ABSTRACT
Objectives
Serum ischemia-modified albumin (IMA) is higher in patients with cerebral infarction (CI). In this study, we aimed at studying the association between IMA and CI.
Methods
Patients with CI were divided to severe stenosis group and mild stenosis group according to the stenosis of vertebrobasilar artery. Digital subtraction angiography (DSA) and magnetic resonance imaging (MRI) were used for diagnosis. Cobalt-combined with Albumin test was used to determine the serum IMA levels. Serum IMA levels in patients with CI and healthy volunteers were compared by t test. Receiver operating characteristic (ROC) curve was performed for evaluating the diagnostic efficiency of serum IMA for CI. The correlation between IMA level and the National Institute of Health Stroke Scale(NIHSS) score after treatment was analyzed.
Results
Serum IMA levels in patients with CI was increased compared to healthy volunteers(P < .05). Patients in severe stenosis group had a higher serum IMA level than mild stenosis group(P < .05). Serum IMA level at admission was associated with NIHSS score on the 7th day after treatment(P < .05).
Conclusions
Serum IMA level was correlated with vertebrobasilar artery stenosis and short-term prognosis in patients with acute CI.
Introduction
Cerebral infarction (CI), also called ischemic stroke, is one of the most common cerebrovascular diseases. The pathogenesis of CI results from interrupting of blood circulation in brain such as thromboembolism and embolic occlusion. Many factors, including hypertension, coronary heart disease, diabetes, smoking and vertebrobasilar artery stenosis, are identified as risk factors for CI (Citation1). In a previous study, the incidence of patients with vertebrobasilar artery stenosis is as high as 10.9% (Citation2). Early diagnosis and treatment of CI are vital for prognosis of patients. The diagnosis of CI currently relies heavily on imagological examination such as computed tomography (CT), magnetic resonance imaging (MRI), electrical impedance tomography (EIT) (Citation3–5). However, imagological examination is not sensitive for early signs of CI. Therefore, it is urgent to find new biomarkers for early diagnosis of CI.
Ischemia-modified albumin (IMA) was first found in the 1990s, which was formed by human serum albumin after the production of ischemia and hypoxia-free radicals or acidosis. Studies have showed that IMA increases within minutes of the onset of ischemia, with a peak lasting for 6–12 h (Citation6–8). The association between IMA and CI has been reported in previous studies (Citation9–11). For instance, IMA has been considered to be a marker of early diagnosis for CI and acute coronary syndrome (Citation9). Abboud et al. (Citation10) found that serum IMA levels of patients with CI are higher than those in healthy population, and the IMA levels of patients with CI gradually increases within the first 24 h. In a case-control study, serum IMA levels are also higher in patients with CI, cerebral hemorrhage, and subarachnoid hemorrhage than those in healthy persons, but there is no significant difference between patients with CIand those with cerebral hemorrhage (Citation11). It is illustrated that measurement of IMA in patients with acute ischemic stroke (AIS) is helpful to assess the clinical status of patients and predict the prognosis of patients with AIS (Citation12).
Vertebrobasilar artery stenosis is a major source of ischemic stroke. IMA may be a novel biochemical marker that can reflect acute ischemia. However, the association between IMA and vertebrobasilar artery stenosis has not been reported. The purpose of this study was to investigate the possibility of IMA as a rapid biomarker for risk stratification in patients with acute CI and for screening high-risk patients with CI.
Materials and methods
Patients collection
This study was approved by the Ethics Committee of Hangzhou Third People’s Hospital (IRB# 201511). Informed consent was obtained from all participants. From November 2015 to December 2016, 80 patients with acute CI including 37 (46.25%) males and 43 (53.75%) females with a mean age of 62.23 ± 14.12 years in Hangzhou First People’s Hospital were enrolled in this study. Meanwhile, 80 healthy volunteers including 32 (40%) males and 48 (60%) females with a mean age of 62.41 ± 11.23 years participated in the study. These patients and healthy volunteers were all diagnosed by physical examination, cranial CT and MRI. Clinical data included age, gender, smoking history, drinking history, blood pressure and body mass index (BMI) were collected from patients and healthy controls.
Inclusion criteria: 1) confirmation of acute CI at posterior circulation by MRI; 2) adult patients (≥21 years old); and 3) patients with clinical evidence of acute cerebrovascular disease, including unilateral hemiplegia without clear cause (according to NIHSS criteria), aphasia and unconsciousness, 6 h after symptoms started.
Exclusion criteria: 1) patients with cerebral hemorrhage and subarachnoid hemorrhage by cranial CT; 2) patients with malignant tumor, end-stage renal disease, chronic liver disease and autoimmune disease; 3) patents taking drugs such as warfarin, captopril, furosemide and others, which may have an effect on serum IMA levels; and 4) patients with contraindication of whole cerebral angiography.
Digital subtraction angiography and grouping
Whole brain digital subtraction angiography (DSA) was performed in patients with acute CI according to national standards and the guideline of Chinese Radiology Association. Stenosis rate was evaluated according North American Symptomatic Carotid Endarterectomy Trial Collaborators (NASCET) criteria. Stenosis = (1-The narrowest blood flow width of internal carotid artery)/ Stenosis lesion distal to normal internal carotid artery diameter.
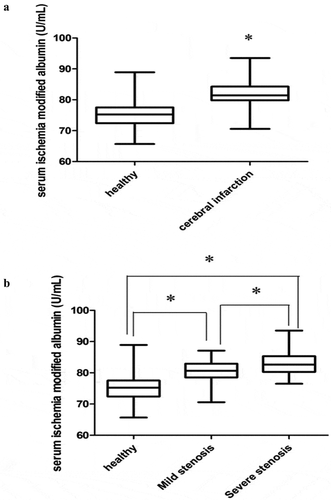
Patients with acute CI were divided into two groups according to the stenosis of vertebrobasilar artery: patients with stenosis of vertebrobasilar artery more than 50% were defined as severe stenosis, and others with stenosis of vertebrobasilar artery less than 50% were defined as mild stenosis.
Detection of serum IMA levels
Peripheral venous blood samples were collected from patients and healthy control into tubes with EDTA, immediately after diagnosed for CI. Samples were centrifuged at 3500 rpm/min for 15 min at room temperature. The plasma samples were extracted and frozen below −80°C for use. A Albumin cobalt assay (ACB) was used to determine the serum IMA levels as described in a previous study (Citation13). ACB Assay for IMA was done colorimetrically as follows: 6200 μL of serum was mixed with 50 μl of CoCl2 (1 g/L) vigorously for 10 min, 50 μL of dithiothreitol (DTT) (1.5 g/L) was added and left for 2 min, and 0.9 N NaCl was then added. Plasma albumin levels were measured in a calibrated and well-controlled autoanalyzer using the bromocresol green method (Architect Plus C8000, Abbott, USA). It was read colorimetrically at 470 nm as ABS U/mL (absorbance units). The conditions of patients after treatment were assessed by National Institutes of Health Stroke Scale (NIHSS). The correlation between serum IMA levels at admission and NIHSS after treatment was also analyzed.
Detection of biochemical indicators
Biochemical indicators were detected by an automatic biochemical analyzer (Architect Plus C8000, Abbott, USA) in clinical laboratory of our hospital, including triglyceride (TG), total cholesterol (TC), high density lipoprotein cholesterol (HDL-C), low density lipoprotein cholesterol (LDL-C).
Statistical analysis
Statistical analyses were performed using Statistical Product and Service Solutions (SPSS) version 19.0 (International Business Machines, corp., Armonk, NY, USA). Patient characteristics were described as frequencies (percentages) or means ± standard deviation (SD). Shapiro-Wilk test was used for normality test. Significant differences between groups were assessed by One-way analysis of variance (ANOVA). Chi-square test was used for counting data. The diagnostic efficiency was calculated by receiver operating characteristic curve (ROC). P value <.05 was defined as a statistically significant difference. Figures were drawn by SPSS Statistics and GraphPad Prism (version 8.0 for Windows, Graph Pad Software, San Diego, CA, USA).
Results
Characteristics of patients with CI
The characteristics of 80 patients with CI and 80 healthy volunteers are shown in . It was indicated that the degree of severe vertebrobasilar artery stenosis was more than 50% (44 of 80 patients), and the degree of mild vertebrobasilar artery stenosis was less than 50% (36 of 80 patients). There were no marked differences between patients with CI and healthy volunteers in variables, including age, gender, BMI, TC, TG, HDL-C and LDL-C (P > .05, respectively).
Table 1. The detailed clinical information of cerebral infarction patients and healthy volunteers (Mean ± SD)
Serum IMA levels
As shown in , serum IMA levels in patients with CI were increased significantly compared to healthy volunteers (81.85 ± 3.69 U/mL vs. 75.07 ± 4.02 U/mL, P = .00). Serum IMA levels in patients with severe or mild stenosis was higher than those in healthy volunteers (P < .05). In addition, serum IMA levels in severe stenosis group was increased compared to mild stenosis group, and there was a significant difference (83.00 ± 3.38 U/mL vs. 80.43 ± 3.55 U/mL, P = .02) ().
ROC curve results
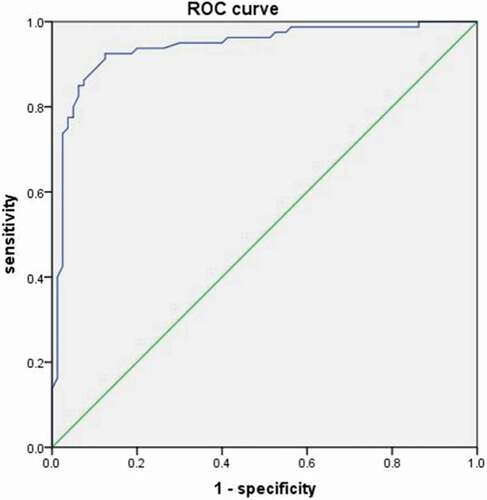
The ROC curve of IMA levels for patients with CI is shown in . The area under curve (AUC) value was 0.94 [95% confidence interval (CI): 0.90–0.98, P = .000]. According to the ROC curve, the cutoff value of serum IMA levels for patients with CI was 78.85 U/mL, with 85% sensitivity and 87.5% specificity.
Correlation between NIHSS score and IMA level
On the 7th day after treatment, all patients with acute CI were assessed by NIHSS score. The results of NIHSS score and serum IMA levels are shown in . Serum IMA levels at admission were associated with NIHSS score on the 7th day after diagnosis (P < .05).
Table 2. Correlation between neurological function score and admission IMA level in patients with acute cerebral infarction treated during 7 days
Discussion
Ischemic stroke is debilitating. Early diagnosis and treatment of ischemic stroke are vital for the prognosis of the patients. It is very important to identify new biomarkers for early diagnosis of ischemic stroke. Many were investigated but none appeared promising. Ischemia-modified albumin (IMA), a modified type of albumin protein, is a candidate biomarker proposed or used for detection of cardiac ischemia (Citation14,Citation15). Many previous studies have demonstrated that serum IMA level is a highly sensitive marker of early acute myocardial infarction (Citation16–18). Recently, IMA has been reported to be uses for diagnosis in other diseases, such as polycystic ovary syndrome (Citation19), acute pancreatitis (Citation20), and acute pancreatitis (Citation21). In the present study, serum IMA levels increased in patients with CI and vertebral basilar artery stenosis, indicating the use of serum IMA levels for diagnosis of CI.
IMA is associated with tissue ischemia, indicating that IMA may also play a role in the differential diagnosis of acute ischemic cerebrovascular disease. It is shown that serum IMA level was 73 U/mL in patient with TIA and 83 U/mL in patient with cerebral infarction within 3 h after the onset (Citation10). In addition, infarct size was reported to be positively correlated with IMA levels (Citation22). A new study suggested that serum IMA significantly increases after transient ischemic attack (TIA) onset, and IMA is significant associated with the secondary CI. Early detection of IMA is of great value in predicting the secondary CI (Citation23). IMA is regarded as “N-terminal modified” albumin which is generated immediately following myocardial ischemia. The detection of IMA is based on reduced cobalt binding affinity to albumin which is attributed mainly to incapability of cobalt to bind at albumin’s modified N-terminus. In this study, Cobalt-combined with Albumin test also found that the serum IMA levels are higher in patients with CI than those in healthy volunteers.
As a previous study reported, serum IMA was affected by many factors, including serum albumin and serum lipid levels (Citation24). It is reported that serum IMA concentrations is positively associated with TC, TG, and LDL-C concentrations (Citation25). In our study, a similar pattern was observed.
There are several possible explanations for the mechanism of elevated serum IMA levels in patients with acute CI within 24 h (Citation26–30). It was found that the surface of the brain cells is rich in unsaturated fatty acids, causing the brain susceptible for ischemia and hypoxia in brain cells (Citation26,Citation27). It has been demonstrated that reactive oxygen species (ROS) can cause an increase in serum IMA levels within minutes (Citation28). With the aggravation of cerebrovascular disease, cerebral tissue ischemia and hypoxia become more serious, leading to the increase and accumulation of lactic acid, which also promote the production of IMA (Citation29,Citation30). Because of ischemia and hypoxia, neuron necrosis induces mitochondrial capacity obstacles and ion pump deficiency, resulting in IMA formation.
In the present study, serum IMA levels differ between severe stenosis and mild stenosis patients, and they were higher in patients with severe stenosis than those in patients with mild stenosis. To the best of our acknowledge, it is the first study reporting that the correlation of serum IMA levels and the stenosis of vertebrobasilar artery. Severe vertebralbasilar artery stenosis increases the incidence of CI. In addition, we found that serum IMA levels in patients with CI in admission are correlated to NIHSS score on the 7th day after diagnosis. It is indicated that serum IMA levels in patients with CI in admission may be a predictive factor for short-term prognosis.
There were some limitations in present study. Firstly, it was a single center observational study, and the sample size of this study was small. But it was the maximum sample size that could be collected. Multi-center large sample prospective study need to be performed to verify the diagnostic values of IMA. Secondly, serum IMA was only detected in admission, and there was no dynamic detection before and after treatment. It is indicated that serum IMA is associated with the severity of CI. In the next study, the real-time changes of IMA in patients with CI need to be investigated.
Conclusions
To sum up, serum IMA level is associated with the stenosis of vertebrobasilar artery, and may be a biomarker for diagnosing CI.
Compliance with Ethical Standards
The study was approved by the Hangzhou First People’s Hospital. Informed consent was obtained.
Author contributions
CYZ is resposible for the guarantor of integrity of the entire study, study concepts, data acquisition, manuscript review; TC is resposible for the study design, experimental studies, manuscript editing; YHS is resposible for the literature research, manuscript preparation; YZ is resposible for the statistical analysis; YL is resposible for the clinical studies; LN is resposible for the definition of intellectual content, data analysis. All authors read and approved the final manuscript.
Acknowledgments
We thank the participants.
Disclosure statement
The authors declare that they have no conflict of interest.
References
- Gjerde G, Naess H. Risk factor burden predicts long-term mortality after cerebral infarction. Acta Neurol Scand. 2014;129:173–77. doi:https://doi.org/10.1111/ane.12159.
- Gondim FA, Cruz-Flores S, Moore J. Angioplasty and stenting for symptomatic basilar artery stenosis. J Neuroimaging. 2002;12:55–58. doi:https://doi.org/10.1111/j.1552-6569.2002.tb00090.x.
- Zhang MJ, Zhang X, Xu YX. Analysis on value of CT and MRI clinical application in diagnosis of middle-aged patients with multiple cerebral infarction. Int J Clin Exp Med. 2015;8(10):17123–27.
- Zhang W, Cheng J, Zhang Y, Wang K, Jin H. Analysis of CT and MRI Combined Examination for the Diagnosis of Acute Cerebral Infarction. J Coll Physicians Surg Pak. 2019;29(9):898–99.doi:https://doi.org/10.29271/jcpsp.2019.09.898.
- Yang B, Shi X, Dai M, Xu C, You F, Fu F, Liu R, Dong X. Real-time imaging of cerebral infarction in rabbits using electrical impedance tomography. J Int Med Res. 2014;42(1):173–83.doi:https://doi.org/10.1177/0300060513499100.
- Myszka W, Wierzchowiecki M, Torliński L, Dziegielewska S. Ischemia modified albumin–a new biochemical marker of myocardial ischemia. Pol Arch Med Wewn. 2005;114(6):1252–59.
- Worster A, Devereaux PJ, Heels-Ansdell D, Guyatt GH, Opie J, Mookadam F, Hill SA. Capability of ischemia-modified albumin to predict serious cardiac outcomes in the short term among patients with potential acute coronary syndrome. CMAJ. 2005;172:1685–90. doi:https://doi.org/10.1503/cmaj.045194.
- Lippi G, Montagnana M, Salvagno GL, Guidi GC. Potential value for new diagnostic markers in the early recognition of acute coronary syndromes. CJEM. 2006;8:27–31. doi:https://doi.org/10.1017/S148180350001335X.
- Sokhanvar S, Mellati AO, Mousavinasab SN, Taran L, Vahdani B, Golmmohamadi Z. Ischemia-modified albumin (IMA) in differential diagnosis of transient myocardial ischemia from non ischemic chest pain. Bratisl Lek Listy. 2012;113:612–15.
- Abboud H, Labreuche J, Meseguer E, Lavallee PC, Simon O, Olivot JM, Mazighi M, Dehoux M, Benessiano J, Steg PG, Amarenco P. Ischemia-modified albumin in acute stroke. Cerebrovasc Dis. 2007;23:216–20. doi:https://doi.org/10.1159/000097644.
- Gunduz A, Turedi S, Mentese A, Altunayoglu V, Turan I, Karahan SC, Topbas M, Aydin M, Eraydin I, Akcan B. Ischemia-modified albumin levels in cerebrovascular accidents. Am J Emerg Med. 2008;26:874–78. doi:https://doi.org/10.1016/j.ajem.2007.11.023.
- Nayak AR, Kashyap RS, Kabra D, Purohit HJ, Taori GM, Daginawala HF. Prognostic significance of ischemia-modified albumin in acute ischemic stroke patients: a preliminary study. Ann Neurosci. 2011;18:5–7. doi:https://doi.org/10.5214/ans.0972.7531.1118103.
- Bar-Or D, Curtis G, Rao N, Bampos N, Lau E. Characterization of the Co(2+) and Ni(2+) binding amino-acid residues of the N-terminus of human albumin. An insight into the mechanism of a new assay for myocardial ischemia. Eur J Biochem. 2001;268(1):42–47.doi:https://doi.org/10.1046/j.1432-1327.2001.01846.x.
- Bhakthavatsala Reddy C, Cyriac C, Desle HB. Role of “Ischemia Modified Albumin” (IMA) in acute coronary syndromes. Indian Heart J. 2014;66:656–62. doi:https://doi.org/10.1016/j.ihj.2014.12.005.
- Mishra B, Pandey S, Niraula SR, Rai BK, Karki P, Baral N, Lamsal M. Utility of Ischemia Modified Albumin as an Early Marker for Diagnosis of Acute Coronary Syndrome. J Nepal Health Res Counc. 2018;16:16–21. doi:https://doi.org/10.3126/jnhrc.v16i1.19383.
- Christenson RH, Duh SH, Sanhai WR, Wu AH, Holtman V, Painter P, Branham E, Apple FS, Murakami M, Morris DL. Characteristics of an Albumin Cobalt Binding Test for assessment of acute coronary syndrome patients: a multicenter study. Clin Chem. 2001;47:464–70. doi:https://doi.org/10.1093/clinchem/47.3.464.
- Sinha MK, Roy D, Gaze DC, Collinson PO, Kaski JC. Role of “Ischemia modified albumin,” a new biochemical marker of myocardial ischaemia, in the early diagnosis of acute coronary syndromes. Emerg Med J. 2004;21:29–34. doi:https://doi.org/10.1136/emj.2003.006007.
- Roy D, Quiles J, Aldama G, Sinha M, Avanzas P, Arroyo-Espliguero R, Gaze D, Collinson P, Carlos Kaski J. Ischemia Modified Albumin for the assessment of patients presenting to the emergency department with acute chest pain but normal or non-diagnostic 12-lead electrocardiograms and negative cardiac troponin T. Int J Cardiol. 2004;97:297–301. doi:https://doi.org/10.1016/j.ijcard.2004.05.042.
- Seshadri Reddy V, Bukke S, Munikumar M. Elevated levels of the circulatory ischemia-modified albumin in patients with polycystic ovary syndrome: a meta-analysis. Gynecol Endocrinol. 2018;34:868–74. doi:https://doi.org/10.1080/09513590.2018.1462318.
- Sahin A, Turkoglu S, Tunc N, Duzenci D, Solmaz OA, Bahcecioglu IH, Yalniz M. Is ischemia-modified albumin a reliable tool for the assessment of acute pancreatitis? Ther Clin Risk Manag. 2018;14:627–35. doi:https://doi.org/10.2147/TCRM.S162690.
- Seshadri Reddy V, Munikumar M, Duggina P, Varma N. A diagnostic test accuracy meta-analysis of maternal serum ischemia-modified albumin for detection of preeclampsia. J Matern Fetal Neonatal Med. 2019;32:2173–81. doi:https://doi.org/10.1080/14767058.2018.1427724.
- Can S, Akdur O, Yildirim A, Adam G, Cakir DU, Karaman HI. Myelin basic protein and ischemia modified albumin levels in acute ischemic stroke cases. Pak J Med Sci. 2015;31:1110–14. doi:https://doi.org/10.12669/pjms.315.7702.
- Ding LD, Xiao ZH, Mao HW, Hang XF, Guo JH, Ke KF, Chen J. 2021. Ischemia Modified Albumin and miR-126 Play Important Role in Diagnosis of Posterior Circulation Transient Ischemic Attack and Prediction of Secondary Cerebral Infarction. Neurol India. 69(1):75–80. doi:https://doi.org/10.4103/0028-3886.310100
- Roy D, Quiles J, Sharma R, Sinha M, Avanzas P, Gaze D, Kaski JC. Ischemia-modified albumin concentrations in patients with peripheral vascular disease and exercise-induced skeletal muscle ischemia. Clin Chem. 2004;50:1656–60. doi:https://doi.org/10.1373/clinchem.2004.031690.
- Han K, Jia N, Yang L, Min LQ. Correlation between ischemia-modified albumin and lipid levels in patients with acute cerebrovascular disease. Mol Med Rep. 2012;6:621–24. doi:https://doi.org/10.3892/mmr.2012.961.
- Wu AH, Morris DL, Fletcher DR, Apple FS, Christenson RH, Painter PC. Analysis of the Albumin Cobalt Binding (ACB) test as an adjunct to cardiac troponin I for the early detection of acute myocardial infarction. Cardiovasc Toxicol. 2001;1:147–51. doi:https://doi.org/10.1385/CT:1:2:147.
- Ulugol H, Aksu U, Kocyigit M, Kilercik M, Karduz G, Okten M, Toraman F. Comparative Effects of Blood and Crystalloid Cardioplegia on Cellular Injury and Oxidative Stress in Cardiovascular Surgery. Ann Thorac Cardiovasc Surg. 2019;25:10–17. doi:https://doi.org/10.5761/atcs.oa.18-00113.
- Roy D, Quiles J, Gaze DC, Collinson P, Kaski JC, Baxter GF. Role of reactive oxygen species on the formation of the novel diagnostic marker ischaemia modified albumin. Heart. 2006;92:113–14. doi:https://doi.org/10.1136/hrt.2004.049643.
- Cichota LC, Moresco RN, Duarte MM, Da Silva JE. Evaluation of ischemia-modified albumin in anemia associated to chronic kidney disease. J Clin Lab Anal. 2008;22:1–5. doi:https://doi.org/10.1002/jcla.20226.
- Yazıcı MU, Ayar G, Savas-Erdeve S, Azapağası E, Neşelioğlu S, Ö E, Çetinkaya S. Role of Ischemia Modified Albumin Serum Levels as an Oxidative Stress Marker in Children with Diabetic Ketoacidosis. Comb Chem High Throughput Screen. 2019;22(8):577–81.doi:https://doi.org/10.2174/1386207322666191008214919.