Introduction
In cataract surgery, the creation of the anterior capsulorhexis as one of the critical steps is of most importance for the surgical success. In challenging initial situations (e.g. in eyes with pseudoexfoliation syndrome, mature, brunescent cataract, juvenile cataract, corneal opacities, or in post-traumatic and postuveitic cases), vital dyes are used as a routine to increase the visibility and plasticity of the ocular structures in the anterior chamber. The range of applications of the vital dye trypan blue (TB) has expanded considerably in recent years due to its excellent staining properties.Citation1 However, its use in ophthalmology as an effective and useful tool requires that the dye has no adverse effects on the cell structures of the eye. As two laboratory studies on cell cultures showed, the time of exposure to TB plays an important role in addition to concentration.Citation2,Citation3 The in vitro studies by Chang et al. with rabbit corneal endothelial cell cultures, and van Dooren et al.Citation3 demonstrated no toxicity of TB with a maximum concentration of 0.4% after 1 minute in cell cultures of human corneal fibroplasts. A significant toxicity of a TB concentration of 0.01% or higher after exposure was observed. At 24-hour exposure, a TB concentration of 0.005% was found to be the threshold for a significant cytotoxicity index. In principle, it is important to note that trypan blue can be cytotoxic at a certain concentration. Monoazo, the most toxic of known impurities found in trypan blue dyes can be carcinogenic. However, the TB concentrations used in eye surgery do not have undesirable effects on the cell structures of the eye and are therefore generally considered safe.Citation3,Citation4
However, a case report showed a transient retinal toxic reaction in the form of transient visual field defect following the entry of TB into the vitreous body space.Citation5
In modern intraocular procedures viscoelastic substances (OVDs, ophthalmic viscoelastic devices) are widely used. Since their introduction in the 1970s, they have been routinely used in cataract surgery and serve to protect sensitive eye structures from mechanical injuries or to create and maintain anatomical spaces such as the anterior chamber or the capsular bag. They increase safety during the procedure and can also shorten overall surgery time by improving visibility and simplifying some surgical steps for the surgeon.Citation6
A shorter surgery time is associated with a lower degree of trauma and a lower risk of complications, and may ultimately be associated with faster recovery and a better final outcome and satisfaction for the patient. In addition, from an economic point of view, the time saving factor is particularly important for high-volume facilities. After the introduction of Healon® in 1979, sodium hyaluronate became the most widely used biopolymer for OVDs in intraocular surgery. Since then, the pharmacological, physiological, and clinical aspects of sodium hyaluronate for ophthalmic applications have been assessed in a large number of studies.Citation7,Citation8
Recently, a combination of a viscoelastic with the vital dye TB has been introduced (Pe-Ha-Blue®PLUS, Albomed, Schwarzenbruck, Germany) and has already been clinically investigated in a prospective case series of 52 eyes with pseudoexfoliation syndrome.Citation6 In addition to a significantly shorter surgery time (due to fewer individual surgical steps) with cost- and safety-relevant advantages of Pe-Ha-Blue®PLUS compared to separate administration of OVD (POLY-HYL® 1. 6%; Polytech Domilens GmbH) and TB (Vision Blue®; DORC, Holland/Blue Color Caps®), the surgeon gains better control over whether the OVD is removed completely at the end of surgery by using the blue OVD. This should also reduce postoperative complications such as hypertension due to OVD residues remaining in the eye. The aim of the present in vitro study was to determine by Raman spectroscopy the amount of residue of the TB dye that remains on a slide during the routine application of two commercial products (TB dye Vision Blue® and Pe-Ha-Blue®PLUS). In Raman spectroscopy, the interaction of light and matter is used to investigate, for example, the properties of a material or to enable the microscopic examination of materials. Excited by monochromatic light, the sample emits scattered light with a specific frequency shift. The frequency shift (the so-called Raman shift) contain information about the vibrational states of the molecules and thus about the chemical composition and structure of the sample. This phenomenon was discovered by Sir C. V. Raman in the early 20th century. Since the beginning of the 20th century, the method, today mostly stimulated by a laser light source, is widely used in fields such as industry, chemistry, archaeology or for the qualitative and quantitative analysis of products in the pharmaceutical industry.
Material and methods
Raman microscopy
Raman spectroscopy is based on the inelastic scattering of light to matter, the so-called Raman scattering. Unlike elastic scattering, in which the photon scattering occurs mostly without energy loss (Rayleigh scattering), a very small part of the photons is scattered at a different frequency (Raman scattering). All Raman measurements in our evaluation were performed with the LabRam 800 HR spectrometer (Horiba Jobin Yvon GmbH, Bensheim, Germany) equipped with a 1024 × 256 CCD camera (Peltier-cooled) adapted to an Olympus BX41 microscope. All measurements were carried out with a laser wavelength of 532 nm (5 mW), an integration time of 1 s per sweep and an Olympus x10 MPlanN (NA = 0. 25) lens. Each spectral image that scans pixel by pixel and has a full spectrum per pixel has a total size of 7200 × 6800 μm with a step width of 50 μm and uses the DuoScanTM imaging system to map the laser focus to the pixel size.
Product information
Pe-Ha-Blue®PLUS (Albomed, Germany) is a combination of biofermented sodium hyaluronate (1.7%) and trypan blue (0.020 mg/ml). The CE approved product is available pre-mixed in a disposable syringe (containing 1. 0 ml) and allows the administration of OVD and TB in one step. VisionBlue® (DORC, The Netherlands) is available as a 0.06% trypane blue dye in a disposable syringe (contents 2.25 ml) and has FDA approval for the anterior segment.
Experimental set-up
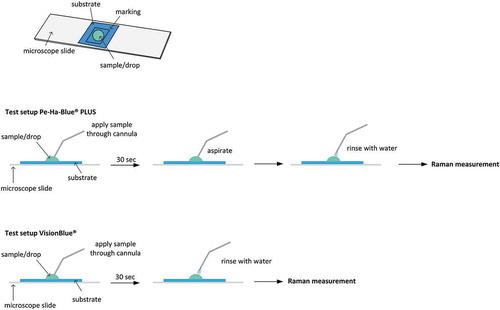
The upper part of shows the prepared samples for both investigated substances. A drop of Vision Blue and Pe-Ha-Blue®PLUS were applied within a marking on a slide. Two samples were taken for each product: a reference sample, which creates a significantly contaminated area by drying the substances, and a test sample. Similar to the procedure in clinical practice, the test sample of the Vision Blue Group was rinsed with water for a maximum of 30 seconds after application of the substances. Five milliliter of water from an injection needle was used at even pressure and from the same distance. In the end, no residues were visible (). In the Pe-Ha-Blue®PLUS group, the substance was first aspirated after the maximum residing time had expired and then rinsed in exactly the same way. The prepared samples of both substances were then imaged with the Raman spectroscope. To demonstrate reproducibility, the entire test procedure was repeated 3 times.
Results
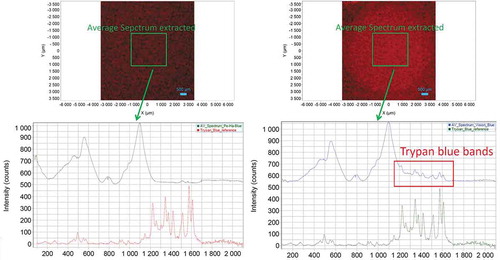
shows the results of spectroscopy measurement of the reference samples of both substances. Clear TB residues can be seen in both the light microscopic image and the false color image. The false color representation was created by integrating over the band typical of TB () from 1547 to 1589 cm−1. This band was chosen because the substrate does not show any Raman signal in this region. (No residue of Pe-Ha-Blue®PLUS is visible on the rinsed samples after mapping; a drop-like residue is visible when mapping Vision Blue®) (). The signal-to-noise ratio in the individual spectra of the mappingin particular, the Vision Blue® test sample, was found to be insufficient for the identification of TB. Therefore, the spectra of the individual pixels collected in the center of the drop were averaged over an area of 2 × 2 mm to achieve an improvement in the signal-to-noise ratio (). The Raman bands of TB are clearly visible in the mean spectrum of the Vision Blue® test specimen at a Raman shift of 1200–1600 cm−1, indicating residues on the test specimen. The test sample of Pe-Ha-Blue®PLUS does not show any Raman bands in the typical TB Raman Shift.
Figure 2. Light microscopy images of the reference samples for Pe-Ha-Blue®PLUS (top left) and Vision Blue® (top right) with marked mapping area of the Raman measurement (blue line). The false-color plots show the distribution of TB as an integral over the Raman band 1547–1589 cm−1 for the Pe-Ha-Blue®PLUS reference sample (bottom left) and the Vision Blue® reference sample (bottom right)
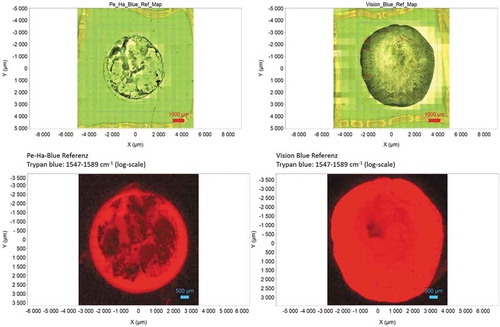
Figure 3. The light microscopic images of the test samples of Pe-Ha-Blue®PLUS (top left) and Vision Blue (bottom right) show no visible TB residues within the mapping area marked (blue line). The false color display does not show any TB residues for Pe-Ha-Blue®PLUS (left bottom), the Vision Blue test sample (right bottom) shows a light, drop-like area from the darker edge area
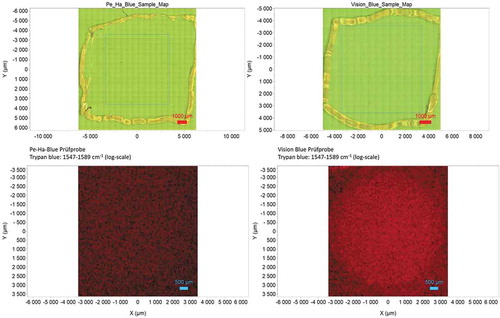
Figure 4. Extracted average spectrum over an area of 2 × 2 mm from the test samples of Pe-Ha-Blue®PLUS (top left) and Vision Blue® (top right). The mean spectrum of the Pe-Ha-Blue®PLUS test specimen (lower left) does not show any specific TB Raman bands; in the Vision Blue® test specimen the Raman bands are clearly visible in the TB-relevant range between 1200 and 1600 cm−1 (lower right)
Discussion
In cases of compromised fundus reflex due to corneal opacities or very advanced lens opacities (cataracta provecta, brunescens, matura) as well as in difficult, challenging situations (e.g. Pseudoexfoliation syndrome, uveitis, post-traumatic cases with phacodonesis and zonule weakness), dye-assisted surgery is preferred to increase the safety of the procedure and achieve a better workflow. The current technique of dyeing is based on a multi-stage process. To stain the anterior lens capsule, the dye (TB) is introduced into the anterior chamber (AC) in a first step. Usually, the surgeon injects a small amount of air into the anterior chamber. This air bubble is used to protect the endothelium from the dye. In a further step, the dye (TB) is removed from the VK by rinsing. The final step is to fill the anterior chamber with a viscoelastic agent (OVD). Good mixing and homogeneous concentration of OVD and dye in the eye or on the scene cannot be controlled in this way. Methods have been described in the past to mix TB and viscoelastic substances before introducing them into the anterior chamber, for example, to limit the contact of the vital dye to the relevant ocular structures. Kayikiçioğlu et al.Citation9 used a combination of TB with sodium hyaluronate to increase the visibility of the anterior lens capsule. In vitreoretinal surgery (peeling), a similar technique using indocyanine green to stain the inner limiting membrane (ILM) has also been described as safe and effectiveCitation10 However, the methods described carry the risk of contamination.
TB is a widely used vital dye whose efficacy in terms of staining properties and cytotoxicity on various structures of the anterior segment of the eye has been investigated in several in vitro studiesCitation2,Citation3,Citation11,Citation12and clinical trialsCitation4,Citation13–16 .TB showed no cytotoxic effects in clinical studies, but in vitro studies proved that TB is toxic to cultured human corneal endothelial cells,Citation11 cultured rabbit corneal endothelial cellsCitation2 or HeLa cells in a concentration- and/or time-dependent manner.Citation12 Basically, the use of TB requires a rapid rinsing of stained areas and a complete removal from the eye, as the critical concentration might be exceeded due to the weak miscibility in the eye. In our opinion, the advantage of the investigated Pe-Ha-Blue®PLUS OVD is that the vital dye TB is present in a much lower concentration and bound in the matrix of a cohesive viscoelastic due to its mixing in the OVD and thus more even distribution in the eye. In the conventional, two-step method of injecting TB into the anterior chamber, a substantially higher dose must be applied to ensure adequate staining of intraocular structures in more remote areas. Cohesive viscoelastic products also have the property of leaving the eye as a whole (bolus) during aspiration. It was shown with our experimental setup based on Raman spectroscopy in the present in vitro study, that the incorporation of TB into such an OVD at the same time as the viscoelastic also enables an easier removal of the dye. This fact reduces the risk of toxic effects and a postoperative pressure rise. In the past, some studies found an effect of ophthalmic dyes on the mechanical properties like elasticity and stiffness of the anterior lens capsule.Citation17,Citation18 In another large clinical evaluation, capsule thickness was positively correlated with increasing age in all groups and the anterior lens capsules of patients with pseudoexfoliation syndrome had more elasticity and less stiffness than the other groups. However, intracameral trypan blue application had no effect on capsule elasticity and stiffness.Citation19 Therefore, studies have not generated a uniform result by now.
The use of a stained OVD may reduce the likelihood of residues of the dye remaining or being overlooked in hard-to-reach regions of the eye (iridocorneal angle). In addition, OVD remaining in the lens capsule or anterior chamber may cause complications postoperatively. If the drainage of the aqueous humor via the trabecular meshwork is reduced by residues of OVD, there, is a higher risk of a postoperative increase in intraocular pressure.Citation20
Further benefits are to be expected with the implantation of toric intraocular lenses (IOLs), since incomplete OVD suction increases the risk of postoperative IOL rotations. It may act as a sliding film between IOL rear surface and posterior capsule), especially for IOLs with large diameter and/or plate-haptic design.Citation21 Furthermore, it has been discussed in the past in various studies whether remaining OVD behind the IOL could act as channels for lens epithelial cell growth (LEC) and thus for faster posterior capsule opacification (PCO) development.Citation22 In addition, further advantages, such as an intraoperative increase in contrast (when using a suitable microscopy light source) and improved control of the amount of OVD injected, could be observed when using Pe-Ha-Blue®PLUS. A so-called “absorption effect” of the blue-stained OVD during larger surgery time may additionally protect the macula from potentially hazardous ultraviolet and blue light of the surgery microscope. Further experimental and clinical study are necessary to proof this effect.
In addition to the effectiveness of the surgical workflow, Pe-Ha-Blue®PLUS may increase the safety of dye-supported eye surgeries. The safe and complete removal of a potentially toxic and carcinogenic substance from the eye is essential. As the concentration of TB in Pe-Ha-Blue®PLUS is lower and the dye is bound in the matrix of the OVD, there is a certain additional safety aspect. Further studies regarding a possible protective effect on the macula as well as investigation on the protective effect for toric lens rotation or refractive stability and postoperative intraocular pressure behavior are necessary to address these issues. Clinical results have shown in a previous study on additional safety benefits in a variety of surgical situations.Citation6 Although this in vitro study is an experimental setup, the findings may support clinical observations. For example, it has been observed in everyday clinical practice that blue OVD (PeHaBlue Plus) can be removed from the anterior chamber more easily and faster using iris hooks or Malyugin rings. The same arrangement with the trypan blue (standard) results in some bluish remnants (like clouds or swaths) on the ring.
In this in-silico experiment we ignored the effect of dispersion of TB in the entire anterior chamber. In fact, in a clinical setting we often see that the TB injected into the anterior chamber does not show a homogeneous distribution, and therefore we feel that our experiment is at least describing how TB which is not in compound with the viscoelastics is washed-out with BSS. The benefit of such an experiment is, that the results are highly repeatable. With experiments in vitro or in vivo the repeatability will be much lower. We feel that Raman spectroscopy is a very powerful tool for measuring the washout effect of TB in this experiment. The measurement technique is well established and offers a good performance, and it allows for a good performance in identification of remnants of TB on our glass plate. Of course, further studies will have to follow to confirm the results.
Conclusion
The results of our in-silico experiment show that a drop of a combination of a vital dye with a viscoelastic could be washed-out more easily with BSS compared to a dye itself without viscoelastics. This suggests that at least in an in silico experiment the removal of both, the viscoelastics and the TB is easier compared to the removal of the TB alone. Further, in-vitro experiment in pig cadaver eyes are planned to prove this washout effect. If this concept can be demonstrated, this could further increase the safety of dye-assisted ophthalmic procedures.
Disclosure of potential conflicts of interest
All authors declare that they do not have any financial interest in products mentioned.
Additional information
Funding
References
- Jhanji V, Chan E, Das S, Zhang H, Vajpayee RB. Trypan blue dye for anterior segment surgeries. Eye (Lond). 2011;25(9):1113–20. doi:https://doi.org/10.1038/eye.2011.139.
- Chang YS, Tseng SY, Tseng SH, Chen YT, Hsiao JH. Comparison of dyes for cataract surgery. Part 1: cytotoxicity to corneal endothelial cells in a rabbit model. J Cataract Refract Surg. 2005;31(4):792–98. doi:https://doi.org/10.1016/j.jcrs.2004.07.020.
- van Dooren BT, Beekhuis WH, Pels E. Biocompatibility of trypan blue with human corneal cells. Arch Ophthalmol. 2004;122:736–42. doi:https://doi.org/10.1001/archopht.122.5.736.
- van Dooren BT, de Waard PW, Poort-van Nouhuys H, Beekhuis WH, Melles GR. Corneal endothelial cell density after trypan blue capsule staining in cataract surgery. J Cataract Refract Surg. 2002;28(4):574–75. doi:https://doi.org/10.1016/s0886-3350(02)01315-9.
- Bacsal KM, Chee SP. Trypan blue-associated retinal toxicity post complicated cataract surgery. Eye (Lond). 2006;20:1310–11. doi:https://doi.org/10.1038/sj.eye.6702164.
- Borkenstein AF, Borkenstein EM. Evaluation of a Novel Blue-Colored Ophthalmic Viscoelastic Device Applied during Phacoemulsification in Eyes with Pseudoexfoliation Syndrome. Case Rep Ophthalmol. 2019;10:101–09. doi:https://doi.org/10.1159/000499339.
- Goa KL, Benfield P. Hyaluronic acid. A review of its pharmacology and use as a surgical aid in ophthalmology, and its therapeutic potential in joint disease and wound healing. Drugs. 1994;47:536–66. doi:https://doi.org/10.2165/00003495-199447030-00009.
- Liesegang TJ. Viscoelastic substances in ophthalmology. Surv Ophthalmol. 1990;34:268–93. doi:https://doi.org/10.1016/0039-6257(90)90027-s.
- Kayikicioglu O, Erakgun T, Guler C. Trypan blue mixed with sodium hyaluronate for capsulorhexis. J Cataract Refract Surg. 2001;27:970. doi:https://doi.org/10.1016/s0886-3350(01)01011-2.
- Kadonosono K, Itoh N, Uchio E, Nakamura S, Ohno S. Staining of internal limiting membrane in macular hole surgery. Arch Ophthalmol. 2000;118:1116–18. doi:https://doi.org/10.1001/archopht.118.8.1116.
- Thaler S, Hofmann J, Bartz-Schmidt KU, Schuettauf F, Haritoglou C, Yoeruek E. Methyl blue and aniline blue versus patent blue and trypan blue as vital dyes in cataract surgery: capsule staining properties and cytotoxicity to human cultured corneal endothelial cells. J Cataract Refract Surg. 2011;37(6):1147–53. doi:https://doi.org/10.1016/j.jcrs.2010.12.051.
- Wilińska J, Mocanu B, Awad D, Gousia D, Hillner C, Brannath W, Mohr A, Gabel D. New stains for anterior capsule surgery. J Cataract Refract Surg. 2019;45(2):213–18. doi:https://doi.org/10.1016/j.jcrs.2018.09.016.
- Dada VK, Sharma N, Sudan R, Sethi H, Dada T, Pangtey MS. Anterior capsule staining for capsulorhexis in cases of white cataract: comparative clinical study. J Cataract Refract Surg. 2004;30(2):326–33. doi:https://doi.org/10.1016/S0886-3350(03)00573-X.
- Jacob S, Agarwal A, Agarwal A, Agarwal S, Chowdhary S, Chowdhary R, Bagmar AA. Trypan blue as an adjunct for safe phacoemulsification in eyes with white cataract. J Cataract Refract Surg. 2002;28(10):1819–25. doi:https://doi.org/10.1016/s0886-3350(01)01316-5.
- Melles GR, de Waard PW, Pameijer JH, Beekhuis WH. Staining the lens capsule with trypan blue for visualizing capsulorhexis in surgery of mature cataracts. Klin Monbl Augenheilkd. 1999;215(6):342–44. doi:https://doi.org/10.1055/s-2008-1034729.
- Kothari K, Jain SS, Shah NJ. Anterior capsular staining with trypan blue for capsulorhexis in mature and hypermature cataracts. A preliminary study. Indian J Ophthalmol. 2001;49:177–80.
- Dick HB, Aliyeva SE, Hengerer F. Effect of trypan blue on the elasticity of the human anterior lens capsule. J Cataract Refract Surg. 2008;34(8):1367–73. doi:https://doi.org/10.1016/j.jcrs.2008.03.041.
- Jardeleza MS, Daly MK, Kaufman JD, Klapperich C, Legutko PA. Effect of trypan blue staining on the elastic modulus of anterior lens capsules of diabetic and nondiabetic patients. J Cataract Refract Surg. 2009;35(2):318–23. doi:https://doi.org/10.1016/j.jcrs.2008.10.040.
- Simsek C, Oto S, Yilmaz G, Altinors DD, Akman A, Gungor SG. Comparison of the mechanical properties of the anterior lens capsule in senile cataract, senile cataract with trypan blue application, and pseudoexfoliation syndrome. J Cataract Refract Surg. 2017;43(8):1054–61. doi:https://doi.org/10.1016/j.jcrs.2017.05.038.
- Grzybowski A, Kanclerz P. Early postoperative intraocular pressure elevation following cataract surgery. Curr Opin Ophthalmol. 2019;30:56–62. doi:https://doi.org/10.1097/ICU.0000000000000545.
- Li S, Li X, He S, Zheng Q, Chen X, Wu X, Xu W. Early Postoperative Rotational stability and its related factors of a single-piece acrylic toric intraocular lens. Eye (Lond). 2020;34(3):474–79. doi:https://doi.org/10.1038/s41433-019-0521-0.
- Chandler HL, Haeussler DJ Jr, Gemensky-Metzler AJ, Wilkie DA, Lutz EA. Induction of posterior capsule opacification by hyaluronic acid in an ex vivo model. Invest Ophthalmol Vis Sci. 2012 6;53(4):1835–45. doi:https://doi.org/10.1167/iovs.11-8735.