Abstract
Objective
There are few studies on fractional exhaled nitric oxide (FeNO) among children in subtropical areas. We studied associations between FeNO and respiratory symptoms, reported diagnosed allergies and indoor and outdoor environmental factors in first grade junior high school students (N = 270) in upper northern Thailand.
Methods
Data on demographics, health and home environment were collected by a questionnaire distributed in dry season (February–March 2018). FeNO was measured when the research team visited the school. Daily outdoor pollution data (PM10 and ozone) were collected from the nearest monitoring station 3 days (lag 3) and 7 days (lag 7) before the FeNO measurements. Two-level (student, school) linear mixed models were used to analyze associations, adjusting for gender and family education level.
Results
In total, 29.6% had elevated FeNO level (>20 ppb) and 7.8% reported any allergy diagnosed by a doctor. Male gender (p = 0.02), diagnosed allergy (p = 0.001), especially to cat (p = 0.001) and house dust mite (HDM) allergies (p = 0.001) were associated with FeNO. Eye symptoms (p = 0.01), rhinitis symptoms (p = 0.03) and dyspnea (p = 0.05) in the last 3 days were associated with FeNO. Household indoor mold (p = 0.03), gas cooking (p = 0.03) and PM10 (lag 3 and lag 7) were negatively (protective) associated with FeNO.
Conclusions
Diagnosed allergy, especially to cat and HDM, can be associated with increased FeNO. Indoor mold and gas cooking can be associated with lower FeNO. Ocular, nasal and dyspnea symptoms reported by students in dry season in northern Thailand can be associated with FeNO, a biomarker of Th2 driven airway inflammation.
Introduction
Air pollution contributes to the global burden of chronic respiratory disease (Citation1) and asthma and allergies are on the rise in Asia (Citation2). In Thailand, a recent study from Bangkok reported that 13.5% of the children (13–14 y) had wheeze and 43.6% had rhinitis (Citation3). Fractional exhaled nitric oxide (FeNO) is a noninvasive biomarker detecting T-helper cell type 2-driven airway inflammation (Citation4). One review identified 27 panel studies on associations between outdoor air pollution and FeNO. Short-term PM10, PM2.5, NO2 and SO2 exposure increased FeNO levels (Citation5). However, all Asian panel studies in this review, except one Japanese study, were from China.
Seasonal haze episodes from biomass burning in South East Asia cause respiratory illnesses (Citation6) and household biomass burning is still common in parts of Asia (Citation1). One panel study from USA found that increased FeNO was associated with personal PM2.5 exposure, but not with ambient PM2.5 or wood smoke combustion biomarkers (Citation7). One study from Honduras found inconsistent associations between household biomass burning and FeNO (Citation8). In upper northern Thailand, farmers burn organic waste materials in the end of the dry season (February-March). This habit causes high outdoor particle pollution of PM10 and PM2.5 (Citation9–11). Moreover, rural families in this area still use biomass for cooking. We found no study from Asia on associations between indoor or outdoor biomass burning and FeNO.
Household air pollution (HAP) can impair respiratory health (Citation12). One study from Sweden reported that window pane condensation, an indicator of high humidity and poor ventilation, increased FeNO in school children (Citation13). Moreover, household paraffin use (Citation14), cooking by liquefied petroleum gas (Citation15) and heating by electric radiators (electric baseboard heating) can increase FeNO (Citation16). Bioaerosol exposure can reduce FeNO, including exposure to endotoxin (Citation17), the gram positive bacteria Streptomyces sp. (Citation18) and dog keeping (Citation16,Citation17). In contrast, exposure to Dermatophagoides pteronyssinus (Der p 1) exposure can increase FeNO (Citation19,Citation20).
We found no previous studies on environmental risk factors for FeNO in Thai school children. Therefore we have performed a study among junior high school students in upper Northern Thailand in dry season, when the farmers burn biomass. The main aim was to study associations between FeNO and HAP and outdoor air pollution (PM10 and ozone). Moreover, we studied associations between FeNO and reported diagnosed allergy and medical symptoms related to asthma and rhinitis. The following hypotheses were tested: FeNO is associated with male gender, family education level, reported doctor diagnosed allergies, current ocular, nasal and respiratory symptoms. Moreover we tested that home environment factors and outdoor PM10 and ozone levels in the school area can be associated with FeNO. The home environment factors tested included cat keeping, dog keeping, different indicators of dampness and mold, second hand smoke (SHS), type of cooking fuel, and biomass burning inside and outside the home.
Materials and methods
Ethics statement
The study protocol was approved by the Ethic Committee of the Research Institute for Health Sciences, Chiang Mai University. Informed consent was obtained from all participating children and their parents.
Study population
The study population came from three randomly selected junior high schools in three cities in upper Northern Thailand (Chiang Rai, Lampun and Mae Hong Son). In Thailand, children start primary school when they are 6 years old and junior high school when they are 12 years old. In each school, all students in eight randomly selected first grade classes were invited to join the study. The students answered a questionnaire the day before the research team arrived to perform the clinical investigation. The questionnaire was answered in the school with assistance from their teachers. The study was performed in dry season (February-March 2018). A total of 270 of 327 invited students (83%) participated. Our studied schools are the main schools of the three provinces and serve students who live in the city area. The area of Chiang Rai province is 1,216 km2, the area of Lamphun province is 479 km2 and the area of Mae Hong Son province is 2,483 km2.
Demographics and allergies
The questionnaire included questions on age, gender, smoking habits, doctor diagnosed allergies. Six yes/no questions asked about doctor diagnosed allergy to cat, dog, pollen, cockroaches, house dust mites and mold, respectively. We combined the information on allergies to one yes/no variable (any doctor diagnosed allergy). Moreover, we asked about the education levels of the father and the mother (elementary school, secondary school, university education). A family education level variable was constructed, coding the education level of the family according to the highest education level among the parents.
Assessment of medical symptoms
There were yes/no questions on doctors’ diagnosed asthma, wheeze, current asthma medication and any asthma attack in the last 12 months adapted from the European Community Respiratory Health Survey (ECRHS) (Citation21). Moreover, there was one question on rhinoconjunctivitis adapted from the International Study of Asthma and Allergies in Childhood (ISAAC) (Citation22) and three questions on severity of symptoms in the last 3 days (Citation23), rhinitis symptoms (irritating, stuffy or runny nose); eye symptoms (itching, burning or irritation of the eyes and breathlessness (dyspnea). For each question, there were seven possible answers on severity of each symptom: (never, very little, slightly, moderately, strongly, very strongly or unbearable). Since few reported severe symptoms, we dichotomized these symptoms variables into any symptom in the main analysis (yes/no).
Assessment of fractional exhaled nitric oxide (FeNO)
FeNO levels were measured by a portable instrument (NIOX-MINO; Aerocrine AB, Solna, Sweden). The sensor in the device was changed periodically according to the manufacturer’s guidance. The FeNO measurements followed ATS guidelines.
Data on the home environment
There were eleven yes/no questions on the home environment in the questionnaire. The asked about cat keeping, dog keeping, any water leakage/flooding in the past 12 months, visible mold on indoor surfaces in the past 12 months, mold odor in the past 12 months, water leakage or indoor mold in the last 5 years, window pane condensation in winter, any tobacco smoking inside the home (SHS), type of cooking fuel (electricity, gas cooking, biomass burning) (more than one alternative possible), biomass burning inside the home (for other reasons than cooking) and biomass burning outside the home. The two questions on water leakage in the last 12 months and water leakage or indoor mold in the last 5 years were combined to a water leakage score (0–2).The two questions on biomass burning inside and outside the home were combined to a biomass burning score (0–2).
Assessment of outdoor air pollution
Air pollution data was collected from the monitoring station located closest to the school. In Lampun, PM10 was measured by Beta Ray Attenuation. In Chiang Rai and Mae Hong Son, PM10 was measured by Tapered Element Oscillating Microbalance (TEOM). Ozone was measured by Ultraviolet Absorbtion Photometry in all cities. The air quality monitoring systems are operated by Thailand Pollution Control Department (PCD) and air pollution data can be assessed at their website (Citation24). The distance between the school and the monitoring station was 0.75 km in Chiang Rai, 0.95 km in Lampun and 3.5 km in Mae Hong Son. We calculated mean concentrations of air pollutants for the past 3 days and 7 days.
Statistical analysis
We categorized FeNO into three groups; low (<20 ppb), intermediate (20–35 ppb and high (>35 ppb), as suggested by the American Thoracic Society (ATS) (Citation25). Moreover FeNO was log transformed (10 logarithm) to obtain a normally distributed variable. Independent t-test was used to test the associations between log transformed FeNO levels and demographic data. Associations between FeNO levels and symptoms severity scores were analyzed by Kendal Tau beta rank correlation test. Two-level linear mixed models (subject and school) were applied to evaluate associations between logFeNO and independent variables. Antilog-beta values were calculated by calculating the 10 exponent (antilog) of the beta values from the models. Initially, we applied one model including the two co-variates (gender and family education) only. In the models, boy and the lowest education level were reference categories. Since very few students were current smokers, we did not include smoking habits as a co-variate in the statistical models. As a next step, we evaluated associations between logFeNO and reported health (diagnosed allergy, diagnosed asthma, and medical symptoms) with one health variable included in each model. Then we evaluated associations between logFeNO, the home environment and outdoor air pollution, adjusting for the two co-variates (one exposure variable in each model). Finally, we analyzed associations between logFeNO and all exposure variables with p < 0.1 in previous models. We used STATA 12.0 (StataCorpLLC, College Station, Texas, USA). Associations were expressed as antilog-beta with 95% confidence intervals (CI) and two-tailed test and 5% level of significance were used.
Results
Descriptive health data
A total of 270 students participated (54.1% girls). The mean age was 12.9 years (range 12–16). Only 0.2% were current smokers and 3.6% were ex-smokers. In total, 52.2% had at least one parent with university education and in 12.1% of the families, both parents had only primary school or lower education. A total of 7.8% had any doctor diagnosed allergy, mainly to house dust mites (6.3%). A total of 11.5% had wheeze but only 5.6% had ever had doctor diagnosed asthma (cumulative incidence) and only 5.3% had current asthma (current asthma attacks or asthma medication). Many students reported rhino conjunctivitis in the last 12 months and eye symptoms (42.8%), rhinitis symptoms (68.8%) and dyspnea (38.3%) in the last three days (). In crude data analysis, boys (p = 0.02) and students with diagnosed allergy (p = 0.001) had higher FeNO, but there was no association between education level and FeNO ().
Table 1. Descriptive data on demographics and doctor diagnosed allergies (N = 270).
Table 2. Descriptive data on FeNO, stratified by gender, family education and allergy.
Normal values for FeNO
We classified FeNO into three groups, low (<20 ppb), intermediate (20–35 ppb) and high (>35 ppb). In total, 70.4% had low, 16.3% intermediate and 13.3% high FeNO values. Students with doctor diagnosed allergy had more often intermediate and high FeNO ().
Table 3. Proportion of students with low, intermediate and high FeNO levels (N = 270).
Household and air pollution data
Data on the home environment and outdoor air pollution are presented in . A total of 14.1% had cats and 24.8% had dogs at home. Signs of dampness, water leakage and mold in the dwelling were common, including window pane condensation in winter (14.6%), visible mold (8.9%), water leakage once in the past 5 years (20.0%) and water leaked more than once in the past 5 years (11.5%). Gas cooking was most common way of cooking (71.3%), almost one fourth used biomass (23.0%) and few homes used only an electric stove (5.7%). Biomass burning inside or outside the home was common (55.6%) and many families burned biomass inside as well as outside the home (36.7%). In the past three days, mean concentration of PM10 (mean of three cities) was 46 µg/m3 (range 31–81) and mean ozone level was 26 µg/m3 (range 23–31) in the past 3 days (lag 3). The air pollution levels in the past 7 days (lag 7) were similar as in the past 3 days (lag 3) ().
Table 4. Descriptive data on home environment and outdoor air pollution for the students (N = 270).
Associations between FeNO, demographics and symptoms
As a next step, we analyzed associations between FeNO and reported heath variables, adjusting for the two co-variates (one health variable per model) (). Any doctor diagnosed allergy, (p = 0.001) cat allergy (p = 0.001) and house dust mite allergy (p = 0.006) were associated with FeNO. Moreover, eye symptoms (p = 0.03), rhinitis symptoms (p = 0.03) and dyspnea (p = 0.09) in the past 3 days were associated with FeNO (). The correlation between FeNO and the dyspnea score was significant (Tau beta 0.10; p = 0.03).
Table 5. Associations between FeNO and reported health (N = 270).
Associations between FeNO and environment
Then we analyzed associations between FeNO and environmental factors, adjusting for the two co-variates (one environmental factor per model). PM10 (lag 3) (p = 0.04) and PM10 (lag 7 (p = 0.04) were negatively associated with FeNO (protective association) (). Finally we included environmental variables from in a mutually adjusted model (). For outdoor air pollution, we included only PM10 (lag 3) since PM10 and ozone and PM10 (lag 3) and PM10 (lag 7) were strongly correlated (r > 0.7). In this mutually adjusted model, indoor mold (p = 0.03), gas cooking (p = 0.03) and PM10 (lag 3) (p = 0.02) were all negatively associated with FeNO. No associations were found between FeNO and gender, family education level or biomass cooking ().
Figure 1. Associations between FeNO, home environment and outdoor PM10 in a mutually adjusted model (N = 270). Analyzed by two level (student, school) linear mixed models, adjusting for gender and family education level (all exposure variable with p < 0.1 in included in the model)
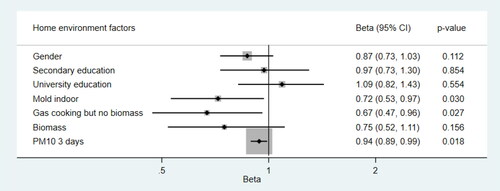
Table 6. Associations between FeNO, home environment and outdoor air pollution (N = 270).
Discussion
In total, 29.6% of the Thai students had elevated FeNO (16.3% intermediate and 13.3% high values) according to normal values suggested by ATS (Citation25). The overall geometric mean value for FeNO was 14.5 ppb (GSD 2.0), somewhat lower (13.9 ppb) in students without doctor diagnosed allergy. This is similar as in a previous study investigating FeNO in healthy Thai volunteers without a history of allergies (Citation26). They found a mean value of 13.6 ppb FeNO in the 11–15 year age group (similar age as in our study). However, in our study only 5.6% had doctor diagnosed asthma and only 3.7% had current asthma medication. Since only a smaller proportion of children with elevated FeNO levels had asthma diagnosis in our study, there could be a number of un-diagnosed asthma cases.
We tested a number of hypotheses. The hypotheses on associations between FeNO and diagnosed allergies, ocular symptoms, nasal symptoms and dyspnea score were verified (all were positive associations). Moreover the hypotheses on associations between FeNO and gas cooking, indoor mold and outdoor PM10 were verified (all were negative associations).
Self-reported doctor diagnosed allergy was a risk factor for FeNO, which is expected since FeNO is an indicator of TH2 driven airway inflammation (Citation4).The main allergy was against house dust mites, other allergies were rare. Skin prick tests have demonstrated that in Thailand, the main allergies to indoor allergens are against house-dust mites, cockroaches, cats and dogs. The main pollen allergies are against Bermuda grass, para grass, sedge and careless weed (Citation27). Thus, we conclude that doctor diagnosed allergy, reported by school children in a questionnaire survey, can reflect ongoing airway inflammation.
We found that current ocular and nasal symptoms and dyspnea in the past 3 days were associated with FeNO, indicating that these symptoms can reflect ongoing airway inflammation. In contrast, we found no significant associations between FeNO and doctor diagnosed asthma. Two previous studies in junior high school students in China (Citation28) and Malaysia (Citation29) found associations between doctor diagnosed asthma and FeNO. The lack of such association in our study could be due to limited statistical power, since the study size was relatively small and few students reported doctor diagnosed asthma. However our results indicate that in a subtropical country, with distinct dry and wet season and fluctuating air pollution levels, it is important to ask about respiratory symptoms in the past days.
In our study, visible indoor mold was negatively associated FeNO (protective). Previous studies have found different associations between indoor mold and FeNO. Five studies did not find any association between visible mold in homes and FeNO (Citation14,Citation16,Citation28,Citation30,Citation31). However, studies measuring specific microbial agents have reported that exposure to endotoxin (Citation17), the gram positive soil bacteria Streptomyces sp. (Citation18) and higher alpha-diversity of mold species in homes (Citation32) can be associated with lower FeNO while exposure to the molds Aspergillus fumigatus (Citation33) and Aspergillus versicolor (Citation29) can be associated with higher FeNO. One study found that outdoor air concentration of Cladosporium spores was associated with higher FeNO, but only in subjects sensitized to this mold (Citation34). Thus, the effect of indoor microbial growth on FeNO can depend on types of mold and bacteria species and more detailed studies on household microbial exposure in Thailand and FeNO are needed.
Gas cooking was associated with lower FeNO as compared to cooking on electric stoves. One previous study found that cooking by liquefied petroleum gas was associated with higher FeNO than cooking by natural gas (Citation15), but we found no previous study comparing gas cooking with electric stoves on FeNO. The group using only electric stoves for cooking was small, only 15 students (5.7%), and thus our unexpected negative association between gas cooking and FeNO could be due to selection effects or residual confounding. More studies are needed on the role of type of cooking fuel in Asia on FeNO.
We had data on two outdoor air pollutants (PM10 and ozone) and PM10 was negatively associated with FeNO. This is an opposite finding as compared to the review on 27 panel studies, concluding that PM10 is associated with higher FeNO (Citation5). All Asian panel studies in this review, except one Japanese study, were from China. PM10 from different pollution sources can have different chemical composition. In China, coal burning is a major source of PM10. In upper northern Thailand the major source of PM10 in dry season is biomass burning. Moreover respiratory health can differ between ethnic groups in Asia. In Malaysia and Singapore, where there three major ethnic groups (Malay, Indian and Chinese ethnicity), ethnic differences in prevalence of asthma and rhinitis symptoms have been reported (with lowest prevalence of symptoms in Chinese population) (Citation35–37). One major limitation of our study is the cross-sectional study design, and moreover the mean PM10 levels was similar as the WHO guideline value of 50 µg/m3 (Citation38). In addition, since some students lived far away from the schools, they could have a different exposure to outdoor air pollution in their residential area. Thus, future panel studies, on associations between PM10 exposure in different Asian countries and FeNO in school children are needed. These studies should assess outdoor exposure at school as well as in their residential area.
The study has some strengths. FeNO is a well-recognized biomarker of TH2 driven airway inflammation (Citation4). We found no previous studies on environmental risk factors of FeNO in Thai school children. Selection bias can influence epidemiological studies. Our study population was recruited from randomly selected classes in randomly selected schools in three cities in upper northern Thailand, with good participation rate (83%). We had somewhat more girls than boys among the participants. However selection bias should not be a major problem. FeNO is a clinical variable not affected by information bias. Another strength is that we adjusted for the education level of the family and used mutually adjusted statistical models However, one limitation is that the study only included three schools. Moreover, there were no measurements or inspections in the homes or data on PM10 or ozone in the residential areas of the participants. In addition, FeNO does not measure neutrophilic inflammation, a type of inflammation caused by some types of air pollutants. Moreover, the cross-sectional study design limits the possibility to draw conclusions on causality.
In conclusion, 30% of junior high school students in upper northern Thailand can have elevated FeNO values in dry season, indicating Th2 driven airway inflammation. Diagnosed allergies, especially to cat and HDM, can increase FeNO while indoor mold and gas cooking may reduce FeNO. Ongoing ocular, rhinitis and dyspnea symptoms in dry season in northern Thailand can be linked to can be associated with FeNO, a biomarker of airway inflammation.
Conflict of interest
The authors declare no conflict of interest. The authors alone are responsible for the content and writing of this article.
Additional information
Funding
References
- GDB 2019. Risk Factor Collaborators. Global burden of 87 risk factors in 204 countries and territories, 1990-2019: a systematic analysis of the Global Burden of Disease Study 2019. Lancet. 2020;396:1223–1249. doi:https://doi.org/10.1016/S0140-6736(20)30752-2.
- Pawankar R, Baena-Cagnani C, Bousquet J, Canonica GW, Cruz AA, Kalinder MA, Lanier BQ. State of World Allergy Report 2008: allergy and chronic respiratory diseases. World Allergy Organ. 2008;(Suppl 1):4–17.
- Chinratanapisit S, Suratannon N, Pacharn P, Sritipsukho P, Vichyanond P. Prevalence and severity of astma, rhinoconjunctivitis and eczema in children from the Bangkok area: the Global Asthma Network (GAN) Phase I. Asian Pac J Allergy Immunol. 2019;37:226–231.
- Alving K, Malinovschi A. Basic aspects of exhaled nitric oxide. In: Horvath I, de Jongste JC, editors. Exhaled biomarkers. Plumouth (UK): European Respiratory Society; 2010. p. 1–32.
- Chen X, Liu F, Niu Z, Mao S, Tang H, Li N, Chen G, Liu S, Lu Y, Xiang H. The association between short-term exposure to ambient air pollution and fractional exhaled nitric oxide level: a systematic review and meta-analysis of panel studies. Environ Pollut. 2020;265(Pt A):114833. doi:https://doi.org/10.1016/j.envpol.2020.114833.
- Ramakreshnan L, Aghamohammadi N, Fong CS, Bulgiba A, Zaki RA, Wong LP, Sulaiman NM. Haze and health impacts in ASEAN countries: a systematic review. Environ Sci Pollut Res Int. 2018;25(3):2096–2111. doi:https://doi.org/10.1007/s11356-017-0860-y.
- Allen RW, Mar T, Koenig J, Liu LJ, Gould T, Simpson C, Larson T. Changes of lung function and airway inflammation among asthmatic children in a wood smoke-impacted urban area. Inhal Toxicol. 2008;20:432–433.
- Benka-Coker ML, Clark ML, Rajkumar S, Young BN, Bachand AM, Balmes JR, Brook R, Nelson TL, Volckens J, Reynolds SJ, Wilson A, et al. Exposure to household air pollution from biomass cook stoves and levels of fractional exhaled nitric oxide (FeNO) among Honduran women. IJERPH. 2018;15(11):2544. doi:https://doi.org/10.3390/ijerph15112544.
- Pongpiachan S, Tipmanee D, Khumsup C, Kittikoon I, Hirunyatrakul P. Assessing risks to adults and preschool children posed by PM2.5-bound polycyclic aromatic hydrocarbons (PAHs) during a biomass burning episode in Northern Thailand. Sci Total Environ. 2015;508:435–444. doi:https://doi.org/10.1016/j.scitotenv.2014.12.019.
- Phairuang W, Suwattiga P, Chetiyanukornkul T, Hongtieab S, Limpaseni W, Ikemori F, Hata M, Furuuchi M. The influence of the open burning of agricultural biomass and forest fires in Thailand on the carbonaceous components in size-fractionated particles. Environ Pollut. 2019;247:238–247. doi:https://doi.org/10.1016/j.envpol.2019.01.001.
- Ruchiraset A, Tantrakarnapa K. Time series modeling of pneumonia admissions and its association with air pollution and climate variables in Chiang Mai province, Thailand. Environ Sci Pollut Res Int. 2018;25(33):33277–33285. doi:https://doi.org/10.1007/s11356-018-3284-4.
- Norbäck D, Wang J. Household air pollution (HAP) and adult respiratory health. Eur Respir J. 2021;57(1):2003520. doi:https://doi.org/10.1183/13993003.03520-2020.
- Janson C, Kalm-Stephens P, Foucard T, Norbäck D, Alving K, Nordvall SL. Exhaled nitric oxide levels in school children in relation to IgE sensitization and window pane condensation. Respir Med. 2005;99(8):1015–1021. doi:https://doi.org/10.1016/j.rmed.2005.02.003.
- Olaniyan T, Dalvie MA, Röösli M, Naidoo R, Künzli N, de Hoogh K, Parker B, Leaner J, Jeebhay M. Astma-related outcomes associated with indoor air pollutants among schoolchildren from four informal settlements in two municipalities in the Western Cape Province of South Africa. Indoor Air. 2019;29(1):89–100. doi:https://doi.org/10.1111/ina.12511.
- Hou F, Huang X, Liu C, Sun H, Zhou T, Song Y, Rong Y, Zhu B, Wang J, He M, et al. Housing characteristics in relation to exhaled nitric oxide in China. Am J Health Behav. 2015;39(1):88–98. doi:https://doi.org/10.5993/AJHB.39.1.10.
- Kovesi TA, Dales RE. Effects of the indoor environment on the fraction of exhaled nitric oxide in school-age children. Can Respir J. 2009;16:18–23.
- Casas L, Tischer C, Wouters IM, Torrent M, Gehring U, Garcia-Esteban R, Thiering E, Postma DS, de Jongste J, Smit HA, et al. Early life microbial exposure and fractional exhaled nitric oxide in school-age children: a prospective birth cohort study. Environ Health. 2013;12:103. doi:https://doi.org/10.1186/1476-069X-12-103.
- Johansson E, Reponen T, Vesper S, Levin L, Lockey J, Ryan P, Bernstein DI, Villareal M, Khurana Hershey GK, Schaffer C, et al. Microbial content of household dust associated with exhaled NO in asthmatic children. Environ Int. 2013;59:141–147. doi:https://doi.org/10.1016/j.envint.2013.05.011.
- Yan DH, Chung FF, Lin SJ, Wan GH. The relationship among Dermatophagoides pteronyssinus exposure, exhaled nitric oxide, and exhaled breath condensate pH levels in atopic asthmatic children. Medicine. 2016;95(39):e4825. doi:https://doi.org/10.1097/MD.0000000000004825.
- Raja S, Xu Y, Ferro AR, Jaques PA, Hopke PK. Resuspension of indoor aeroallergens and relationship to lung inflammation in asthmatic children. Environ Int. 2010;36(1):8–14. doi:https://doi.org/10.1016/j.envint.2009.09.001.
- Janson C, Anto J, Burney P, Chinn S, de Marco R, Heinrich J, Jarvis D, Kuenzli N, Leynaert B, Luczynska C, et al. The European Community Respiratory Health Survey: what are the main results so far? European Community Respiratory Health Survey II. Eur Respir J. 2001;18(3):598–611. doi:https://doi.org/10.1183/09031936.01.00205801.
- Asher MI, Montefort S, Björkstén B, Lai CKW, Strachan DP, Weiland SK, Williams H, ISAAC Phase Three Study Group. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC phases one and three repeat multicountry cross-sectional surveys. Lancet. 2006;368(9537):733–743. doi:https://doi.org/10.1016/S0140-6736(06)69283-0.
- Norbäck D, Lindgren T, Wieslander G. Changes in ocular and nasal signs and symptoms among air crew in relation to air humidification on intercontinental flights. Scand J Work Environ Health. 2006;32(2):138–144. doi:https://doi.org/10.5271/sjweh.989.
- Thailand Pollution Control Department (PCD) (PCD air4thai). http://air4thai.pcd.go.th/webV2/.
- Dweik RA, Boggs PB, Erzurum SC, Irvin CG, Leigh MW, Lundberg JO, Olin A-C, Plummer AL, Taylor DR. An official ATS clinical practice guideline: interpretation of exhaled nitric oxide levels (FENO) for clinical applications. Am J Respir Crit Care Med. 2011;184(5):602–615. doi:https://doi.org/10.1164/rccm.9120-11ST.
- Suksawat Y, Pacharn P, Jirapongsananurur O, Visitsunthorn N. Determination of fractional exhaled nitric oxide (FENO) reference values in healthy Thai population. Asian Pac J Allegy Immunol. 2017;35:127–131.
- Bunnag C, Jareoncharsri P, Tantilipikorn P, Vichyanond P, Pawankar R. Epidemiology and current status of allergic rhinitis and asthma in Thailand – ARIA Asia-Pacific Workshop report. Asian Pac J Allergy Immunol. 2009;27(1):79–86.
- Zhao Z, Huang C, Zhang X, Xu F, Kan H, Song W, Wieslander G, Norback D. Fractional exhaled nitric oxide in Chinese children with asthma and allergies—a two-city study. Respir Med. 2013;107(2):161–171. doi:https://doi.org/10.1016/j.rmed.2012.11.001.
- Norbäck D, Hashim JH, Hashim Z, Cai GH, Sooria V, Ismail SA, Wieslander G. Respiratory symptoms and fractional exhaled nitric oxide (FeNO) among students in Penang, Malaysia in relation to signs of dampness at school and fungal DNA in school dust. SciTotal Environ. 2017;577:148–154. doi:https://doi.org/10.1016/j.scitotenv.2016.10.148.
- Lim FL, Hashim Z, Md Said S, Than LT, Hashim JH, Norbäck D. Fractional exhaled nitric oxide (FeNO) among office workers in an academic institution Malaysia—associations with asthma, allergies and office environment. J Asthma. 2016;53:353–361.
- Mustonen K, Karvonen AM, Kirjavainen P, Roponen M, Schaub B, Hyvärinen A, Frey U, Renz H, Pfefferle PI, Genuneit J, et al. Moisture damage in home associates with systemic inflammation in children. Indoor Air. 2016;26(3):439–447. doi:https://doi.org/10.1111/ina.12216.
- Vandenborght LE, Enaud R, Urien C, Coron N, Girodet PO, Ferreira S, Berger P, Delhaes L. Type 2-high asthma is associated with a specific indoor mycobiome and microbiome. J Allergy Clin Immunol. 2021;147(4):1296–1305. doi:https://doi.org/10.1016/j.jaci.2020.08.035.
- Stark HJ, Randell JT, Hirvonen MR, Purokivi MK, Roponen MH, Tukiainen HO. The effects of Aspergillus fumigatus challenge on exhaled and nasal NO levels. Eur Respir J. 2005;26(5):887–893. doi:https://doi.org/10.1183/09031936.05.00061405.
- Tham R, Erbas B, Dharmage SC, Tang ML, Aldakheel F, Lodge CJ, Thomas PS, Taylor PE, Abramson MJ, Lowe AJ. Outdoor fungal spores and acute respiratory effects in vulnerable individuals. Environ Res. 2019;178:108675. doi:https://doi.org/10.1016/j.envres.2019.108675.
- Ng TP, Niti M, Tan WC. Trends and ethnic differences in asthma hospitalization rates in Singapore, 1991 to 1998. Ann Allergy Asthma Immunol. 2003;90(1):51–55. doi:https://doi.org/10.1016/S1081-1206(10)63614-9.
- Ng TP, Hui KP, Tan WC. Prevalence of asthma and risk factors among Chinese, Malay and Indian adults in Singapore. Thorax. 1994;49(4):347–351. doi:https://doi.org/10.1136/thx.49.4.347.
- Norback D, Hashim Z, Ali F, Hashim JF. Asthma symptoms and respiratory infections in Malaysian students-associations with ethnicity and chemical exposure at home and school. Environ Res. 2021;197:111061. doi:https://doi.org/10.1016/j.envres.2021.111061.
- WHO. Health effects of air pollution: an overview. In: Air quality guidelines global update 2005: particulate matter, ozone, nitrogen dioxide and sulfur dioxide. Copenhagen (Denmark): WHO Regional Office for Europe; 2006. p. 87–102.
