elements, log standard methods, or age profiles (e.g. Hongo and Meadow Citation1998; Cucchi et al. Citation2011, Citation2016; Peters, Arbuckle, and Pöllath Citation2015; Dong and Yuan Citation2020), similarly reflect protracted change, making simple assignment of specimens to wild or domesticated status difficult.
To assess the pace and variability of domestication processes, we require a metric applicable across a wide variety of species. The ‘Haldane’ unit offers an estimate of how much a population shifts in the distribution of measurements of a trait per generation. A Haldane of 1 indicates a change in one standard deviation unit in a single generation. Previous estimates of plant traits have calculated averages over entire domestication ‘episodes’ (i.e. early periods of change), and detected similarity in the pace of seed size changes across plant families, which tends to be slower than the evolution of non-shattering (Fuller et al. Citation2014). To appreciate the process, we need to embrace variability in rate within and across taxa (Allaby et al. Citation2017; Allaby, Stevens, and Fuller Citation2020).
In , rates of change are illustrated with examples from China and western Asia. All taxa display episodes of accelerated phenotypic change, raising questions of causal linkage with local cultural processes. In Yangtze rice (), a rapid increase in selection for non-shattering took place in the 5th millennium BC, while ~4000 BC there was a rapid increase in grain size during a period of intensive management of rice and water in small field systems (Weisskopf et al. Citation2015). Pigs in the Yellow River region () show accelerated size reduction in the 6th millennium BC, corresponding with marked increases in sedentism, demographic growth and millet agriculture (Stevens and Fuller Citation2017; Leipe et al. Citation2019). In western Asia, increasing grain size in einkorn in the 9th millennium BC preceded an upturn in proportions of cereal non-shattering (). The non-shattering increase in the 8th millennium BC coincides with a continuing trend of increased specialisation and frequency in sickle blades, suggesting some kind of co-evolutionary process, though it is clear that sickle use per se did not drive the evolution of non-shattering (Maeda et al. Citation2016). Lithic microwear suggests that the proportion of mature (as opposed to green) cereal plants harvested also increased over this period (Ibáñez et al. Citation2016), reflecting cereal populations that shifted towards non-shattering and more even ripening.
Figure 2. (a). Selected examples of morphological change from China, assemblage mean and standard deviation plotted against median age: rice non-shattering (Allaby et al. Citation2017), rice grain size and soybean size (Fuller et al. Citation2014), and pig molar size (Luo Citation2007; Wang et al. Citation2015). (b). The same dataset converted to Haldane rate estimates over subsets of the data in comparison to average Haldane rate based on a regression through the whole dataset (assumes 2-year generation for pigs, 1-year for crops).
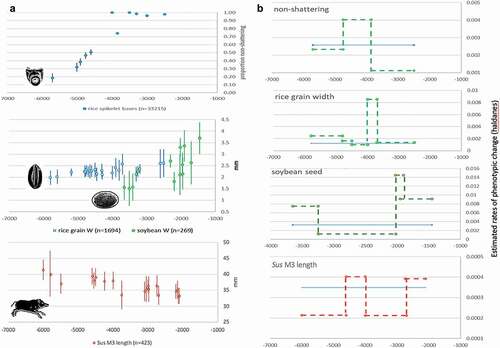
Figure 3. (a). Selected examples of morphological change in western Asia: einkorn wheat non-shattering (Allaby et al. Citation2017), einkorn grain size (Fuller et al. Citation2014; Kabukcu et al. Citation2021) and sheep size based on talus breadth (data compiled by authors Arbuckle and Peters based on their own work and supplemented with published (Küchelmann Citation2019) and unpublished data). (b). The same dataset converted to Haldane rate estimates over subsets of the data in comparison to average Haldane rate based on a regression through the whole dataset (assumes 3-year generation for sheep, 1-year for crops).
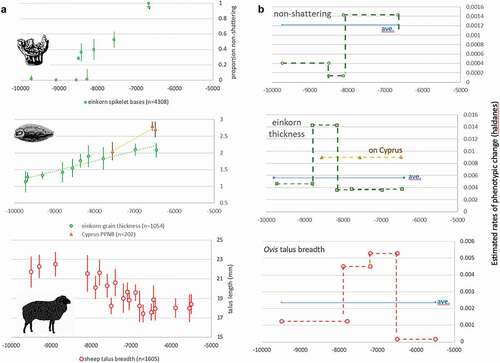
In western Asia, the emergence of non-shattering cereals coincides with increased rates of change in sheep (), suggesting links between domestication processes in different taxa. At Aceramic Neolithic Aşıklı Höyük (central Anatolia), dung concentrations reveal that sheep and goats were penned between houses by c. 8300 BC (Stiner et al. Citation2014). However, restrictions on mobility and dense corralling of caprines caused lasting joint damage (Zimmermann et al. Citation2018), likely increasing the abortion and neonate death rates (Pöllath et al. Citation2021). Waste products from caprine penning were subsequently integrated in small-scale intensive cultivation, indicated by stable isotope values derived from Neolithic grains (Styring et al. Citation2017) and weed assemblages at Catalhöyuk from c. 6700 BC (Green, Charles, and Bogaard Citation2018), where the evolution of non-shattering continued in the ‘new glume wheat’ (Triticum timopheevii group) (Charles et al. Citation2021). Thus, the dynamics of domestication processes entail interlinked agencies of animals, plants, people and material culture. The evidence of rate fluctuations makes clear that domestication was a complex, dynamic process, one that we have only begun to comprehend.
From domestication ‘syndromes’ and ‘pathways’ to processes
Domestic organisms are often described as possessing a suite of shared phenotypic characteristics that distinguish them from their wild progenitors. In plants, these include an increase in the size of the edible portion (fruit, tuber, grain), the loss of seed dispersal (shattering) in seed crops, increased apical dominance, and shifts in the timing of production (i.e. greater synchronicity in seed crops and less in some tuber crops) (Denham et al. Citation2020). Domestic animals share traits including, but not limited to, variable, non-camouflage colouring, giant and miniature sizes, and tamer dispositions (Lord et al. Citation2020a).
Though Darwin (Citation1868) observed and discussed the co-occurrence of these traits in a wide variety of domestic taxa, the term ‘domestication syndrome’ was first coined by botanists in the later 20th century (Harlan, De Wet, and Price Citation1973; Hammer Citation1984). The domestication syndrome was developed by tabulating parallel traits distinguishing cultivated cereals from their wild relatives. Harlan, De Wet, and Price (Citation1973) explored parallels across wheat, barley, oats, rice, sorghum, maize and pearl millet, while Zohary and Hopf (Citation1973) drew attention to parallel changes in pulses. Hammer (Citation1984) later recognized that many of these same traits could be found much more widely, and distinguished different combinations in seed-grown ‘field’ and ‘garden’ crops.
Exploration of common traits of vegetative crops, including tubers and bananas, suggests the presence of environmentally influenced tendencies not only of hard-wired traits, and also that the expressions of some are opposite to those of seed crops (Denham et al. Citation2020). These contrasts highlight the lack of a universally applicable ‘domestication syndrome’. The syndrome concept has aided comparative studies, but variation across crops and crop types highlights its limitations.
Despite widespread use since the 1980s, the domestication syndrome in animals has not been rigorously evaluated. A recent study (Lord et al. Citation2020a) questioned the direct link between selection for tameness (as in the Russian Farm-Fox Experiment (Belyaev, Plyusnina, and Trut Citation1985)) and the appearance of traits associated with the domestication syndrome. The same study also questioned the evidence for numerous syndrome traits, including the assertion (Kruska Citation1988) that domestic animals possess smaller brains than their wild counterparts (Lord, Larson, and Karlsson Citation2020b).
Domestication studies have benefited from the recognition of multiple ‘pathways’ through which different animal species entered the human niche. Zeder’s three domestication pathways, the commensal route, the prey route, and the directed route, categorised species by the manner in which they first began their association with people (Zeder Citation2012a). The ‘pathways’ concept has since been applied to plants, yielding at least six different routes through which the association between plants and people arose (Fuller and Denham CitationIn press): the cereal pathway (including most seed crops), the segetal pathway by which new cereals and many fodder crops were recruited from weeds, and additional pathways for tubers, tree fruits, fibre crops or fibre varieties of crops like hemp and flax. Another pathway involves forms of ecosystem engineering that transform landscapes for production of species like oil palm or sago palm, which were accompanied, at least initially, by few obvious trait adaptations.
Like domestication syndromes, ‘pathways’ can be understood as hypotheses about processes, but where causal mechanisms are necessarily generalised. The emerging challenge is to move from checklists of traits or generalised scenarios of domestication and towards evidence-based accounts of situated processes. Jones et al. (Citation2021) have recently taken a step in this direction through experimental work to improve understanding of the ecological processes underlying selection for domestication traits in western Asian cereals. A key finding is that increases in seed size are likely a by-product of selection for larger, more competitive plants through improved growing conditions (e.g. fertility) resulting from human management, and that larger seeds did not necessarily increase yield. This lends further support to the view that human intention and rationality played a complex and indirect role in the selection of domestication traits (see also Allaby, Stevens, and Fuller Citation2020).
Cooperation versus competition
The interconnectedness of all life is a fundamental principle of process philosophy in biology: all organisms, including viruses, bacteria and complex multicellular organisms, can only survive as part of entanglements with other organisms (Dupré and Nicholson Citation2018). Dupré and O’Malley (Citation2009) describe these relationships as ‘collaborations’, ranging from cooperative to competitive. Many explanatory frameworks for the origins of domesticated animals and plants, however, emphasise competition over cooperation, especially between humans. We argue that most if not all domestication relationships contain a strong cooperative element. Not all cooperative relationships begin with mutual or equal benefits for all partners, and many involve the mere tolerance of organisms in an environment, conventionally termed commensalism (O’Connor Citation1997; Vigne Citation2011; Zeder Citation2012a).
Competition between human individuals or groups plays a central role in economy- and ownership-centred narratives of the emergence of farming. In his influential model, Hayden (Citation1990) argued that individual ‘aggrandizers’ used rare plants and animals in feasts to gain prestige and status, thereby initiating the management of rare food resources. Although the focus on individual ‘aggrandizers’ and rare foods has diminished in importance in recent years due to the lack of supporting archaeological evidence (Hayden Citation2009; Zeder Citation2009), competition continues to play a pivotal role in models that place early agricultural intensification and domestication primarily in the context of competitive feasts and/or emerging private property (e.g. Lueck and Torrens Citation2020).
These models are too narrow to account for all processes that culminate in domestication relationships. By concentrating on ‘selfish individuals’ competition-centred models fail to acknowledge widespread evidence for cooperation in early farming societies. For example, early Neolithic communities in western Asia maintained relatively egalitarian social and economic structures, with communal activities and funerary rituals that promoted community cohesion and household integration (Kuijt Citation2004, Citation2008; Twiss Citation2008; Bogaard et al. Citation2009; Makarewicz, Finlayson, and Biehl Citation2018). This does not mean that social differentiation and individuals with access to special knowledge would not have existed, but a focus on these competitive elements masks the inherently cooperative nature of early Neolithic farming communities.
Cooperation in domestication relationships also developed between humans and non-humans. This took place, for example, when humans became seed dispersers in place of large herbivores in numerous small-seeded crop progenitors (Spengler and Mueller Citation2019; Mueller et al. Citation2020), and when commensal animals were mutually tolerated in anthropogenic environments (Zeder Citation2012a). Both the wolf (Lahtinen et al. Citation2021) and the wild boar (Price and Hongo Citation2020) were likely initially attracted to human settlements by human food waste before entering a cooperative relationship, obtaining protection against predators and a secure food source from the human community. Small felids colonized early farming settlements in both western and eastern Asia, a process that predated cat domestication (Vigne et al. Citation2004; Hu et al. Citation2014). While cats potentially obtained shelter and access to prey, humans would have benefited from their role as protectors of granaries against pests who were themselves attracted to the human niche. This narrative has been undermined by a recent study that questioned the efficiency of felids at controlling rodent populations (Mahlaba et al. Citation2017). A lack of detrimental effects may be sufficient to foster tolerance of commensals.
The integration of plants and animals in mixed agricultural systems further demonstrates how cooperation in domestication relationships can emerge between species that are otherwise competitors. In early farming systems in western Asia, integrating livestock with crop cultivation provided manure and fodder/stubble grazing, driving the intensification of early plant and animal husbandry practices (Bogaard Citation2005). Outside of such multispecies collectives, however, wild cereals and other palatable, large-seeded species are sensitive to heavy grazing (Noy-Meir Citation1990). These dynamics were turned around with the development of mixed farming systems, where the growth of cereals and other grain crops significantly benefits from their cooperation with grazers. In the context of African savannas, the palatability of sorghum to early livestock may account for why this species appears to undergo domestication in the eastern Sudan (Winchell et al. Citation2018).
Human and non-human agency
Decentering domestication away from humans has several important consequences. First, agency is shifted from questions of human intentionality and distributed to the members of multi-species hybrid communities. Research on goats (Capra hircus) suggests that their ecological network was crucial in directing the domestication relationship. Within the mountainous range of the bezoar (Capra aegagrus), a combination of altitudinally structured landscape, strong diurnal and seasonal variations in resources and the behavior and physiology of the bezoar (Korshunov Citation1994; Lomolino Citation2001) created a unique collaborative relationship between bezoars and people in the central Zagros mountains of western Asia by the 9th millennium BC. At this time, archaeological signatures of goat husbandry, such as sex- and age-specific culling, penning and dung use, are first evident in the mid-altitudinal winter habitats of wild goats. This period has also revealed the first evidence for zoonoses (Fournié, Pfeiffer, and Bendrey Citation2017), suggesting goats and people were in closer proximity as a result of goat seasonal mobility between mid and upper elevations and were accompanied by human co-dwellers. Recent aDNA analysis of early managed goats in the central Zagros suggests some genetic divergence from local wild populations, but without a genetic bottleneck or phenotypic differentiation (e.g. coat pigmentation) from wild goats (Daly et al. Citation2021). Goat management appears in adjacent lowland regions in the 8th millennium BCE (Saña Segui Citation2000), further reflecting the agency of goats whose physiology and disease ecology promoted transhumant co-residence with humans. The ecological and behavioural patterns of goats prevented them from being translocated to lowland regions for more than a millennium, at which point they became morphologically distinct in the absence of gene flow with wild populations (Daly et al. Citation2021).
Second, by focusing on networks of interaction, a process approach emphasizes that each community member actively constructs niches. Domesticatory co-evolutionary relationships are reincorporated into their broader ecological contexts. This approach encompasses historically unique collaborations such as those between humans and foxes (Vulpes vulpes) in prehistoric western Asia (Yeshurun, Bar-Oz, and Weinstein-Evron Citation2009), and relationships with organisms not involved in ‘modern’ global economies such as ‘reindeer herding’, and management of nondomestic crops and aquatic resources in neo-tropical forests (Fausto and Neves Citation2018). One promise of an expanded view of domestication is recognition of resource management techniques and economically useful plant/animals hiding in plain sight beyond the temperate cereal and livestock zones. Recent examples include the açai (Euterpe oleracea) and pequi (Caryocar brasiliense) (Fausto and Neves Citation2018).
Third, if domestication is perceived as an emergent property of the process of multi-species collaboration, and one that is never completed but always in the process of becoming, single-cause explanations (e.g. demographic growth, game depression, comet impacts, feasting) recede in favour of more complex, multi-causal accounts that accommodate non-human agencies. Moreover, simple explanations of domestication tend to underestimate the productivity and resilience of forager-hunter economies and to project later agricultural economies back in time. Food insecurity is frequently assumed to instigate domestication relationships even though increased demand for food is not related to domestication in any straightforward manner (Arbuckle Citation2015). Critique of the 1960s processual Broad Spectrum Revolution model has pointed to indications of diverse resource use in situations of resource abundance rather than scarcity (Zeder Citation2012b), of which early agricultural Çatalhoyuk, central Anatolia, now provides a clear example through the 7th millennium BC (Wallace et al. Citation2019; Bogaard et al. Citation2021).
Turning to yield, recent experiments with North American crop progenitors have shown that wild plants are capable of producing similar yields to their domesticated relatives when sufficient labour is invested in cultivation (Mueller, White, and Szilagyi Citation2019). Since the selective pressures associated with cultivation are the most likely explanation for how domestic annual plants emerged, these results suggest that some increases in yield pre-date domestication. The implication is that domestication was an incidental byproduct of increased investment in cultivating certain crops, not the mechanism by which people achieved these increases, as also hypothesised also for west Asian cereals (Jones et al. Citation2021).
For animals, behavioural plasticity has long been appreciated as an important factor in domestication (Zeder Citation2006). Recent experimental work with wild boar has also demonstrated that the entire skeleton of animals, and not just their behaviours, show immediate responses to life in anthropogenic ecosystems (Harbers et al. Citation2020), facilitating the emergence of new morphologies (Neaux et al. Citation2021). Similarly, wild crop progenitors may be especially plastic, capable of responding to cultivation or anthropogenic ecosystems (Ménard et al. Citation2013; Piperno et al. Citation2015, Citation2019; Matesanz and Milla Citation2018; Mueller, White, and Szilagyi Citation2019).
We encourage a shift of focus from perceived human needs to the processes and contexts of interaction. The question of ‘how’ this interaction operates becomes the answer to ‘why’ domestication occurs (Stépanoff and Vigne Citation2018). Mueller et al. have argued that the domestication of certain annual seed crops in eastern North America arose from deep evolutionary relationships between these plants and bison herds (Mueller et al. Citation2020). They envision the early Holocene landscape created by grazing bison as a template for the later agroecosystems created by people: hotspots (Marshall et al. Citation2018) harbouring dense stands of seed-bearing plants in nitrogen-enriched soils exploited and then recreated by people. This process may have played out in various forms in other regions as people became effective seed dispersers (see Guimarães, Galetti, and Jordano Citation2008 for South America), thereby altering the selective pressures acting on plant communities that spurred phenotypic change (Spengler and Mueller Citation2019). Domestication of small-seeded annual plants can be seen as an emergent property of interactions between humans, grazing animals, and grasslands, playing out over extended periods of time.
From ‘domestic versus wild’ to ‘hybrid communities’
The conceptual dichotomy between ‘wild’ and ‘domestic’ emphasises arbitrary thresholds rather than processes, ignoring intermediate forms or taxa/communities that do not meet the wild/domestic criteria. This focus obscures the significance of hybrid communities.
A textbook example of hybrid communities that are conventionally separated into categories of ‘cultivated/domestic’ and ‘weed/wild’ are arable fields, which often support several crops associated with a weed community. In evolutionary terms, however, this distinction becomes obsolete when we consider cultivated fields as multispecies communities in highly disturbed habitats where all species experience selection pressures (Harlan and deWet Citation1965; Hammer Citation1984). In fact, ‘wild’ progenitors of crops are associated with an accompanying ‘weed’ flora, an observation that has only recently begun to enter into discussions of early cultivation scenarios in western Asia (Weide et al. Citation2018, Citation2021). Many weeds developed effective adaptations to different arable environments and ‘crop mimics’ exhibit many traits also found in crop species (Bogaard, Ater, and Hodgson Citation2018). The cereal mimicking Aegilops (goat-face grass) became a dominant and economically important component of crop harvests in extensive early urban agrosystems of western Asia; other weeds with ‘opportunistic’ habits offered valued sources of food or fodder in more intensively managed arable fields since the Neolithic (Bogaard, Ater, and Hodgson Citation2018).
The fundamental role non-crops play in arable communities includes pollinators (insects, bats), providers of nutrients or defence against pathogens (rhizobia bacteria in legume roots, endo- and exophytic microbes on roots and leaves in livestock manure and other degrading biological materials), grazers to encourage tillering/prevent lodging (Halstead Citation2006) and draught animals. No organism is truly independent, but rather part of a ‘holobiont’ (Margulis Citation1991) that typically consists of a larger scaffold organism interacting with a diverse array of (principally) microbial symbionts. While the role of such microbial partners was once assumed to be largely limited to nutrient uptake and conversion on plant roots and in animal digestive tracts, we are beginning to appreciate the range of roles they confer on their host, including defence mechanisms against pathogens (Rosshart et al. Citation2017), adaptive capacity (Alberdi et al. Citation2016), and in animals, even conditioning behaviour (Liang, Wu, and Jin Citation2018). Recent metagenomic research has found that wild and cultivated cereals affect the microbiome composition of the soil in different ways, with higher microbiome diversity in wild forms (Bulgarelli et al. Citation2015; Shenton et al. Citation2016) and implications for nutrient uptake or disease vectors (Pérez-Jaramillo, Mendes, and Raaijmakers Citation2016). Palaeogenomic analyses are creating new opportunities to identify ancient microbiomes in, for example, dental calculus, raising the prospect of reconstructing past holobionts (Warinner et al. Citation2014).
The domestic and wild dichotomy does not account for the continuum that often exists within a species. In pigs and rabbits, for example, selection over the past 10,000 years has resulted in a myriad of intermediary forms across the globe. Hybridisation is likely still taking place between populations of pigs along the domestication continuum, as in southern Europe (Frantz et al. Citation2019), where multiple lineages of pigs often cohabit, and interbreed, from highly selected ‘modern’ domestic pigs to free-living pigs and wild boar (Albarella, Manconi, and Trentacoste Citation2011).
Although most episodes of gene flow in pigs were likely not intentional, there is evidence of both unintentional and intentional breeding among highly differentiated ‘domestic’ and ‘wild’ forms in donkeys and camels. Donkey’s solitary behaviour and largely unsupervised foraging led to encounter-based breeding among managed, feral donkeys and African wild asses over 6,000 years (Marshall and Weissbrod Citation2009; Marshall et al. Citation2014). To maintain strong, hardy transport animals, Roman period and historic herders also intentionally encouraged interbreeding with large, arid adapted wild asses (Marshall and Weissbrod Citation2011). Donkey’s roles in trade led to further interbreeding among domestic donkeys in diverse human networks and over long distances.
As unattended foraging is also typical in camels, addressing an animal’s domestication status within the native distribution of the wild form is difficult. Following long-distance trade with Bactrian camels into regions populated naturally by (wild) dromedaries more than 3,000 years ago, cross-breeding two- with one-humped camels became widespread practice in order to obtain performance and therefore highly valued hybrids for the caravan trade (Heide and Peters Citation2021).
Hybridisation along the domestication continuum is also common in plants. Flax absorbed genetic variability from local populations while spreading from western Asia into Europe during the Neolithic expansion (Gutaker et al. Citation2019). Multiple waves of intermediary forms of maize left Mesoamerica (Kistler et al. Citation2018), and later expansions carried altitudinal adaptations from a second wild progenitor species (Zea mexicana) which were taken up by populations in both South and North America, allowing for the occupation of the mountainous Andes (Calfee et al. Citation2021).
The extent of hybridisation along the domestication continuum varies dramatically. Palaeogenomic data suggest that hybridisation between wolves and dogs was extremely limited over the last ~11,000 years, implying some form of selection against gene flow (Bergström et al. Citation2020). This is particularly striking given that, though dogs, pigs, cattle and flax were all translocated into Europe during the Neolithic expansion, dogs did not admix significantly with local European wild populations of the same or related species (Larson and Fuller Citation2014).
Conclusions: process lessons for domestication, and vice versa
The process perspective as developed in the philosophy of biology, since emerging in archaeology (Gosden and Malafouris Citation2015), offers crucial metaphysical ‘scaffolding’ for the challenges of niche construction and the extended evolutionary synthesis, for which domestication has emerged as a model case study (Zeder Citation2016, Citation2017). To embrace these opportunities, we advocate combining the rich and multi-stranded nature of current (bio)archaeological data with the insights of process ontology, to investigate the causal background of domestication relationships. This shift of perspective is not simply an intellectual exercise, but fundamental to redirecting approaches to contemporary dilemmas surrounding food security and sustainability. Archaeologists are uniquely positioned to debunk the myth, invested in the very word ‘domestication’, that these relationships depend on human control and rationality. By striving for a more sophisticated deep-time understanding that depends on the variable dynamics, diversity, cooperative aspects, diffuse agency and hybrid nature of domestication relationships, the way is open to improve future strategies.
We also hope to have shown that the development of domestication studies in archaeology holds interest for process philosophy. By taking a topic such as ‘domestication’ that is inherently, unarguably processual in nature, we are confronted by the challenges of situating complex processes in time and space, and of addressing ‘process’ in a way that 1960s ‘processual’ archaeology neither attempted nor recognised.
Acknowledgments
This contribution and its wide authorship stem from a Catalysis Meeting held in March 2020, supported and hosted by the Santa Fe Institute. The meeting was also supported by a Starting ERC grant (ERC-2013-StG-337574-UNDEAD) awarded to G.L. H. Buitenhuis, H.C. Küchelmann and N. Pöllath kindly provided osteometric data for the diachronic analysis of sheep size (); funding by the Deutsche Forschungsgemeinschaft (DFG) to JP (Project No. 165831460) is gratefully acknowledged.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
Notes on contributors
Amy Bogaard
The authors of this paper are social and/or life scientists who share an interest in domestication processes in their widest sense.
References
- Albarella, U., F. Manconi, and A. Trentacoste. 2011. “A Week on the Plateau: Pig Husbandry, Mobility and Resource Exploitation in Central Sardinia.” In Ethnozooarchaeology. The Present and past of Human-Animal Relationships, edited by U. Albarella and A. Trentacoste, 143–159. Oxford: Oxbow.
- Alberdi, A., O. Aizpurua, K. Bohmann, M. L. Zepeda-Mendoza, and M. T. P. Gilbert. 2016. “Do Vertebrate Gut Metagenomes Confer Rapid Ecological Adaptation?” Trends in Ecology & Evolution 31 (9): 689–699. doi:https://doi.org/10.1016/j.tree.2016.06.008.
- Allaby, R. G., C. Stevens, L. Lucas, O. Maeda, and D. Q. Fuller. 2017. “Geographic Mosaics and Changing Rates of Cereal Domestication.” Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences 372: 1735. doi:https://doi.org/10.1098/rstb.2016.0429.
- Allaby, R. G., C. J. Stevens, and D. Q. Fuller. 2020. “A Novel Cost Framework Reveals Evidence for Competitive Selection in the Evolution of Complex Traits during Plant Domestication.” bioRxiv. doi:https://doi.org/10.1101/2020.10.30.362236.
- Arbuckle, B. S. 2015. “Large Game Depression and the Process of Animal Domestication in the near East.” In Climate Change in Ancient Societies, edited by M. S. Kerner, R. Dann, and P. Bangsgaard, 215–243. Museum Tusculanum Press: Copenhagen.
- Bellwood, P. 2005. First Farmers: The Origins of Agricultural Societies. Oxford: Blackwell.
- Belyaev, D. K., I. Z. Plyusnina, and L. N. Trut. 1985. “Domestication in the Silver Fox (Vulpes Fulvus Desm): Changes in Physiological Boundaries of the Sensitive Period of Primary Socialization.” Applied Animal Behaviour Science 13 (4): 359–370. doi:https://doi.org/10.1016/0168-1591(85)90015-2.
- Bergström, A., L. Frantz, R. Schmidt, E. Ersmark, O. Lebrasseur, L. Girdland-Flink, A. T. Lin, et al. 2020. “Origins and Genetic Legacy of Prehistoric Dogs.” Science 370 (6516): 557–564. DOI:https://doi.org/10.1126/science.aba9572.
- Binford, L. R. 1968. “Some Comments on Historical versus Processual Archaeology.” Southwestern Journal of Anthropology 24 (3): 267–275. doi:https://doi.org/10.1086/soutjanth.24.3.3629348.
- Bogaard, A. 2005. “‘Garden Agriculture’ and the Nature of Early Farming in Europe and the near East.” World Archaeology 37 (2): 177–196. doi:https://doi.org/10.1080/00438240500094572.
- Bogaard, A., M. Ater, and J. G. Hodgson. 2018. “Arable Weeds as a Case Study in Plant-human Relationships beyond Domestication.” In Hybrid Communities: Biosocial Approaches to Domestication and Other Trans-species Relationships, edited by C. Stépanoff and J.-D. Vigne, 97–112. London: Routledge.
- Bogaard, A., M. Charles, D. Filipović, D.Q. Fuller, L. Gonzalez Carretero, L. Green, C. Kabukcu, E. Stroud, and P. Vaiglova. 2021. “The Archaeobotany of Çatalhöyük: Results from 2009–2017 Excavations and Final Synthesis.” In Peopling the Landscape of Çatalhöyük: Reports from the 2009–2017 Seasons., edited by I. Hodder, 91–123. London: British Institute at Ankara.
- Bogaard, A., M. Charles, K. C. Twiss, A. Fairbairn, N. Yalman, D. Filipović, G. A. Demirergi, F. Ertuğ, N. Russell, and J. Henecke. 2009. “Private Pantries and Celebrated Surplus: Storing and Sharing Food at Neolithic Çatalhöyük, Central Anatolia.” Antiquity 83 (321): 649–668. doi:https://doi.org/10.1017/S0003598X00098896.
- Bogaard, P. A. 2020. “Whitehead and His Philosophy of Evolution.” In Whitehead at Harvard, 1924–1925, edited by V. Rothwell, B. G. Henning, and J. Petek, 75–88. Edinburgh: Edinburgh University Press.
- Bollongino, R., J. Burger, A. Powell, M. Mashkour, J.-D. Vigne, and M. G. Thomas. 2012. “Modern Taurine Cattle Descended from Small Number of near-Eastern Founders.” Molecular Biology and Evolution 29 (9): 2101–2104. doi:https://doi.org/10.1093/molbev/mss092.
- Bulgarelli, D., R. Garrido-Oter, P. C. Münch, A. Weiman, J. Dröge, Y. Pan, A. C. McHardy, and P. Schulze-Lefert. 2015. “Structure and Function of the Bacterial Root Microbiota in Wild and Domesticated Barley.” Cell Host & Microbe 17 (3): 392–403. doi:https://doi.org/10.1016/j.chom.2015.01.011.
- Caldwell, J. R. 1959. “The New American Archeology.” Science 129 (3345): 303–307. doi:https://doi.org/10.1126/science.129.3345.303.
- Calfee, E., D. Gates, A. Lorant, M. T. Perkins, G. Coop, and J. Ross-Ibarra. 2021. “Selective Sorting of Ancestral Introgression in Maize and Teosinte along an Elevational Cline.” bioRxiv. doi:https://doi.org/10.1101/2021.03.05.434040.
- Charles, M., D.Q. Fuller, T. Roushannafas, and A. Bogaard. 2021. “An Assessment of Crop Plant Domestication Traits at Çatalhöyük.” In Humans and Environments of Çatalhöyük: Reports from the 2009–2017 Seasons, edited by I. Hodder, 125–136. London: British Institute at Ankara.
- Cucchi, T., A. Hulme-Beaman, J. Yuan, and K. Dobney. 2011. “Early Neolithic Pig Domestication at Jiahu, Henan Province, China: Clues from Molar Shape Analyses Using Geometric Morphometric Approaches.” Journal of Archaeological Science 38 (1): 11–22. doi:https://doi.org/10.1016/j.jas.2010.07.024.
- Cucchi, T., L. Dai, M. Balasse, C. Zhao, J. Gao, Y. Hu, J. Yuan, J.-D. Vigne, and J P. Hart. 2016. “Social Complexification and Pig (Sus Scrofa) Husbandry in Ancient China: A Combined Geometric Morphometric and Isotopic Approach.” PLOS ONE 11 (7): e0158523. doi:https://doi.org/10.1371/journal.pone.0158523.
- Daly, K. G., V. Mattiangeli, A. J. Hare, H. Davoudi, H. Fathi, S. B. Doost, S. Amiri, et al. 2021. “Herded and Hunted Goat Genomes from the Dawn of Domestication in the Zagros Mountains.” Proceedings of the National Academy of Sciences 118 (25): e2100901118. doi:https://doi.org/10.1073/pnas.2100901118.
- Darwin, C. 1868. The Variation of Animals and Plants under Domestication. London: J. Murray.
- Darwin, C. 1881. The Formation of Vegetable Mould, through the Action of Worms: With Observations on Their Habits. London: J. Murray.
- De Buffon, G. L. L. 1753. Histoire naturelle: Générale et particulière. Paris: De l’Imprimerie royale.
- Denham, T., H. Barton, C. Castillo, A. Crowther, E. Dotte-Sarout, S. A. Florin, J. Pritchard, A. Barron, Y. Zhang, and D. Q. Fuller. 2020. “The Domestication Syndrome in Vegetatively Propagated Field Crops.” Annals of Botany 125 (4): 581–597. doi:https://doi.org/10.1093/aob/mcz212.
- Digard, J.-P. 1990. L’homme et les animaux domestiques: Anthropologie d’une passion. Paris: Fayard.
- Dong, N., and J. Yuan. 2020. “Rethinking Pig Domestication in China: Regional Trajectories in Central China and the Lower Yangtze Valley.” Antiquity 94 (376): 864–879. doi:https://doi.org/10.15184/aqy.2020.122.
- Dupré, J. 2012. Processes of Life: Essays in the Philosophy of Biology. Oxford: Oxford University Press.
- Dupré, J. 2017. “The Metaphysics of Evolution.” Interface Focus 7 (5): 20160148. doi:https://doi.org/10.1098/rsfs.2016.0148.
- Dupré, J., and D. Nicholson. 2018. “A Manifesto for A Processual Philosophy of Biology.” In Everything Flows: Towards a Processual Philosophy of Biology, edited by D. J. Nicholson and J. Dupré. Oxford: Oxford University Press. doi:https://doi.org/10.1093/oso/9780198779636.003.0001.
- Dupré, J. 2020. “Processes within Processes: A Dynamic Account of Living Beings and Its Implications for Understanding the Human Individual.” In Biological Identity: Perspectives from Metaphysics and the Philosophy of Biology, edited by A. S. Meincke and Dupré, 149–166. London: Routledge.
- Dupré, J., and M. A. O’Malley. 2009. “Varieties of Living Things: Life at the Intersection of Lineage and Metabolism.” Philosophy and Theory in Biology 1. doi:https://doi.org/10.3998/ptb.6959004.0001.003.
- Fausto, C., and E. G. Neves. 2018. “Was There Ever a Neolithic in the Neotropics? Plant Familiarisation and Biodiversity in the Amazon.” Antiquity 92 (366): 1604–1618. doi:https://doi.org/10.15184/aqy.2018.157.
- Flannery, K. V. 1967. “Culture History V. Culture Process: A Debate in American Archaeology.” Scientific American 217 (2): 119–122. doi:https://doi.org/10.1038/scientificamerican0867-119.
- Flannery, K. V. 1969. “Origins and Ecological Effects of Early Domestication in Iran and the Near East.” In The Domestication and Exploitation of Plants and Animals, edited by P. J. Ucko and G. W. Dimbleby, 73–100. London: Duckworth.
- Flannery, K. V. 1972. “The Cultural Evolution of Civilizations.” Annual Review of Ecology and Systematics 3 (1): 399–426. doi:https://doi.org/10.1146/annurev.es.03.110172.002151.
- Fournié, G., D. U. Pfeiffer, and R. Bendrey. 2017. “Early Animal Farming and Zoonotic Disease Dynamics: Modelling Brucellosis Transmission in Neolithic Goat Populations.” Royal Society Open Science 4 (2): 160943. doi:https://doi.org/10.1098/rsos.160943.
- Frantz, L. A. F., J. Haile, A. T. Lin, A. Scheu, C. Geörg, N. Benecke, M. Alexander, et al. 2019. “Ancient Pigs Reveal a near-Complete Genomic Turnover following Their Introduction to Europe.” Proceedings of the National Academy of Sciences of the United States of America 116 (35): 17231–17238. DOI:https://doi.org/10.1073/pnas.1901169116.
- Fuller, D. Q., and T. Denham. In press. “Coevolution in the Arable Battlefield: Pathways to Crop Domestication, Cultural Practices and Parasitic Domesticoids.” In The Convergent Evolution of Agriculture in Humans and Insects, edited by T. Schulz, P. N. Peregrine, and R. Gawne. Cambridge, MA: MIT Press.
- Fuller, D. Q., A. Barron, L. Champion, C. Dupuy, D. Commelin, M. Raimbault, and T. Denham. 2021. “Transition from Wild to Domesticated Pearl Millet (Pennisetum Glaucum) Revealed in Ceramic Temper at Three Middle Holocene Sites in Northern Mali.” African Archaeological Review 38 (2) (March): 211–230. doi:https://doi.org/10.1007/s10437-021-09428-8.
- Fuller, D. Q., T. Denham, M. Arroyo-Kalin, L. Lucas, C. J. Stevens, L. Qin, R. G. Allaby, and M. D. Purugganan. 2014. “Convergent Evolution and Parallelism in Plant Domestication Revealed by an Expanding Archaeological Record.” Proceedings of the National Academy of Sciences of the United States of America 111 (17): 6147–6152. doi:https://doi.org/10.1073/pnas.1308937110.
- Gibson, J. J. 1979. The Ecological Approach to Visual Perception. Boston: Houghton Mifflin.
- Gosden, C., and L. Malafouris. 2015. “Process Archaeology (P-Arch).” World Archaeology 47 (5): 701–717. doi:https://doi.org/10.1080/00438243.2015.1078741.
- Green, L., M. Charles, and A. Bogaard. 2018. “Exploring the Agroecology of Neolithic Çatalhöyük, Central Anatolia: An Archaeobotanical Approach to Agricultural Intensity Based on Functional Ecological Analysis of Arable Weed Flora.” Paléorient 44 (2): 29–44.
- Gudeman, S., and A. Rivera. 1990. Conversations in Colombia: The Domestic Economy in Life and Text. Cambridge: Cambridge University Press.
- Guimarães, P. R., M. Galetti, and P. Jordano. 2008. “Seed Dispersal Anachronisms: Rethinking the Fruits Extinct Megafauna Ate.” PloS One 3 (3): e1745. doi:https://doi.org/10.1371/journal.pone.0001745.
- Gutaker, R. M., M. Zaidem, Y.-B. Fu, A. Diederichsen, O. Smith, R. Ware, and R. G. Allaby. 2019. “Flax Latitudinal Adaptation at LuTFL1 Altered Architecture and Promoted Fiber Production.” Scientific Reports 9 (1): 976. doi:https://doi.org/10.1038/s41598-018-37086-5.
- Halstead, P. 2006. “Sheep in the Garden: The Integration of Crop and Livestock Husbandry in Early Farming Regimes of Greece and Southern Europe.” In Animals in the Neolithic of Britain and Europe, edited by D. Serjeantson and D. Field, 42–55. Oxford: Oxbow.
- Hammer, K. 1984. “Доместикационный Синдром.” Die Kulturpflanze 32 (1): 11–34. doi:https://doi.org/10.1007/bf02098682.
- Harbers, H., C. Zanolli, M. Cazenave, J.-C. Theil, K. Ortiz, B. Blanc, Y. Locatelli, et al. 2020. “Investigating the Impact of Captivity and Domestication on Limb Bone Cortical Morphology: An Experimental Approach Using a Wild Boar Model.” Scientific Reports 10 (1): 19070. DOI:https://doi.org/10.1038/s41598-020-75496-6.
- Harlan, J. R., J. M. J. De Wet, and E. G. Price. 1973. “Comparative Evolution of Cereals.” Evolution 27 (2): 311–325. doi:https://doi.org/10.1111/j.1558-5646.1973.tb00676.x.
- Harlan, J. R., and J. M. J. deWet. 1965. “Some Thoughts about Weeds.” Economic Botany 19 (1): 16–24. doi:https://doi.org/10.1007/BF02971181.
- Hayden, B. 1990. “Nimrods, Piscators, Pluckers, and Planters: The Emergence of Food Production.” Journal of Anthropological Archaeology 9 (1): 31–69. doi:https://doi.org/10.1016/0278-4165(90)90005-X.
- Hayden, B. 2009. “The Proof Is in the Pudding: Feasting and the Origins of Domestication.” Current Anthropology 50 (5): 597–601. doi:https://doi.org/10.1086/605110.
- Heide, M., and J. Peters. 2021. Camels in the Biblical World. University Park: Pennsylvania State University Press.
- Hillman, G. C., and M. S. Davies. 1990. “Measured Domestication Rates in Wild Wheats and Barley under Primitive Cultivation, and Their Archaeological Implications.” Journal of World Prehistory 4 (2): 157–222. doi:https://doi.org/10.1007/BF00974763.
- Hongo, H., and R. H. Meadow. 1998. “Pig Exploitation at Neolithic Cayonu Tepesi (Southeastern Anatolia).” MASCA Research Papers in Science and Archaeology 15:77–98.
- Hu, Y., S. Hu, W. Wang, X. Wu, F. B. Marshall, X. Chen, L. Hou, and C. Wang. 2014. “Earliest Evidence for Commensal Processes of Cat Domestication.” Proceedings of the National Academy of Sciences of the United States of America 111 (1): 116–120. doi:https://doi.org/10.1073/pnas.1311439110.
- Ibáñez, J. J., P. C. Anderson, J. González-Urquijo, and J. Gibaja. 2016. “Cereal Cultivation and Domestication as Shown by Microtexture Analysis of Sickle Gloss through Confocal Microscopy.” Journal of Archaeological Science 73: 62–81. doi:https://doi.org/10.1016/j.jas.2016.07.011.
- Ingold, T. 1996. “Growing Plants and Raising Animals: An Anthropological Perspective on Domestication.” In The Origins and Spread of Agriculture and Pastoralism in Eurasia, edited by D.R. Harris, 12–24. London: UCL Press.
- Jones, G., T. Kluyver, C. Preece, J. Swarbrick, E. Forster, M. Wallace, M. Charles, M. Rees, and C. P. Osborne. 2021. “The Origins of Agriculture: Intentions and Consequences.” Journal of Archaeological Science 125: 105290. doi:https://doi.org/10.1016/j.jas.2020.105290.
- Kabukcu, C., E. Asouti, N. Pöllath, J. Peters, and N. Karul. 2021. “Pathways to Plant Domestication in Southeast Anatolia Based on New Data from Aceramic Neolithic Gusir Höyük.” Scientific Reports 11 (1): 2112. doi:https://doi.org/10.1038/s41598-021-81757-9.
- Kistler, L., S. Y. Maezumi, J. Gregorio de Souza, N. A. S. Przelomska, F. Malaquias Costa, O. Smith, H. Loiselle, et al. 2018. “Multiproxy Evidence Highlights a Complex Evolutionary Legacy of Maize in South America.” Science 362 (6420): 1309–1313. DOI:https://doi.org/10.1126/science.aav0207.
- Korshunov, V. M. 1994. “Ecology of the Bearded Goat (Capra Aegagrus Ersleben 1777) in Turkmenistan.” In Biogeography and Ecology of Turkmenistan, edited by V. Fet and K. I. Atamuradov, 231–246. Dordrecht, Netherlands: Springer.
- Kruska, D. 1988. “Effects of Domestication on Brain Structure and Behavior in Mammals.” Human Evolution 3 (6): 473–485. doi:https://doi.org/10.1007/BF02436333.
- Küchelmann, H. C. 2019. “Archaeozoological Analysis of Domestic Sheep in the Area between South-West Asia and South-East Europe (7th-2nd Millennium BC)”. online-database, Edition Topoi, Berlin. http://repository.edition-topoi.org/collection/WOLL/object/BONES
- Kuijt, I. 2004. “When the Walls Came Down: Social Organization, Ideology, and the ‘Collapse’of the Pre-Pottery Neolithic.” In Central Settlements in Neolithic Jordan, edited by H. D. Bienert, H. G. K. Gebel, and R. Neef, 183–199. Berlin: ex oriente.
- Kuijt, I. 2008. “The Regeneration of Life: Neolithic Structures of Symbolic Remembering and Forgetting.” Current Anthropology 49 (2): 171–197. doi:https://doi.org/10.1086/526097.
- Kushner, G. 1970. “A Consideration of Some Processual Designs for Archaeology as Anthropology.” American Antiquity 35 (2): 125–132. doi:https://doi.org/10.2307/278141.
- Lahtinen, M., D. Clinnick, K. Mannermaa, J. S. Salonen, and S. Viranta. 2021. “Excess Protein Enabled Dog Domestication during Severe Ice Age Winters.” Scientific Reports 11 (1): 7. doi:https://doi.org/10.1038/s41598-020-78214-4.
- Larson, G., and D. Q. Fuller. 2014. “The Evolution of Animal Domestication.” Annual Review of Ecology, Evolution, and Systematics 45 (1): 115–136. doi:https://doi.org/10.1146/annurev-ecolsys-110512-135813.
- Leipe, C., T. Long, E. A. Sergusheva, M. Wagner, and P. E. Tarasov. 2019. “Discontinuous Spread of Millet Agriculture in Eastern Asia and Prehistoric Population Dynamics.” Science Advances 5 (9): eaax6225. doi:https://doi.org/10.1126/sciadv.aax6225.
- Lewontin, R. C. 1978. “Adaptation.” Scientific American 239 (3): 212–231. doi:https://doi.org/10.1038/scientificamerican0978-212.
- Lewontin, R. C. 1983. “Gene, Organism and Environment.” In Evolution from Molecules to Men, edited by D. S. Bendall, 273–285. Cambridge: Cambridge University Press.
- Lewontin, R. C. 1985. “The Organism as Subject and Object of Evolution.” In The Dialectical Biologist, edited by R. Levins and R. Lewontin, 85–106. Cambridge, MA: Harvard University Press.
- Liang, S., X. Wu, and F. Jin. 2018. “Gut-Brain Psychology: Rethinking Psychology from the Microbiota-Gut-Brain Axis.” Frontiers in Integrative Neuroscience 12: 33.
- Lomolino, M. V. 2001. “Elevation Gradients of Species-Density: Historical and Prospective Views.” Global Ecology and Biogeography: A Journal of Macroecology 10 (1): 3–13. doi:https://doi.org/10.1046/j.1466-822x.2001.00229.x.
- Lord, K. A., G. Larson, and E. K. Karlsson. 2020b. “Brain Size Does Not Rescue Domestication Syndrome.” Trends in Ecology & Evolution 35: 1061-1062.
- Lord, K. A., G. Larson, R. P. Coppinger, and E. K. Karlsson. 2020a. “The History of Farm Foxes Undermines the Animal Domestication Syndrome.” Trends in Ecology & Evolution 35 (2): 125–136. doi:https://doi.org/10.1016/j.tree.2019.10.011.
- Lueck, D., and G. Torrens. 2020. “Property Rights and Domestication.” Journal of Institutional Economics 16 (2): 199–215. doi:https://doi.org/10.1017/S1744137419000390.
- Luo, Y. B.2007. The Origin of the Domestic Pig in China (Zhongguo Gudai Jiazhu Qiyuan Yanjiu). PhD dissertation, Institute of Archaeology, Chinese Academy of Social Science (CASS), Beijing, China. (In Chinese)
- Maeda, O., L. Lucas, F. Silva, K-I. Tanno, and D. Q. Fuller. 2016. “Narrowing the Harvest: Increasing Sickle Investment and the Rise of Domesticated Cereal Agriculture in the Fertile Crescent.” Quaternary Science Reviews 145: 226–237. doi:https://doi.org/10.1016/j.quascirev.2016.05.032.
- Mahlaba, T. A. M., A. Monadjem, R. McCleery, S. R. Belmain, and B L. Allen. 2017. “Domestic Cats and Dogs Create a Landscape of Fear for Pest Rodents around Rural Homesteads.” PloS One 12 (2): e0171593. doi:https://doi.org/10.1371/journal.pone.0171593.
- Makarewicz, C. A., B. Finlayson, and P F. Biehl. 2018. “Constructing Community in the Neolithic of Southern Jordan: Quotidian Practice in Communal Architecture.” PLOS ONE 13 (6): e0193712. doi:https://doi.org/10.1371/journal.pone.0193712.
- Margulis, L. 1991. “Symbiogenesis and Symbionticism.” In Symbiosis as a Source of Evolutionary Innovation: Speciation and Morphogenesis, edited by L. Margulis and R. Fester, 1–14. Cambridge, MA: MIT Press.
- Marshall, F., and L. Weissbrod. 2009. “The Consequences of Women’s Use of Donkeys for Pastoral Flexibility: Maasai Ethnoarchaeology.” Documenta Archaeobiologiae 7: 59–79.
- Marshall, F., and L. Weissbrod. 2011. “Domestication Processes and Morphological Change: Through the Lens of the Donkey and African Pastoralism.” Current Anthropology 52 (S4): S397–S413. doi:https://doi.org/10.1086/658389.
- Marshall, F., R. E. B. Reid, S. Goldstein, M. Storozum, A. Wreschnig, L. Hu, P. Kiura, R. Shahack-Gross, and S. H. Ambrose. 2018. “Ancient Herders Enriched and Restructured African Grasslands.” Nature 561 (7723): 387–390. doi:https://doi.org/10.1038/s41586-018-0456-9.
- Marshall, F. B., K. Dobney, T. Denham, and J. M. Capriles. 2014. “Evaluating the Roles of Directed Breeding and Gene Flow in Animal Domestication.” Proceedings of the National Academy of Sciences of the United States of America 111 (17): 6153–6158. doi:https://doi.org/10.1073/pnas.1312984110.
- Matesanz, S., and R. Milla. 2018. “Differential Plasticity to Water and Nutrients between Crops and Their Wild Progenitors.” Environmental and Experimental Botany 145: 54–63. doi:https://doi.org/10.1016/j.envexpbot.2017.10.014.
- Ménard, L., D. McKey, G. S. Mühlen, B. Clair, N. P. Rowe, and J P. Hart. 2013. “The Evolutionary Fate of Phenotypic Plasticity and Functional Traits under Domestication in Manioc: Changes in Stem Biomechanics and the Appearance of Stem Brittleness.” PLoS ONE 8 (9): e74727. doi:https://doi.org/10.1371/journal.pone.0074727.
- Mueller, N. G., A. White, and P. Szilagyi. 2019. “Experimental Cultivation of Eastern North America’s Lost Crops: Insights into Agricultural Practice and Yield Potential.” Journal of Ethnobiology 39 (4): 549–566. doi:https://doi.org/10.2993/0278-0771-39.4.549.
- Mueller, N. G., R. N. Spengler, A. Glenn, and K. Lama. 2020. “Bison, Anthropogenic Fire, and the Origins of Agriculture in Eastern North America.” The Anthropocene Review, doi:https://doi.org/10.1177/2053019620961119.
- Müller, G. B. 2017. “Why an Extended Evolutionary Synthesis Is Necessary.” Interface Focus. doi:https://doi.org/10.1098/rsfs.2017.0015.
- Neaux, D., B. Blanc, K. Ortiz, Y. Locatelli, R. Schafberg, A. Herrel, V. Debat, and T. Cucchi. 2021. “Constraints Associated with Captivity Alter Craniomandibular Integration in Wild Boar.” Journal of Anatomy 239 (2): 489–497. doi:https://doi.org/10.1111/joa.13425.
- Nicholson, D. J., and R. Gawne. 2015. “Neither Logical Empiricism nor Vitalism, but Organicism: What the Philosophy of Biology Was.” History and Philosophy of the Life Sciences 37 (4): 345–381. doi:https://doi.org/10.1007/s40656-015-0085-7.
- Noy-Meir, I. 1990. “The Effect of Grazing on the Abundance of Wild Wheat, Barley and Oat in Israel.” Biological Conservation 51 (4): 299–310. doi:https://doi.org/10.1016/0006-3207(90)90115-6.
- O’Connor, T. P. 1997. “Working at Relationships: Another Look at Animal Domestication.” Antiquity 71 (271): 149–156. doi:https://doi.org/10.1017/S0003598X00084635.
- Outram, A. K., and A. Bogaard. 2019. Subsistence and Society in Prehistory: New Directions in Economic Archaeology. Cambridge: Cambridge University Press.
- Pérez-Jaramillo, J. E., R. Mendes, and J. M. Raaijmakers. 2016. “Impact of Plant Domestication on Rhizosphere Microbiome Assembly and Functions.” Plant Molecular Biology 90 (6): 635–644. doi:https://doi.org/10.1007/s11103-015-0337-7.
- Peters, J., B. S. Arbuckle, and N. Pöllath. 2015. “Subsistence and Beyond: Animals in Neolithic Anatolia.” The Neolithic in Turkey 6: 135–203.
- Piperno, D. R., I. Holst, J. E. Moreno, and K. Winter. 2019. “Experimenting with Domestication: Understanding Macro- and Micro-Phenotypes and Developmental Plasticity in Teosinte in Its Ancestral Pleistocene and Early Holocene Environments.” Journal of Archaeological Science 108: 104970. doi:https://doi.org/10.1016/j.jas.2019.05.006.
- Piperno, D. R., I. Holst, K. Winter, and O. McMillan. 2015. “Teosinte before Domestication: Experimental Study of Growth and Phenotypic Variability in Late Pleistocene and Early Holocene Environments.” Quaternary International: The Journal of the International Union for Quaternary Research 363: 65–77. doi:https://doi.org/10.1016/j.quaint.2013.12.049.
- Pöllath, N., R. García-González, S. Kevork, U. Mutze, M. I. Zimmermann, M. Özbaşaran, and J. Peters. 2021. “A Non-Linear Prediction Model for Ageing Foetal and Neonatal Sheep Reveals Basic Issues in Early Neolithic Husbandry.” Journal of Archaeological Science 130: 105344. doi:https://doi.org/10.1016/j.jas.2021.105344.
- Pouvreau, D., and M. Drack. 2007. “On the History of Ludwig Von Bertalanffy’s ‘General Systemology’, and on Its Relationship to Cybernetics.” International Journal of General Systems 36 (3): 281–337. doi:https://doi.org/10.1080/03081070601127961.
- Price, M., and H. Hongo. 2020. “The Archaeology of Pig Domestication in Eurasia.” Journal of Archaeological Research 28 (4): 557–615.
- Rindos, D. 1984. The Origins of Agriculture: An Evolutionary Perspective. London: Academic Press.
- Rindos, D., H. Aschmann, P. Bellwood, L. Ceci, M. N. Cohen, J. Hutchinson, R. S. Santley, J. G. Shaffer, and T. Shaw. 1980. “Symbiosis, Instability, and the Origins and Spread of Agriculture: A New Model [And Comments and Reply].” Current Anthropology 21 (6): 751–772. doi:https://doi.org/10.1086/202569.
- Rosshart, S. P., B. G. Vassallo, D. Angeletti, D. S. Hutchinson, A. P. Morgan, K. Takeda, H. D. Hickman, et al. 2017. “Wild Mouse Gut Microbiota Promotes Host Fitness and Improves Disease Resistance.” Cell 171 (5): 1015–1028.e13. DOI:https://doi.org/10.1016/j.cell.2017.09.016.
- Rowley-Conwy, P., and R. Layton. 2011. “Foraging and Farming as Niche Construction: Stable and Unstable Adaptations.” Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences 366 (1566): 849–862. doi:https://doi.org/10.1098/rstb.2010.0307.
- Saña Segui, M. 2000. “Animal Resource Management and the Process of Animal Domestication at Tell Halula (Euphrates Valley-Syria) from 8800 BP to 7800 BP.” In Archaeozoology of the near East IVA, edited by M. Mashkour, A.M. Choyke, H. Buitenhuis, and F. Poplin, 241–256. Groningen: ARC Publication 32.
- Shenton, M., C. Iwamoto, N. Kurata, and K. Ikeo. 2016. “Effect of Wild and Cultivated Rice Genotypes on Rhizosphere Bacterial Community Composition.” Rice 9 (1). doi:https://doi.org/10.1186/s12284-016-0111-8.
- Smith, B. D. 2007. “Niche Construction and the Behavioral Context of Plant and Animal Domestication.” Evolutionary Anthropology: Issues, News, and Reviews 16 (5): 188–199. doi:https://doi.org/10.1002/evan.20135.
- Spengler, R. N., and N. G. Mueller. 2019. “Grazing Animals Drove Domestication of Grain Crops.” Nature Plants 5 (7): 656–662. doi:https://doi.org/10.1038/s41477-019-0470-4.
- Stépanoff, C. 2017. “The Rise of Reindeer Pastoralism in Northern Eurasia: Human and Animal Motivations Entangled.” Journal of the Royal Anthropological Institute 23 (2): 376–396. doi:https://doi.org/10.1111/1467-9655.12612_1.
- Stépanoff, C., and J-D. Vigne. 2018. “Introduction.” In Hybrid Communities, edited by C. Stépanoff and J.-D. Vigne, 1–20. London: Routledge.
- Stevens, C. J., and D. Q. Fuller. 2017. “The Spread of Agriculture in Eastern Asia: Archaeological Bases for Hypothetical Farmer/language Dispersals.” Language Dynamics and Change 7 (2): 152–186. doi:https://doi.org/10.1163/22105832-00702001.
- Stiner, M. C. 2001. “Thirty Years on the ‘Broad Spectrum Revolution’ and Paleolithic Demography.” Proceedings of the National Academy of Sciences of the United States of America 98 (13): 6993–6996. doi:https://doi.org/10.1073/pnas.121176198.
- Stiner, M. C., H. Buitenhuis, G. Duru, S. L. Kuhn, S. M. Mentzer, N. D. Munro, N. Pöllath, J. Quade, G. Tsartsidou, and M. Özbaşaran. 2014. “A Forager-Herder Trade-Off, from Broad-Spectrum Hunting to Sheep Management at Aşıklı Höyük, Turkey.” Proceedings of the National Academy of Sciences of the United States of America 111 (23): 8404–8409. doi:https://doi.org/10.1073/pnas.1322723111.
- Styring, A. K., M. Charles, F. Fantone, M. M. Hald, A. McMahon, R. H. Meadow, G. K. Nicholls, et al. 2017. “Isotope Evidence for Agricultural Extensification Reveals How the World’s First Cities Were Fed.” Nature Plants 3 (6). doi:https://doi.org/10.1038/nplants.2017.76.
- Tanno, K.-I., and G. Willcox. 2006. “How Fast Was Wild Wheat Domesticated?” Science 311 (5769): 1886. doi:https://doi.org/10.1126/science.1124635.
- Trigger, B. G. 2006. A History of Archaeological Thought. Cambridge: Cambridge University Press.
- Trut, L. N. 1999. “Early Canid Domestication: The Farm-Fox Experiment: Foxes Bred for Tamability in a 40-Year Experiment Exhibit Remarkable Transformations that Suggest an Interplay between Behavioral Genetics and Development.” American Scientist 87 (2): 160–169. doi:https://doi.org/10.1511/1999.2.160.
- Twiss, K. C. 2008. “Transformations in an Early Agricultural Society: Feasting in the Southern Levantine Pre-Pottery Neolithic.” Journal of Anthropological Archaeology 27 (4): 418–442. doi:https://doi.org/10.1016/j.jaa.2008.06.002.
- Vigne, J.-D. 2011. “The Origins of Animal Domestication and Husbandry: A Major Change in the History of Humanity and the Biosphere.” Comptes Rendus Biologies 334 (3): 171–181. doi:https://doi.org/10.1016/j.crvi.2010.12.009.
- Vigne, J-D, J. Guilaine, K. Debue, L. Haye, and P. Gérard. 2004. “Early Taming of the Cat in Cyprus.” Science 304 (5668): 259. doi:https://doi.org/10.1126/science.1095335.
- Von Bertalanffy, L. 1941. “Die organismische Auffassung und ihre Auswirkungen.” Der Biologe 10 (247–258): 337–345.
- Von Bertalanffy, L. 1949. “Zu einer allgemeinen Systemlehre.” Biologia Generalis 19: 114–129.
- Von Bertalanffy, L. 1950. “An Outline of General System Theory.” The British Journal for the Philosophy of Science 1 (2): 134–165. doi:https://doi.org/10.1093/bjps/I.2.134.
- Wallace, M., G. Jones, M. Charles, E. Forster, E. Stillman, V. Bonhomme, A. Livarda, et al. 2019. “Re-Analysis of Archaeobotanical Remains from Pre- and Early Agricultural Sites Provides No Evidence for a Narrowing of the Wild Plant Food Spectrum during the Origins of Agriculture in Southwest Asia.” Vegetation History and Archaeobotany 28 (4): 449–463. DOI:https://doi.org/10.1007/s00334-018-0702-y.
- Walsh, D. M. 2018. “Objectcy and Agency: Towards a Methodological Vitalism.” In Everything Flows: Towards a Processual Philosophy of Biology, edited by D.J. Nicholson and J. Dupré. Oxford: Oxford University Press. doi:https://doi.org/10.1093/oso/9780198779636.003.0008.
- Wang, H., L. Martin, W. Wang, 2015. “Morphometric analysis of Sus remains from neolithic sites in the Wei river valley, China, with implications for domestication. ” International Journal of Osteoarcheology 25: 877–889.
- Warinner, C., J. F. Matias Rodrigues, R. Vyas, C. Trachsel, N. Shved, J. Grossmann, A. Radini, et al. 2014. “Pathogens and Host Immunity in the Ancient Human Oral Cavity.” Nature Genetics 46 (4): 336–344. DOI:https://doi.org/10.1038/ng.2906.
- Weide, A., J. G. Hodgson, H. Leschner, G. Dovrat, J. Whitlam, N. Manela, Y. Melamed, Y. Osem, and A. Bogaard. 2021. “The Association of Arable Weeds with Modern Wild Cereal Habitats: Implications for Reconstructing the Origins of Plant Cultivation in the Levant.” Environmental Archaeology.
- Weide, A., S. Riehl, M. Zeidi, N. J. Conard, and R. Bussmann. 2018. “A Systematic Review of Wild Grass Exploitation in Relation to Emerging Cereal Cultivation Throughout the Epipalaeolithic and Aceramic Neolithic of the Fertile Crescent.” PloS One 13 (1): e0189811. doi:https://doi.org/10.1371/journal.pone.0189811.
- Weisskopf, A., L. Qin, J. Ding, P. Ding, G. Sun, and D. Q. Fuller. 2015. “Phytoliths and Rice: From Wet to Dry and Back Again in the Neolithic Lower Yangtze.” Antiquity 89 (347): 1051–1063. doi:https://doi.org/10.15184/aqy.2015.94.
- Whitehead, A. N. 1925. Science and the Modern World. London: Macmillan.
- Whitehead, A. N. 1929. Process and Reality: An Essay in Cosmology. New York: Free Press.
- Winchell, F., M. Brass, A. Manzo, A. Beldados, V. Perna, C. Murphy, C. Stevens, and D. Q. Fuller. 2018. “On the Origins and Dissemination of Domesticated Sorghum and Pearl Millet across Africa and into India: A View from the Butana Group of the Far Eastern Sahel.” African Archaeological Review 35 (4): 483–505. doi:https://doi.org/10.1007/s10437-018-9314-2.
- Woodger, J. H. 1929. Biological Principles: A Critical Study. London: Routledge & Kegan Paul.
- Yeshurun, R., G. Bar-Oz, and M. Weinstein-Evron. 2009. “The Role of Foxes in the Natufian Economy.” Before Farming 2009 (1): 1–15. doi:https://doi.org/10.3828/bfarm.2009.1.3.
- Zeder, M. A. 2006. “Central Questions in the Domestication of Plants and Animals.” Evolutionary Anthropology: Issues, News, and Reviews 15 (3): 105–117. doi:https://doi.org/10.1002/evan.20101.
- Zeder, M. A. 2009. “The Neolithic Macro-(R)evolution: Macroevolutionary Theory and the Study of Culture Change.” Journal of Archaeological Research 17 (1): 1–63.
- Zeder, M. A. 2012a. “The Domestication of Animals.” Journal of Anthropological Research 68 (2): 161–190. doi:https://doi.org/10.3998/jar.0521004.0068.201.
- Zeder, M. A. 2012b. “The Broad Spectrum Revolution at 40: Resource Diversity, Intensification, and an Alternative to Optimal Foraging Explanations.” Journal of Anthropological Archaeology 31 (3): 241–264. doi:https://doi.org/10.1016/j.jaa.2012.03.003.
- Zeder, M. A. 2016. “Domestication as a Model System for Niche Construction Theory.” Evolutionary Ecology 30 (2): 325–348. doi:https://doi.org/10.1007/s10682-015-9801-8.
- Zeder, M. A. 2017. “Domestication as a Model System for the Extended Evolutionary Synthesis.” Interface Focus 7 (5): 20160133. doi:https://doi.org/10.1098/rsfs.2016.0133.
- Zimmermann, M. I., N. Pöllath, M. Özbaşaran, and J. Peters. 2018. “Joint Health in Free-Ranging and Confined Small Bovids - Implications for Early Stage Caprine Management.” Journal of Archaeological Science 92: 13–27. doi:https://doi.org/10.1016/j.jas.2018.02.004.
- Zohary, D., and M. Hopf. 1973. “Domestication of Pulses in the Old World: Legumes Were Companions of Wheat and Barley When Agriculture Began in the near East.” Science 182 (4115): 887–894. doi:https://doi.org/10.1126/science.182.4115.887.
