Abstract
Background: Primary tumor volume is as an important and independent prognostic factor in Ewing sarcoma. However, the observer variability of magnetic resonance imaging (MRI)-based primary tumor volume measurements in newly diagnosed Ewing sarcoma has never been investigated. Furthermore, it is unclear how MRI-based volume measurements compare to 18F-fluoro-2-deoxy-D-glucose (FDG) positron emission tomography (PET)-based volume measurements. This study aimed to determine the observer variability of simplified MRI-based primary tumor volume measurements in newly diagnosed treatment-naive Ewing sarcoma and to compare them to the actual primary tumor volume at MRI and the FDG-PET-based metabolically active tumor volume (MATV).
Material and methods: Twenty-nine newly diagnosed Ewing sarcoma patients with pretreatment MRI (of whom 11 also underwent FDG-PET) were included. Both exact and dichotomized (according to the proposed threshold of 200 mL) primary tumor volume measurements were analyzed.
Results: Mean inter- and intraobserver differences of MRI-based simplified tumor volume ± limits of agreement varied between 15–42 ± 155–204 mL and between 9–16 ± 64–250 mL, respectively. Inter- and intraobserver agreements of dichotomized MRI-based simplified tumor volume measurements was very good (κ = 0.827–1.000). Mean difference between simplified and actual tumor volumes at MRI ± limits of agreement was 60 ± 381 mL. Agreement between dichotomized simplified and actual tumor volumes at MRI was very good (κ = 0.839). Mean difference between MRI-based simplified tumor volume and MATV ± limits of agreement was 181 ± 549 mL and almost significantly different (p = .0581). Agreement between dichotomized MRI-based simplified tumor volume and MATV was moderate (κ = 0.560).
Conclusions: Exact MRI-based simplified primary tumor volume measurements in Ewing sarcoma suffer from considerable observer variability, but observer agreement of dichotomized measurements (≤200 mL vs. >200 mL) is very good and generally matches MRI-based actual volume measurements. MRI-based primary tumor volume measurements poorly-moderately agree with and tend to be higher than the MATV.
Introduction
Ewing sarcoma is a high grade primary tumor of bone, most commonly with soft tissue extension [Citation1]. In rare cases the lesion is purely in the soft tissue [Citation1]. Its peak incidence is during adolescence and young adulthood [Citation1]. The overall annual incidence of Ewing sarcoma is approximately 2.93 cases/1,000,000 [Citation2]. Metastatic status at diagnosis is the strongest prognostic factor across different treatment strategies [Citation1]. Five-year overall survival remains 30% for patients with initially metastatic disease [Citation1]. Accurate staging, for which imaging plays an important role, is thus important for correct prognostication and treatment planning. Primary tumor volume has also been recognized as an important and independent prognostic factor, both in localized disease and primary disseminated multifocal Ewing sarcoma [Citation1]. In Europe, primary tumor volume is even used to tailor maintenance chemotherapy in patients with localized disease [Citation1]. The optimal cutoff value for prognostic stratification is still underinvestigated, although a primary tumor volume of more than 200 mL has been proposed by authoritative experts to identify those patients with a worse outcome [Citation1], which is based on several previous studies [Citation3–6]. The imaging modality at which these measurements should be done is also unclear, although they are currently usually performed at magnetic resonance imaging (MRI), which is regarded as the optimal radiological modality for local evaluation of Ewing sarcoma, thanks to its high contrast resolution [Citation7].
Importantly, the observer variability of MRI-based primary tumor volume measurements in newly diagnosed Ewing sarcoma has never been investigated. Such knowledge is crucial for reliable prognostic stratification. Another issue is that primary tumor volume at MRI is currently calculated in a simplified manner by multiplying measured tumor dimensions in three orthogonal directions with a conversion factor [Citation3–6]. However, it is unknown whether these MRI-based simplified tumor volume measurements are interchangeable with the actual tumor volume at MRI as measured by manual slice-by-slice segmentation. Another relevant issue is that signal abnormalities at MRI may not only reflect viable tumor, but are likely to represent a combination of viable tumor, necrotic tissue and non-neoplastic surrounding tissue alterations due to phenomena such as tumor-induced inflammation, mechanical stress and compression. On the other hand, MRI may fail to detect tumor in diffusely involved tissue without structural or signal abnormalities. Meanwhile, 18F-fluoro-2-deoxy-D-glucose (FDG) positron emission tomography (PET) is increasingly used in Ewing sarcoma as an alternative to computed tomography (CT) alone and bone scintigraphy and as an adjunct to MRI, for staging, therapy response assessment and restaging [Citation8]. An advantage of FDG-PET is its high tumor-to-background contrast and the availability of software to automatically measure metabolically active tumor volume (MATV) with proven high feasibility of tumor segmentation and scan-rescan repeatability [Citation9,Citation10]. A comparison of primary tumor volume measurements at MRI to FDG-PET-based MATV measurements is relevant to understand to what extent the former appears to over- or underestimate the viable tumor volume.
The purpose of this study was therefore to determine the inter- and intraobserver agreement of simplified MRI-based primary tumor volume measurements in newly diagnosed treatment-naive Ewing sarcoma and to compare them to the actual primary tumor volume at MRI and the MATV as measured at FDG-PET.
Material and methods
Study design and patients
The local institutional review board approved this retrospective study and waived the requirement for written informed consent. The hospital's database was searched for all patients who were newly diagnosed with Ewing sarcoma between March 2009 and April 2017. The hospital is a university clinic and one of the four national designated centers for treatment of bone tumors. Inclusion criteria for this study were: histopathologically proven newly diagnosed treatment-naive Ewing sarcoma in bone and/or soft tissue and availability of a pretreatment MRI examination. Exclusion criteria for this study were: chemotherapy, radiation therapy or tumor resection before MRI, recurrent Ewing sarcoma and MRI without gadolinium-enhanced sequences. For the subgroup analysis in which MRI was compared to FDG-PET, an FDG-PET scan had to be performed within 14 days of MRI, FDG-PET had to be performed with a resEARch 4 Life (EARL)-accredited scanner [Citation11] and patients should have remained treatment-naive before the FDG-PET examination.
MRI acquisition
MRI scans were performed using different clinical 1.5-T MRI systems. MRI sequences were adapted to body region and local tumor extent. Therefore, MRI protocols were not uniform. Nevertheless, all patients were scanned with both unenhanced T1-weighted, (fat-suppressed) T2-weighted and gadolinium-enhanced sequences. Applied slice thicknesses varied between 1–4 mm (three-dimensional isotropic acquisition). Sequences or reconstructed images (in case a three-dimensional isotropic MRI sequence was acquired) were oriented axially and in at least one longitudinal direction with regard to the involved bone.
FDG-PET acquisition
Patients fasted for at least six hours and blood glucose levels were checked to be less than 11 mmol/L before 3 MBq FDG/kg body weight was administered intravenously. Approximately 60 min after FDG administration, PET scanning was performed using an EARL-accredited combined PET/CT system (Biograph mCT PET/CT, Siemens, Knoxville, TN, USA). Data acquisition and reconstruction were performed according to European Association of Nuclear Medicine guidelines [Citation11].
MRI evaluation
Simplified primary tumor volume measurements at MRI were done by three different radiologists (Ö.K., T.C.K. and J.O), with four, six and nine years of experience in musculoskeletal MRI, respectively) using a PACS workstation (Carestream Vue PACS version 11.4.1.1102, Carestream Health, Inc, Rochester, NY, USA). All three readers were aware of the diagnosis of Ewing sarcoma, but were blinded to original MRI reports, FDG-PET findings and each other's measurements. Radiographs and other imaging tests, if available, were not reviewed during MRI evaluation. Each reader measured the maximum left-right (LR), anterior-posterior (AP) and cranio-caudal (CC) tumor dimensions (). Tumor tissue at MRI was defined as all areas with increased contrast-enhancement relative to normal background on gadolinium-enhanced sequences [Citation7]. The simplified tumor volume was then calculated according to the formula LR × AP × CC tumor dimensions × 0.52, as published previously [Citation3–6]. This simplified determination of primary tumor volume is the currently used measurement method for Ewing sarcoma in clinical practice. Under the same conditions and while being blinded to their first series of measurements, each reader measured simplified tumor volume in each patient a second time. Time interval between these two series of measurements was at least two weeks to avoid recognition bias.
Figure 1. A 9-year-old boy with Ewing sarcoma in and around the right clavicle. Tumor dimension measurements (left-right [LR], anterior-posterior [AP] and cranio-caudal [CC]) are shown on axial (A) and coronal (B) gadolinium-enhanced images. Simplified MRI-based tumor volume was then calculated as LR × AP × CC tumor dimensions × 0.52 [Citation3–6]. Actual MRI-based tumor volume was calculated by first drawing free-hand tumor regions of interest on each axial slice (two slice examples are shown (C), multiplying tumor area by the slice thickness for each slice and then summing the tumor volumes of the slices. Coronal FDG-PET/CT (D) shows the tumor to be FDG-avid relative to mediastinum and liver. Using in-house-developed software (ACCURATE), supervised automated tumor segmentation was performed and MATV (50% threshold of SUVpeak corrected for local background) was calculated [Citation9,Citation10]. Segmentation results are shown on coronal (E) and sagittal (F) maximum intensity projection images.
![Figure 1. A 9-year-old boy with Ewing sarcoma in and around the right clavicle. Tumor dimension measurements (left-right [LR], anterior-posterior [AP] and cranio-caudal [CC]) are shown on axial (A) and coronal (B) gadolinium-enhanced images. Simplified MRI-based tumor volume was then calculated as LR × AP × CC tumor dimensions × 0.52 [Citation3–6]. Actual MRI-based tumor volume was calculated by first drawing free-hand tumor regions of interest on each axial slice (two slice examples are shown (C), multiplying tumor area by the slice thickness for each slice and then summing the tumor volumes of the slices. Coronal FDG-PET/CT (D) shows the tumor to be FDG-avid relative to mediastinum and liver. Using in-house-developed software (ACCURATE), supervised automated tumor segmentation was performed and MATV (50% threshold of SUVpeak corrected for local background) was calculated [Citation9,Citation10]. Segmentation results are shown on coronal (E) and sagittal (F) maximum intensity projection images.](/cms/asset/8d614b5b-abab-422f-a058-ff23eea1a3b3/ionc_a_1398411_f0001_c.jpg)
Yet another two weeks later and while being blinded to all previous measurements, one radiologist (T.C.K.) measured tumor areas on all axial gadolinium-enhanced slices on which each tumor was visible, using freehand regions of interest (). The actual tumor volume at MRI in each patient was calculated by multiplying tumor area by the slice thickness for each slice and then summing the tumor volumes of the slices.
All MRI-based tumor volumes were also dichotomized into ≤200 vs. >200 mL groups, which is the cutoff that was recently suggested by authoritative experts [Citation1].
FDG-PET evaluation
Primary tumor locations were identified on the whole-body FDG-PET scans. Using in-house-developed software (ACCURATE), volumes of interest encompassing the primary tumors were automatically generated using a 50% threshold of the peak standardized uptake value (SUVpeak) adapted for local background, as described previously [Citation9,Citation10]. For each volume of interest, SUVpeak (1.2 cm3 spheric region positioned to maximize its mean value) and MATV (50% threshold of SUVpeak corrected for local background) were calculated [Citation9,Citation10]. FDG-PET-based MATV was also dichotomized into ≤200 and >200 mL groups [Citation1].
Statistical analysis
Inter- and intraobserver agreements of MRI-based simplified tumor volume measurements were determined as mean absolute difference (bias) and 95% confidence interval of the mean difference (limits of agreement) according to the methods of Bland and Altman [Citation12]. Intra- and interobserver agreements of dichotomized MRI-based simplified tumor volume measurements (≤200 mL vs. > 200 mL) were analyzed using the unweighted κ statistic, defined as poor (<0.2), fair (>0.2 to ≤0.4), moderate (>0.4 to ≤0.6), good (>0.6 to ≤0.8) and very good (>0.8 to ≤1) agreement.
The mean MRI-based simplified tumor volume in each patient was then calculated based on the average of the six measurements in each patient (three observers with two measurements) for further analyses. Agreement between MRI-based simplified tumor volume, MRI-based actual tumor volume and FDG-PET-based MATV was determined using Bland-Atman analyses [Citation12] and agreement between the three dichotomized volume metrics (≤200 vs. >200 mL) was analyzed using the unweighted κ statistic. Kolmogorov-Smirnov tests were used to check whether MRI-based simplified tumor volume, MRI-based actual tumor volume, and FDG-PET-based MATV were normally distributed. Differences between MRI-based simplified tumor volume and FDG-PET-based MATV and between MRI-based actual tumor volume and FDG-PET-based MATV were then assessed using two-tailed paired t-tests. p values < .05 were considered statistically significant.
Statistical analyses were executed using MedCalc version 17.2 Software (MedCalc, Mariakerke, Belgium).
Results
Patients
A total of 40 patients with newly diagnosed Ewing sarcoma were potentially eligible for inclusion. Of these 40 patients, five were excluded because no MRI was performed, three were excluded because gadolinium-enhanced sequences were not acquired and three more were excluded because primary tumor resection was done before MRI. Thus, 29 patients (17 males and 12 females, with mean age of 20.4 ± 15.6 years [range: 5–64 years]) were finally included. Twenty-four patients had skeletal involvement, with primary tumors located in the pelvic bone (n = 5), femur (n = 4), ribs (n = 4), vertebrae (n = 3), tibia (n = 2), skull (n = 1), clavicle (n = 1), humerus (n = 1), sacrum (n = 1), fibula (n = 1) and calcaneus (n = 1). Five patients only had extra-osseous involvement, with primary tumor locations in the maxillary sinus (n = 1), intraspinal extradural at the cervical level (n = 1), gluteal muscles (n = 1), quadriceps muscles (n = 1) and hamstrings (n = 1). Twenty-one patients had localized disease, whereas eight patients had metastatic disease.
Twelve of these 29 (41%) patients had also undergone FDG-PET, of whom one was not included for further analysis due to a time interval of 41 days between MRI and FDG-PET. In the remaining 11 patients (seven males and four females, with mean age of 11.8 ± 3.6 years [range: 5–17 years]), MRI was performed before FDG-PET in six patients and after FDG-PET in five patients, with a time interval of 3.6 ± 3.6 days (range: 0–10 days) between the two examinations. The tumors were located in the pelvic bone (n = 3), femur (n = 3), ribs (n = 2), clavicle (n = 1), vertebra (n = 1) and humerus (n = 1). Whole-body FDG-PET/CT (cranial vertex to toes) was performed in five patients, FDG-PET/CT from cranial vertex to knees was performed in three patients and FDG-PET/CT from cranial vertex to mid-thighs was performed in three patients. Ten patients had localized disease, whereas one patient had metastatic disease.
All measured primary tumor volumes are displayed in .
Table 1. Measured primary volumes per patient.
Inter- and intraobserver agreement MRI-based simplified tumor volume measurements
Mean interobserver differences in MRI-based simplified tumor volume ± limits of agreement were 27 ± 204 mL for observer 1 vs. observer 2, 42 ± 155 mL for observer 1 vs. observer 3 and 15 mL ± 186 mL for observer 2 vs. observer 3. Mean intraobserver differences in MRI-based simplified tumor volume ± limits of agreement were 9 ± 64 for observer 1, 16 ± 134 for observer 2 and 14 ± 250 mL for observer 3, respectively. Corresponding Bland-Altman plots for inter- and intraobserver agreement ( and , respectively) show that measurement variations appear to depend to some degree on the magnitude of measurements. Inter- and intraobserver agreements of dichotomized MRI-based simplified tumor volume measurements (≤200 vs. >200 mL) was very good (κ = 0.827–1.000), with only four discrepancies among a total of 174 interobserver (29 × 3) and intraobserver (29 × 3) comparisons.
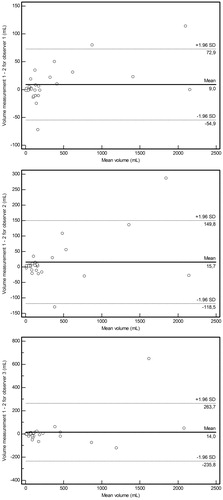
Figure 2. Interobserver agreement of MRI-based simplified tumor volume measurements. Bland-Altman plots of difference of MRI-based simplified tumor volume measurements (y-axis) against mean volume (x-axis), with mean absolute difference (bias) (continuous line) and 95% confidence interval of the mean difference (limits of agreement) (dashed lines) for observer 1 vs. 2 (upper panel), observer 1 vs. 3 (middle panel) and observer 2 vs. 3 (lower panel).
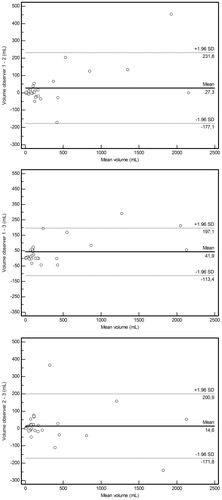
Figure 3. Intraobserver agreement of MRI-based simplified tumor volume measurements. Bland-Altman plots of difference of MRI-based simplified tumor volume measurements (y-axis) against mean volume (x-axis), with mean absolute difference (bias) (continuous line) and 95% confidence interval of the mean difference (limits of agreement) (dashed lines) for observer 1 (upper panel), observer 2 (middle panel) and observer 3 (lower panel).
MRI-based simplified tumor volume versus MRI-based actual tumor volume
Mean difference between MRI-based simplified tumor volume and MRI-based actual tumor volume ± limits of agreement was 60 ± 381 mL (). Agreement between dichotomized MRI-based simplified tumor volume and MRI-based actual tumor volume measurements (≤200 vs. >200 mL) was very good (κ = 0.839, two discrepancies).
Figure 4. Agreement between MRI-based simplified tumor volume, MRI-based actual tumor volumeand FDG-PET-based MATV measurements. Bland-Altman plots of difference of tumor volume measurements (y-axis) against mean volume (x-axis), with mean absolute difference (bias) (continuous line) and 95% confidence interval of the mean difference (limits of agreement) (dashed lines) for MRI-based simplified tumor volume vs. MRI-based actual tumor volume (upper panel), MRI-based simplified tumor volume vs. FDG-PET-based MATV (middle panel) and MRI-based actual tumor volume vs. FDG-PET-based MATV (lower panel).
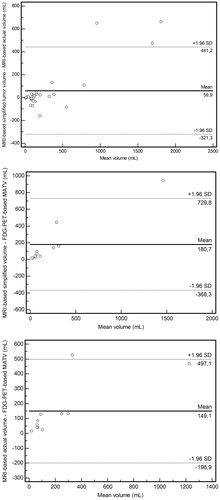
MRI-based tumor volumes versus FDG-PET-based MATV
Mean difference between MRI-based simplified tumor volume and FDG-PET-based MATV ± limits of agreement was 181 ± 549 mL (). Mean difference between MRI-based actual tumor volume and FDG-PET-based MATV ± limits of agreement was 149 ± 348 mL (). Agreement between dichotomized MRI-based simplified tumor volume and FDG-PET-based MATV measurements (≤200 vs. >200 mL) and agreement between dichotomized MRI-based actual tumor volume and FDG-PET-based MATV measurements were both moderate (κ = 0.560, two discrepancies).
Kolmogorov-Smirnov tests confirmed that MRI-based simplified tumor volume (p = .271, 341 ± 551 mL), MRI-based actual tumor volume (p = .296, 309 ± 417 mL), and FDG-PET-based MATV (p = 0.163, 160 ± 285 mL) were normally distributed, justifying comparison with two-tailed paired t-tests. MRI-based simplified tumor volume was nearly significantly higher than FDG-PET-based MATV (p = .0581) and MRI-based actual tumor volume was significantly higher than FDG-PET-based MATV (p = .0193).
Discussion
The results of this study show that the observer variability of exact MRI-based simplified tumor volume measurements is rather high (especially for larger tumors), but both inter- and intraobserver agreement of dichotomized measurements (≤200 vs. >200 mL) are very good. The same results apply to the agreement between MRI-based simplified tumor volume measurements and MRI-based actual tumor volume measurements according to manual slice-by-slice tumor segmentation. Therefore, as long as exact measurements on a continuous scale are not required, MRI-based simplified primary tumor volume measurements can be regarded as quite robust to stratify patients into good and poor prognostic groups according to the 200 mL threshold. Nevertheless, even dichotomized MRI-based simplified primary tumor volume measurements are not perfect, given the fact that these did not agree with MRI-based actual tumor volume measurements in two of 29 (7%) cases in this study. The non-negligible observer variability in MRI-based simplified volume measurements may in part be explained by the limited specificity of MRI acquisitions to depict tumor boundaries. Another important finding of the present study is that both exact and dichotomized (≤200 vs. >200 mL) MRI-based volume measurements poorly to moderately agree with the FDG-PET-based MATV, with primary tumor volumes according to the former being (nearly) significantly higher than primary tumor volumes according to the latter. This may be explained by the fact that gadolinium-enhancement is not specific in that it may represent both tumor tissue and non-neoplastic surrounding tissue alterations. On the other hand, it has not been proven that the FDG-PET-based MATV more accurately reflects the true primary tumor volume due to a lack of histopathological correlation. It should be noted that FDG is not tumor-specific either and may also accumulate in surrounding inflammatory tissue [Citation13].
Until now, there were no studies that addressed primary tumor volume measurement methodology in Ewing sarcoma. Before the routine availability of cross-sectional imaging, primary tumor volumes were assessed on conventional radiographs, based on the formula LR × AP × CC tumor dimensions × 0.52 [Citation3–6]. This formula was proposed for extremity tumors with extraosseous extensions or metaphyseal sites, assuming an ellipsoidal tumor configuration [Citation3–6]. For diaphyseal tumors without extraosseous extension, the formula LR × AP × CC tumor dimensions × 0.785 was proposed, assuming a cylindrical tumor configuration [Citation3–6]. However, since an associated soft-tissue mass in seen in about 96% of Ewing sarcoma patients [Citation7], the former formula can be used in virtually all patients, as was the case in the present study. MRI has nowadays replaced plain radiography for local tumor evaluation, but primary tumor volumes are still measured according to this simplified formula, in part due to the fact that manual slice-by-slice tumor segmentation is too cumbersome and time-consuming to be clinically feasible and in part due to the fact that (validated) software for automatic tumor segmentation is not available at every institution. Given the disagreements between MRI and FDG-PET with regard to both exact and dichotomized primary tumor volume measurements, future studies are required to compare the prognostic value of MRI- and FDG-PET-based primary tumor volume measurements and to determine whether MRI should remain the method of choice for this purpose. These future studies should also aim to standardize imaging protocols to allow for more widespread clinical implementation.
This study had several limitations. First, advanced MRI sequences such as diffusion-weighted imaging [Citation14], which may potentially more accurately discriminate viable tumor volume from surrounding non-neoplastic tissue than gadolinium-enhanced sequences, have not been investigated. Second, the software that was used for FDG-PET analysis (ACCURATE) [Citation9,Citation10] was developed in-house and a software like this may not be available in other institutions. Third, pathological assessment of tumor volume was impossible, because patients receive neoadjuvant chemotherapy before primary tumor resection. Note, however, that all included patients were still treatment-naive before MRI and FDG-PET were performed. Fourth, patients were dichotomized into groups according to the 200 mL primary tumor volume threshold [Citation1]. This cutoff was proposed by an international working group on the management of Ewing sarcoma [Citation1], which is based on several previous studies [Citation3–6]. However, whether 200 mL is the best cutoff for prognostic stratification remains under investigated. Nevertheless, it is nowadays commonly used in clinical practice. In addition, the present study also provided measures of inter- and in intraobserver traobserver agreement on a continuous scale. Fifth, FDG-PET was not performed in all patients, since this was at the discretion of the treating oncologist.
In conclusion, exact MRI-based simplified primary tumor volume measurements in Ewing sarcoma suffer from considerable inter- and intraobserver variability and moderately agree with MRI-based actual primary tumor volume measurements. However, inter- and intraobserver agreement of dichotomized MRI-based simplified primary tumor volume measurements (≤200 mL vs. >200 mL) is very good and generally matches MRI-based actual volume measurements. Finally, MRI-based primary tumor volume measurements poorly-moderately agree with and tend to be higher than the MATV.
Disclosure statement
The authors declare they have no potential conflict of interest.
References
- Gaspar N, Hawkins DS, Dirksen U, et al. Ewing sarcoma: current management and future approaches through collaboration. J Clin Oncol. 2015;33:3036–3046.
- Esiashvili N, Goodman M, Marcus RB, Jr. Changes in incidence and survival of Ewing sarcoma patients over the past 3 decades: Surveillance Epidemiology and End Results data. J Pediatr Hematol Oncol. 2008;30:425–430.
- Göbel V, Jürgens H, Etspüler G, et al. Prognostic significance of tumor volume in localized Ewing's sarcoma of bone in children and adolescents. J Cancer Res Clin Oncol. 1987;113:187–191.
- Jürgens H, Exner U, Gadner H, et al. Multidisciplinary treatment of primary Ewing's sarcoma of bone. A 6-year experience of a European Cooperative Trial. Cancer. 1988;61:23–32.
- Hense HW, Ahrens S, Paulussen M, et al. Factors associated with tumor volume and primary metastases in Ewing tumors: results from the (EI)CESS studies. Ann Oncol. 1999;10:1073–1077.
- Oberlin O, Deley MC, Bui BN, et al. French Society of Paediatric Oncology. Prognostic factors in localized Ewing's tumours and peripheral neuroectodermal tumours: the third study of the French Society of Paediatric Oncology (EW88 study). Br J Cancer. 2001;85:1646–1654.
- Murphey MD, Senchak LT, Mambalam PK, et al. From the radiologic pathology archives: Ewing sarcoma family of tumors: radiologic-pathologic correlation. Radiographics. 2013;33:803–831.
- Treglia G, Salsano M, Stefanelli A, et al. Diagnostic accuracy of 18F-FDG-PET and PET/CT in patients with Ewing sarcoma family tumours: a systematic review and a meta-analysis. Skeletal Radiol. 2012;41:249–256.
- Frings V, van Velden FH, Velasquez LM, et al. Repeatability of metabolically active tumor volume measurements with FDG PET/CT in advanced gastrointestinal malignancies: a multicenter study. Radiology. 2014;273:539–548.
- Kramer GM, Frings V, Hoetjes N, et al. Repeatability of quantitative whole-body 18F-FDG PET/CT uptake measures as function of uptake interval and lesion selection in non-small cell lung cancer patients. J Nucl Med. 2016;57:1343–1349.
- Boellaard R, Delgado-Bolton R, Oyen WJ, et al. European Association of Nuclear Medicine (EANM). FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42:328–354.
- Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1:307–310.
- Hess S, Alavi A, Basu S. PET-based personalized management of infectious and inflammatory disorders. PET Clin. 2016;11:351–361.
- Vilanova JC, Baleato-Gonzalez S, Romero MJ, et al. Assessment of musculoskeletal malignancies with functional MR imaging. Magn Reson Imaging Clin N Am. 2016;24:239–259.
