 ?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.ABSTRACT
Clinical Relevance
Vision disorders in children impact health-related quality of life, with early detection and intervention improving outcomes and educational performance. Eye health professionals should be aware of paediatric vision screening guidelines and their development to understand the components of local programmes and the differences in sensitivity and specificity between protocols.
Background
High-quality clinical practice guidelines (CPGs) for vision screening enable the early detection of common vision disorders; however, they require rigorous development to ensure optimal accuracy in detecting vision disorders, enabling timely interventions. This study evaluated the quality of available vision screening CPGs on vision screening of children in Australia and New Zealand.
Methods
A systematic search of academic databases, guideline databases, professional associations and Google search engines was conducted to identify relevant paediatric vision screening CPGs. Four independent reviewers used the Appraisal of Guidelines, Research and Evaluation (AGREE II) instrument to assess the quality of individual guidelines and scores were aggregated and reported as the percentage of the total possible score across the six AGREE II domains: scope and purpose, stakeholder involvement, rigour of development, clarity of presentation, applicability, and editorial independence.
Results
Initial 2,999 items were evaluated, with seven guidelines included. AGREE-II quality score agreement ranged from 43.3% to 95.8%. All guidelines scored >60.0% in the scope and purpose, however, most had poor scores of <26.5% in the rigour of development and <3.3% in editorial independence domains. All guidelines recommended screening using measures of habitual distance vision.
Conclusion
Of the guidelines developed for use in Australia and New Zealand, most guidelines scored poorly when assessed against the AGREE II tool, because of lack of editorial independence and rigour of development. Paediatric vision screening guidelines should prioritise systematic review of literature to inform practice and include statements regarding competing interests.
Introduction
Vision screening is an important public health initiative for detecting childhood vision conditions, particularly in vulnerable or economically disadvantaged communities, as it delivers economical vision assessments and improves outcomes for common childhood vision disorders.Citation1 Untreated vision problems in children can lead to amblyopia,Citation2 poor vision from abnormal visual experience early in life, reduce quality of life through to adulthood,Citation3 and negatively impact learningCitation4 and future earning potential.Citation3
Vision screening programs identify children with reduced vision and refer them for comprehensive assessment. The predominant causes of poor vision in childhood are refractive error, strabismus or amblyopia, however poor vision can be subsequent to ocular or systemic pathology.Citation2,Citation3 Whilst identifying and treating vision disorders is a cost-effective health intervention for improving productivity and quality of life,Citation3 there are challenges to effectively capturing a paediatric population and developing a screening programme that balances efficiency and cost-effectiveness with a high sensitivity for detecting paediatric eye conditions.
Most developed countries have some form of children’s vision screening or testing,Citation5 but there is limited consensus regarding evidence-based screening protocols that are effective in detecting common vision disorders in children.Citation6 Children’s vision screening programmes vary significantly in terms of their objectives, testing approach and personnel employed.Citation7
Given the importance of identifying children at risk of amblyopia and other common vision disorders, a paediatric vision screening programme must have high sensitivity, but also high specificity to ensure appropriate delivery without overwhelming health care resources. Therefore, protocols and procedures should be rigorously developed, using the appropriate evidence and stakeholder engagement. Clinical practice guidelines (CPGs), which are statements developed to help people make clinical, system- and policy-related decisions, can vary widely in content and quality.Citation8
The Appraisal of Guidelines, Research and Evaluation (AGREE) Collaboration established a generic instrument, the AGREE II tool, to assess the process of guideline development and quality.Citation5,Citation6,Citation9 With 23 items, AGREE II critically assesses published guidelines across six domains, and two global scores, looking at the overall quality of the guideline and its suitability for use with or without modification. The domains evaluate scope and purpose covering target population and overall aims; stakeholder involvement assessing the input from relevant groups; rigour of development analysing the links to the evidence and how recommendations were developed; clarity of presentation of the overall guideline; applicability including barriers to implementation; and editorial independence.Citation9
Individual items within each domain are outlined in Supplementary Table S1. Using a 7-point response scale, with a maximum score of 7 (strongly agree) and a minimum score of 1 (strongly disagree), the instrument differentiates between high quality and low-quality guideline content. There is a correlation between favourable outcomes of guideline implementation and positive ratings in multiple AGREE domains with every 10% change in the rigour of development domain predicting a statistical improvement in clinician endorsement.Citation9
Despite the prevalence of clinically significant refractive errors in children ranging from 13.0% to 30.0%,Citation10,Citation11 and amblyopia prevalence reported as between 1.9% and 6.1%Citation2,Citation10 there has been no systematic investigation of the quality of paediatric vision screening guidelines developed for in Australia and New Zealand. Therefore, the aim of this study was to review the quality, current evidence, and recommendations from published paediatric vision screening CPGs in Australia and New Zealand using the AGREE II tool.
Methods
Search strategy
A single, systematic literature search for published vision screening guidelines from Australia and New Zealand in academic databases (Medline, PsycINFO, Web of Science Core Collection, CCENTRAL, LILACs and ProQuest Scopus) was conducted in January 2023, using pre-defined selection criteria and search strategies outlined in and Supplementary Table S2. Additionally, guideline databases and optometry and ophthalmology professional society websites (Supplementary Table S3) were searched using the terms and criteria in Supplementary Table S3.Citation12
Table 1. Criteria used for systematic search and selection of the clinical practice guidelines (CPGs) and exclusion criteria.
Finally, a detailed Google search was undertaken to identify any relevant CPGs in public domains using specific search terms, as outlined in Supplementary Table S3.Citation13 The Google search was terminated for each dataset when 10 consecutive irrelevant results were found. All searches were limited to the last 15 years and only to English language CPGs, to ensure evidence was current.
Clinical practice guideline selection
Two authors (CH and RC) independently screened the titles and abstracts, where available, of items identified from the systematic literature, Google, professional society, and guideline database searches. CPGs that met the exclusion criteria outlined in were removed. CPGs that met title screening underwent independent full text screening by CH and RC.
Guidelines that did not meet the inclusion criteria outlined in , or were not based in Australia and New Zealand, were excluded. Any discrepancies were resolved by discussion between the two authors conducting the full text screening or, in the event a consensus could not be reached, by discussion with a third author (NA).
Quality appraisal and analysis
The quality of the included guidelines was assessed by four independent reviewers (CH, KK, JH and NM) using all 23 key items across the six domains and the two global ratings of the AGREE II tool. Differences of more than two points for any item were resolved through discussion within the appraisal group or, in the event a consensus could not be reached, by discussion with a fifth reviewer (NA).
A quality score was calculated for each of the six AGREE II domains by summing individual item scores and by scaling the total as a percentage of the maximum possible score for that domain,Citation14 with the minimum possible score for each domain of one and the maximum score for each domain of seven. Each domain score was calculated by obtaining the sum of scores from all reviewers, then producing an agreement percentage using the equationCitation9:
A good-quality guideline was defined as the one with ≥60.0% score for the rigour of development, the scope and purpose, and at least one other domain.Citation15
Data on recommendations were extracted from included CPGs using a standardised online form. Key vision screening recommendations including the tests used for screening, age of screening, referral criteria, individuals involved in the screening programme, how participants were notified about the outcomes, and the preferred referral pathways were extracted by one author (CH), and independently checked by a second author (NA).
Results
Search results
shows the number of items screened, assessed for eligibility, and included in the final review. Initially, 2,999 results were identified, with only 115 (3.8%) remaining after title and abstract screening. Systematic literature search of published vision screening guidelines yielded no results. All CPGs were found on guideline databases, professional association websites, or general websites. Full text screening further excluded 59 items due to relevance and 47 because they were not developed for vision screening in Australia or New Zealand, leaving nine potential CPGs.
Figure 1. Preferred reporting items for systematic literature search, search through guideline databases and professional association websites, and Google search.
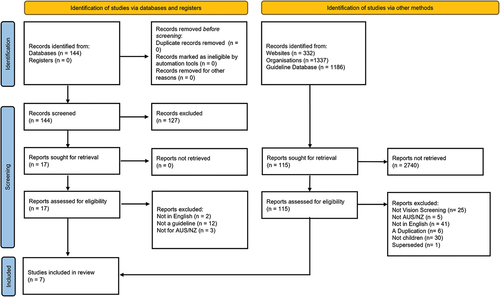
After a detailed appraisal, an additional two CPGs were excluded; one because it was not a vision screening guideline, and the other because it had been replaced by a newer version. This left seven guidelines selected for quality appraisal using the AGREE II tool: Western Australia (WA) Clinical Nursing Manual,Citation16 the Victorian Maternal and Child Health Service Practical Guidelines and Supplementary material Melbourne Initial Screening test,Citation17,Citation18 Vision 2020 National Framework for Vision Screening (Vision 2020), Lions Eye Health Program Children’s Vision Screening Screener Manual,Citation19 New Zealand Well Child Programme and supplementary material,Citation20,Citation21 the B4 School Check (B4 School Check)Citation22 and the National Vision and Hearing Screening Protocols (New Zealand National Protocols).Citation23
Guideline characteristics
The seven guidelines, four from Australia and three from New Zealand, were published between 2008 and 2023. Five guidelines were commissioned by government bodies, and two were published by charities with an interest in vision screening (). Two CPGs also included supplementary materials.
Table 2. Characteristics of the seven clinical practice guidelines (CPGs) selected for quality appraisal using the AGREE II tool.
CPG quality assessment using the AGREE II tool
Domain-specific results for all guidelines are summarised in and . The mean (± standard deviation) overall percentage score for all the guidelines was 69.0 ± 15.3%. The New Zealand Well Child and Supplementary material: Brief Evidence Reviews for the Well Child Tamariki Ora Programme CPG had the highest overall score of 95.8%, and the Lions Eye Health Program had the lowest overall score of 43.3%. The only guideline that met the accepted standard for a high-quality guideline with ≥60.0% in the scope and purpose, rigour of development and one other domainCitation15 was the New Zealand Well Child CPG.
Figure 2. Radar Chart showing individual AGREE II domain scores (%) for the seven selected clinical practice guidelines (CPGs).
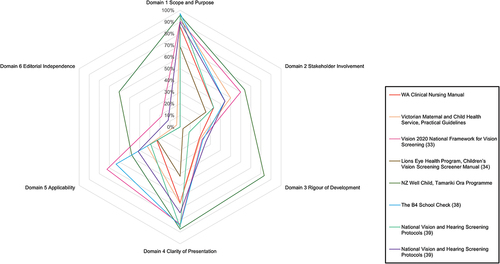
Table 3. Individual AGREE II domain scores (%) for the seven selected clinical practice guidelines (CPGs). Percentage scores for each domain were calculated by summing individual item scores and by reporting the total as a percentage of the maximum possible score for that domain.
All other reviewed CPGs scored poorly in the rigour of development domain with scores ≤22.4%. The editorial independence domain also scored poorly, with five CPGs scoring <3.3%. Conversely, the scope and purpose domain scored well, with all CPGs scoring >60.0%, and the clarity of presentation domain scored >60.0% for all but one CPG.Citation25
The final two questions of the AGREE II tool ask the assessors to rate the overall quality of the guideline and whether the assessor would recommend the guideline for use, with or without modification. Although no CPGs were considered unsuitable for use by the examiners, modifications were recommended in most of them. Only the New Zealand Well Child CPG had 100.0% agreement from examiners that it should be used, while 60.0% of examiners agreed the Melbourne Initial Screening Test CPG should be used without modification. There was agreement that the B4 School Check, the WA Clinical Nursing Manual CPG, and the Lions Eye Health CPGs could be used, but with modifications.
Results of the individual AGREE II domains
Domain 1 scope and purpose was the best scoring domain, with a mean ± SD score of 89.8% ± 9.9%. All CPGs clearly outlined the screening population, the conditions targeted and why vision screening is an important health intervention for these populations. Clarity of presentation (Domain 4) was the next best scoring domain, with a mean score of 73.5% ± 16.8% with six out of seven CPG scoring above 60.0% agreement. Overall, the CPGs scored poorly (44.4% ± 14.5%) in the stakeholder engagement domain due to lack of engagement with professional groups, and minimal consultation with schools, teachers, parents, and children on the screening programme.
Likewise, the mean score for the applicability of the guideline (Domain 5), which examined funding, barriers to guideline application, and monitoring or auditing criteria, was low (41.6% ± 20.1%) with five of the seven CPGs (WA Clinical Nursing Manual, Melbourne Initial Screening Test, Lions Eye Health, the B4 School Check, and the New Zealand National Protocols) achieving the lowest possible score for monitoring and auditing CPG protocols, recommendations or outcomes.
The two poorest scoring domains were rigour of development (Domain 3, mean score 25.5% ± 26.5%) and editorial independence (Domain 6, mean score 11.7% ± 22.5%). Domain 3 represents the largest section of the AGREE II tool, with questions addressing the evidence basis behind the development of the CPG, how evidence was selected and how guidelines will be reviewed and updated in the future. Most CPGs had limited protocols for updating the guideline and lacked linkages between protocol selection and the evidence base used. The editorial independence domain was the worst scoring of the six, as most guidelines, except for the New Zealand Well Child and Vision 2020, scored 0–3.3% for this section.
Clinical recommendations
summarises the key recommendations from each of the CPGs, all of which recommend conducting paediatric vision screening by measuring habitual distance vision. Most guidelines recommended that children should be referred for a comprehensive eye examination when acuity was 0.3 logMAR (Snellen equivalent 6/12) or worse, with the guidelines from Aotearoa New Zealand also identifying the need to rescreen children who had visual acuity of 6/9 (0.175 logMAR) in the worse eye and 6/6 (0.0 logMAR) in the better eye.
Table 4. Clinical recommendations from the selected clinical practice guidelines (CPGs).
Most CPGs recommended screening at 4–5 years of age, with some offering repeat screening at different ages. There was no consistency in recommendations between the visual acuity chart used, with some CPGs recommending letter-based charts, while others recommended picture optotypes. Only the Vision 2020, New Zealand Well Child, B4 School Check and New Zealand National Protocols specifically mentioned using vision charts with crowding.
Three of the seven guidelines also included the use of supplementary tests to screen for colour vision defects,Citation19,Citation23 strabismus,Citation16,Citation19 refractive errorCitation19 and ocular pathology.Citation16 All CPGs recommended that the vision screening be conducted by nurses or lay screeners with children who failed the vision screening referred to orthoptists, optometrists, or ophthalmologists.
Discussion
This study identified seven paediatric vision screening guidelines that have been developed for use in Australia and New Zealand, although none of these guidelines had been published in peer-reviewed journals. Most guidelines scored poorly when assessed against the AGREE II tool, primarily due to lack of editorial independence and poor rigour of development. Only the New Zealand Well Child CPG was considered as a good guideline as it had AGREE II scores >60.0% for the rigour of development, the scope and purpose, and at least one other domain.Citation25
Most of the guidelines reviewed in this study were rated as poor to moderate quality based on quantitative scores, which was also reflected in the appraisers' overall qualitative rating based on the final two holistic AGREE II questions. All assessors agreed that the only guideline suitable to use in its current form was the New Zealand Well Child check with all other guidelines requiring some modifications. In general, most guidelines could be improved by including a well-designed systematic literature review and justifying how the evidence base had been used to develop the CPG recommendations.
Furthermore, explicit information regarding the editorial board and guideline developers, as well as a conflict of interest disclosure were missing from most guidelines. The AGREE II tool recommends explicit discussion on any conflict of interest from members of the development group, and clear links between funding bodies and the guideline content to ensure clear editorial independence in the interests of best practice for patients.Citation9 Finally, further stakeholder engagement is warranted during the development process to ensure patient-centred care and applicability in a clinical setting.
Clinical practice guidelines are designed to optimise patient care and assist health care practitioners with clinical decision-making and should be informed by a systematic review of the evidence, therefore Domain 3 rigour of development and Domain 6 editorial independence are particularly critical during the CPG development process.Citation28 The rigour of development domain is regarded as having the most direct effect on CPG quality as it captures the need for a systematic review of the literature and the use of a clear evidence base for guideline development.Citation29
Alonso-Coella et al. noted that the leading deficiencies in this area of guideline development were lack of methodological expertise and a lack of information provided about the methods used to influence guideline development.Citation29 The authors therefore recommended involving librarians and guideline methodologists with expertise in methodological design, clinical epidemiology and statistics in guideline development, and to specifically include information about search strategies, literature selection processes and systems used to evaluate the quality of evidence as an addendum. The New Zealand Well Child CPG scored well in Domain 3 through the associated brief evidence review that was published by paediatric vision researchers in 2019,Citation21 6 years after the CPG was first developed, which provides the evidence base to support the screening protocol.
However, the lack of evidence base utilised in developing these paediatric vision screening CPGs may be reflected in the range of screening tests suggested. While all guidelines recommend measuring distance vision as the primary screening technique, the recommended acuity chart varies between different screening guidelines with both letter matching (Melbourne Initial Screening Test chart,Citation17,Citation18 Parr 4 mCitation20,Citation21,Citation23 and HOTVCitation24 chart) and picture optotype (Lea symbolsCitation16,Citation24) at either three or 4 m suggested.
Some guidelines (New Zealand Well Child, B4 School Check and New Zealand National Protocols) recommend a re-screen when vision was 6/9 in one eye and 6/6 in the fellow eye, with other guidelines recommending referral with VA worse than 6/12 (0.3 logMAR) or 3/5 (0.2 logMAR for Melbourne Initial Screening Test), other than the Vision 2020 with a referral criterion of 6/15.
Recent evidence by Findlay et al. found that removing restrictions around the two-line inter-eye difference improved the sensitivity of the screening, with no reduction in specificity for amblyopia risk factors.Citation27 Acute or urgent referral is recommended by the WA Clinical Nursing Manual at 6/19 and the Vision 2020 at 6/18. Almost all programmes other than the B4 School check, which suggests orthoptist referral, and the WA Clinical Nursing Manual, which recommends ‘medical practitioner’ referral, recommend referral to optometrists and ophthalmologists (). Only the Lions Eye Health programme recommended the use of autorefraction (Spot Vision Screener) as an adjunct test, despite the literature suggesting that handheld autorefractors demonstrate higher specificity and equivalent sensitivity to Lea symbols, when screening for significant refractive error and amblyopia risk factors.Citation30,Citation31
The WA Clinical Nursing Manual, Lions Eye Health and New Zealand National Protocols CPGs all recommend supplementary testing including cover test, corneal light reflex test, stereoacuity, and colour vision screening, however there is little published evidence to support the use of these procedures. While the inclusion of cover test (in screenings conducted by eye health professionals) or stereopsis (when the screening is conducted by lay screeners) improves the sensitivity of detecting strabismus,Citation30 measuring stereoacuity alone does not increase the sensitivity for detecting amblyopia, except when combined with autorefraction, which may increase the efficacy of screening protocols.Citation32
There was broad general agreement across all guidelines recommending screening children between 3 and 5 years of age, with the aim of identifying amblyopia and amblyopia risk factors such as strabismus, anisometropia and significant refractive error.Citation33 Screening children at younger ages was discussed only in the WA Clinical Nursing Manual, and repeat vision screening of school-aged children only referenced in the New Zealand Well Child and the Lions Eye Health CPGs. These CPG referral recommendations are broadly consistent with international best practice,Citation34 which shows there is limited evidence for screening children younger than 3 years,Citation34 and most children with vision worse than 0.2 logMAR require refractive correction.Citation35
All the seven CPGs recommend that vision screening should be conducted by nurses or lay screeners. Nurses could achieve high sensitivity and specificity with appropriate training.Citation36 The Vision in Preschoolers Study compared the performance of different vision screening tests in 3- to 5-year-old children by trained nurses and lay screeners, using the results of cycloplegic eye examinations administered by an ophthalmologist or optometrist as the gold standard.Citation30,Citation37
Although the sensitivities for all tests, including Lea symbols, autorefractor and stereoacuity testing, were marginally higher when administered by nurse screeners compared to lay screeners, these differences were not statistically different. The study concluded that nurse and lay screeners can achieve similar sensitivity to optometrists and ophthalmologists for detecting vision disorders in preschool children, when specificity is set at 0.90.Citation37 It is recommended that a collaborative effort of eye-care professionals, nurses and lay screeners may be an ideal and cost-effective approach for vision screening in paediatric populations.Citation5
A major strength of the study is that it included a comprehensive literature review encompassing guideline databases, professional association websites, and Google searches of all available paediatric vision screening CPGs in the last 15 years. Another strength of the study was the inclusion of four independent reviewers as recommended by the AGREE II protocol, which improves the reliability of scores in each individual domain.Citation38
A limitation of this study is that the AGREE II tool does not assess the quality of the guideline recommendations, and some studies have found that high AGREE II scores do not guarantee optimal CPG recommendations.Citation39 Furthermore, given the nature of guidelines, there may be other CPGs that met our inclusion criteria but were not published in literature or available publicly on the internet. Comparison of guidelines from other geographical areas may have strengthened the study, but due to the diverse nature of eye health care practices internationally including the different status of eye-care professionals, this study focused only on paediatric vision screening guidelines in Australia and New Zealand, which share similar health care systems through trans-Tasman mutual recognition.
Conclusion
This review identified seven paediatric vision screening CPGs developed in Australia and New Zealand, all of which were found on guideline databases, professional association websites or through a detailed Google search. The majority of these paediatric vision screening CPGs scored poorly on the AGREE II tool, primarily due to poor scores in the domains of editorial independence and rigour of development.
Conversely, most CPGs scored well on the scope and purpose and the clarity of presentation domains on the AGREE II tool. The highest scoring CPG was the New Zealand Well Child check, which scored >60.0% in six AGREE-II domains, including rigour of development. There were significant variations in the recommendations from these CPGs regarding referral criteria for comprehensive eye examination, the age of screening and the tests for screening refractive errors and other vision disorders.
These findings suggest the need for the development of a robust evidence-based paediatric vision screening guideline for use in Australian and Aotearoa New Zealand through improved editorial independence, increased rigour of development by incorporating current evidence and published literature and better stakeholder engagement.
Supplemental Material
Download PDF (190.2 KB)Acknowledgements
We would like to thank our librarian, Ms Shannon Brown, for helping us with the literature search.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Supplementary material
Supplemental data for this article can be accessed online at https://doi.org/10.1080/08164622.2024.2339276.
References
- Wahl MD, Fishman D, Block SS et al. A comprehensive review of state vision screening mandates for schoolchildren in the United States. Optom Vis Sci 2021; 98: 490–499. doi:10.1097/OPX.0000000000001686.
- Pai A-I, Rose KA, Leone JF et al. Amblyopia prevalence and risk factors in Australian preschool children. Ophthalmology 2012; 119: 138–144. doi:10.1016/j.ophtha.2011.06.024.
- Resnikoff S, Pascolini D, Mariotti SP et al. Global magnitude of visual impairment caused by uncorrected refractive errors in 2004. Health Organ Bull 2008; 86: 63–70. doi:10.2471/BLT.07.041210.
- White SLJ, Wood JM, Black AA et al. Vision screening outcomes of grade 3 children in Australia: differences in academic achievement. Int J Educ Res 2017; 83: 154–159. doi:10.1016/j.ijer.2017.03.004.
- Atowa UC, Wajuihian SO, Hansraj R. A review of paediatric vision screening protocols and guidelines. Int J Ophthalmol 2019; 12: 1194–1201. doi:10.18240/ijo.2019.07.22.
- Metsing IT, Hansraj R, Jacobs W et al. Review of school vision screening guidelines. Afr Vis Eye Health 2018; 77: 1–11. doi:10.4102/aveh.v77i1.444.
- Chen AH, Abu Bakar NF, Arthur P. Comparison of the pediatric vision screening program in 18 countries across five continents. J Curr Ophthalmol 2019; 31: 357–365. doi:10.1016/j.joco.2019.07.006.
- Shaneyfelt TM, Mayo-Smith MF, Rothwangl J. Are guidelines following guidelines? The methodological quality of clinical practice guidelines in the peer-reviewed medical literature. JAMA 1999; 281: 1900–1905. doi:10.1001/jama.281.20.1900.
- Brouwers MC, Kho ME, Browman GP et al. AGREE II: advancing guideline development, reporting and evaluation in health care. J Clin Epidemiol 2010; 63: 1308–1311. doi:10.1016/j.jclinepi.2010.07.001.
- Findlay R, Black J, Anstice NS et al. The prevalence of refractive error and visual impairment among New Zealand children in a community with significant socioeconomic disadvantage: is current preschool vision screening effective? N Z Med J 2020; 133: 33–41.
- French AN, Morgan IG, Burlutsky G et al. Prevalence and 5- to 6-year incidence and progression of myopia and hyperopia in Australian schoolchildren. Ophthalmology 2013; 120: 1482–1491. doi:10.1016/j.ophtha.2012.12.018.
- Lingham G, Thakur S, Safi S et al. A systematic review of clinical practice guidelines for childhood glaucoma. BMJ Open Ophthalmol 2022; 7: 1–8. doi:10.1136/bmjophth-2021-000933.
- Adams J, Hillier-Brown FC, Moore HJ et al. Searching and synthesising ‘grey literature’ and ‘grey information’ in public health: critical reflections on three case studies. Syst Rev 2016; 5: 164–174. doi:10.1186/s13643-016-0337-y.
- The AGREE Enterprise. The AGREE II instrument. AGREE Trust Website. [accessed 2023 May 19]. http://www.agreetrust.org.
- Gyawali R, Toomey M, Stapleton F et al. Eyes on guidelines: evaluating the quality of diabetic eye disease clinical practice guidelines [abstract]. Invest Ophthalmol Vis Sci 2021; 62: 3522.
- Government of Western Australia Child and Adolescent Health Service. Vision and eye health guideline. [accessed 2023 May 19]. https://cahs.health.wa.gov.au/~/media/HSPs/CAHS/Documents/Community-Health/CHM/Vision-and-eye-health.pdf?thn=0
- Victorian State Government. Melbourne Initial Screening Test (MIST). [accessed 2023 May 19]. https://www.education.vic.gov.au/Documents/childhood/professionals/health/MIST_info-for-nurses%28Oct17%29.pdf
- Victoria State Govenrnment. Department of education and early childhood development. Maternal and Child health service: practice guidelines 2009. [accessed 2023 May 19]. https://www.education.vic.gov.au/Documents/childhood/professionals/support/mchpracguidel.pdf
- Lions Eye Health Program – Australia. Lions eye health program, children’s vision screening, screener manual. [accessed 2023 May 19]. https://lehp.org.au/wp-content/uploads/2022/11/SCREENER-MANUAL-CVSP-July-2022-FINAL-APPROVED1.pdf
- Ministry of Health. Well Child/Tamariki ora programme practitioner handbook: supporting families and whānau to promote their child’s health and development – Revised 2014. Wellington: Ministry of Health. [accessed 2023 May 19]. https://www.health.govt.nz/system/files/documents/publications/wcro-practitioner-handbook-october-2015-updates-v2.pdf
- Hamm LRF, Black J. Vision screening in infancy and childhood. Brief Evidence Revs Well Child Tamariki Ora Programme 2019; 168–216.
- Ministry of Health. The B4 school check: a handbook for practitioners. Wellington: Ministry of Health. [accessed 2023 May 19]. https://www.moh.govt.nz/notebook/nbbooks.nsf/0/9de5d356a2c8f2cacc2577140005fad9/$FILE/b4sc-practitionershandbook-march2010.pdf
- Ministry of Health. National vision and hearing screening protocols: revised 2021. Wellington: Ministry of Health. [accessed 2023 May 19]. https://www.health.govt.nz/system/files/documents/publications/national-vision-and-hearing-screening-protocols-jan22.pdf
- Vision 2020. National Framework for vision screening for 3.5–5-year-olds. [accessed 2023 May 19]. https://www.vision2020australia.org.au/wp-content/uploads/2022/10/National-Framework-for-Vision-Screening_final.pdf
- Ciapponi A, Tapia-López E, Virgilio S et al. The quality of clinical practice guidelines for preoperative care using the AGREE II instrument: a systematic review. Syst Rev 2020; 9: 159–174. doi:10.1186/s13643-020-01404-8.
- Anstice NS, Jacobs RJ, Simkin SK et al. Do picture-based charts overestimate visual acuity? Comparison of Kay pictures, Lea symbols, HOTV and Keeler logMAR charts with Sloan letters in adults and children. PLOS ONE 2017; 12: e0170839. doi:10.1371/journal.pone.0170839.
- Findlay R, Black J, Goodman L et al. Diagnostic accuracy of the Parr vision test, single crowded Lea symbols and spot vision screener for vision screening of preschool children aged 4-5 years in Aotearoa/New Zealand. Ophthalmic Physiol Opt 2021; 41: 541–552. doi:10.1111/opo.12816.
- Hoffmann-Eßer W, Siering U, Neugebauer EAM et al. Guideline appraisal with AGREE II: online survey of the potential influence of AGREE II items on overall assessment of guideline quality and recommendation for use. BMC Health Serv Res 2018; 18: 143–151. doi:10.1186/s12913-018-2954-8.
- Alonso P, Irfan A, Solà I et al. The quality of clinical practice guidelines over the last two decades: a systematic review of guideline appraisal studies. Qual Saf Health Care 2010; 19: 1–7. doi:10.1136/qshc.2010.042077.
- Kulp MT. Findings from the vision in preschoolers (VIP) study. Optom Vis Sci 2009; 86: 619–623.
- Kapoor V, Shah SP, Beckman T et al. Community based vision screening in preschool children; performance of the spot vision screener and optotype testing. Ophthalmic Epidemiol 2022; 29: 417–425. doi:10.1080/09286586.2021.1962918.
- The Vision in Preschoolers Study Group. Does assessing eye alignment along with refractive error or visual acuity increase sensitivity for detection of strabismus in preschool vision screening? Invest Ophthalmol Vis Sci 2007; 48: 3115–3125. doi:10.1167/iovs.06-1009.
- US Preventative Services Task Force. Vision screening for children 1 to 5 years of age: US preventive services task force recommendation statement. Pediatrics 2011; 127: 340–346. doi:10.1542/peds.2010-3177.
- Cotter SA, Cyert LA, Miller JM et al. Vision screening for children 36 to <72 months: recommended practices. Optom Vis Sci 2015; 92: 6–16. doi:10.1097/OPX.0000000000000429.
- McCullough S, Saunders K. Visual profile of children who passed or failed the UK school vision screening protocol. Br Ir Orthopt J 2019; 15: 36–46. doi:10.22599/bioj.121.
- Mathers M, Keyes M, Wright M. A review of the evidence on the effectiveness of children’s vision screening. Child Care Health Dev 2010; 36: 756–780. doi:10.1111/j.1365-2214.2010.01109.x.
- The Vision in Preschoolers Study Group. Preschool vision screening tests administered by nurse screeners compared with lay screeners in the vision in preschoolers study. Invest Ophthalmol Vis Sci 2005; 46: 2639–2648. doi:10.1167/iovs.05-0141.
- Shrout PE, Fleiss JL. Intraclass correlations: uses in assessing rater reliability. Psychol Bull 1979; 86: 420–428. doi:10.1037/0033-2909.86.2.420.
- Vlayen J, Aertgeerts B, Hannes K et al. A systematic review of appraisal tools for clinical practice guidelines: multiple similarities and one common deficit. Int J Qual Health Care 2005; 17: 235–242. doi:10.1093/intqhc/mzi027.
