Abstract
Calcific uremic arteriolopathy (or calciphylaxis) is a severe complication of renal failure characterized by subcutaneous calcification of the small arteries and tissue necrosis. We describe the case of a woman receiving continuous cycling peritoneal dialysis with calciphylaxis involving upper and lower extremities. After intolerance of intravenous sodium thiosulfate and limited intravenous access options, we administered sodium thiosulfate intraperitoneally and quantitated the amount of extra calcium removed. Intraperitoneal administration of sodium thiosulfate was well tolerated and led to removal of extra calcium with peritoneal dialysis.
INTRODUCTION
Calcific uremic arteriolopathy (CUA), also known as calciphylaxis, is a severe and progressive ischemic necrosis of the skin and subcutis primarily seen in patients with end-stage renal disease, and is associated with calcium deposition in the wall of subcutaneous small arteries and arterioles.Citation[1] The pathogenesis of CUA is poorly understood and its treatment is largely empirical and anecdotal. Recent studies have emphasized the crucial role of a multidisciplinary therapeutic approach focusing on the correction of the underlying abnormalities of the plasma calcium and phosphorus concentrations (using non-calcium containing phosphate binders), local wound care with debridement of necrotic tissues, and aggressive treatment of infectious complications.Citation[2],Citation[3]
The prognosis remains poor: the mortality rate is as high as 80% and is principally due to secondary infection of the ulcers, and sepsis.Citation[4] One of the more recently suggested therapies is the use of sodium thiosulfate to increase the solubility of soft tissue calcium deposits.
Thiosulfate binds to calcium and forms a highly soluble calcium thiosulfate salt.Citation[5] In a reported case of CUA in a peritoneal dialysis patient, sodium thiosulfate was delivered via an indwelling central catheter.Citation[6] We report the administration of sodium thiosulfate intraperitoneally (i.p.) in a patient on continuous cycling peritoneal dialysis (CCPD) with severe CUA.
CASE
A 26–year-old white female diagnosed with focal segmental glomerulosclerosis at age 7, with two failed kidney transplants, had been on CCPD for the past 4 years. She underwent a subtotal parathyroidectomy a year and a half prior to her presentation. The patient was evaluated at our institution with a 6-week history of violaceous mottling of the skin followed by painful ulcerations on proximal left arm and right lateral and left inner thigh. Two skin biopsies at outside institutions were non-diagnostic and reportedly revealed epidermal necrosis. The two biopsy sites demonstrated delayed healing and eschar formation. Skin biopsies were negative for oxalate crystals, and serological work-up was negative for ANA, ANCA, C3, C4, cryoglobulin, HbsAg, and anti-HCV antibody. Laboratory results on presentation included serum calcium 8.6 mg/dL, albumin 2.7 g/dL, corrected calcium 9.6 mg/dL, phosphorus 5.5 mg/dL, calcium x phosphorus product 53, ionized calcium 1.22 mmol/L, intact PTH 89 pg/mL, BUN 22 mg/dL, serum creatinine 8.1 mg/dL, Hb10.2 g/dL. The patient was not on any vitamin D products. The only phosphate binder was sevelamer. The patient was treated at our institution for severe upper and lower extremity CUA ulcers with CCPD using low-calcium (2.5 meq/L) dialysate (Dianeal, Baxter Healthcare Corporation, Deerfield, IL, USA), wound care, parenteral antibiotics, and thrice weekly intravenous sodium thiosulfate administered through a central indwelling catheter. There was improvement in skin ulcerations, per family and plastic surgeon's report. However, sodium thiosulfate was discontinued after 2 months due to nausea and vomiting that coincided with its administration.
Four months later, the patient presented with more extensive CUA. There was also a history of noncompliance with medications and peritoneal dialysis. Admission laboratory data were as follows: BUN 118 mg/dL, creatinine 17.7 mg/dL, serum calcium 8.7 mg/dL, albumin 1.8 g/dL, corrected serum calcium 10.5 mg/dL, phosphorus 6.5 mg/dL, calcium x phosphorus product 68. CCPD with low-calcium Dianeal, aggressive wound care with surgical debridement, and antibiotics, were started. Lanthanum carbonate was added to sevelamer to control hyperphosphatemia. Access to central veins had become difficult due to prior catheter-associated thrombosis. It was then decided to administer sodium thiosulfate i.p. A dose of 25 g of sodium thiosulfate was added to 2L of low-calcium Dianeal in the long day dwell. This was repeated every other day. On the remaining days, 2L of low-calcium Dianeal alone was used in the long day dwell. Calcium concentration was measured in the peritoneal effluent following the long dwell containing sodium thiosulfate, as well as at 3, 6, and 9 hours on the cycler. Similar measurements were performed on the days that she did not receive i.p. sodium thiosulfate. Two sets of measurements were obtained following the long day dwell with and without sodium thiosulfate (). The total duration of i.p. sodium thiosulfate treatment was 3 months.
Table 1 Calcium concentration in peritoneal effluent following the long day dwell with and without sodium thiosulfate
With the above measures, the median serum phosphorus was 3.5 mg/dL and median ionized calcium was 1.15 mmol/L (range 1.09–1.24 mmol/L). Despite the multifaceted approach to treatment, CUA lesions in our patient progressed, leading to sepsis and death.
RESULTS
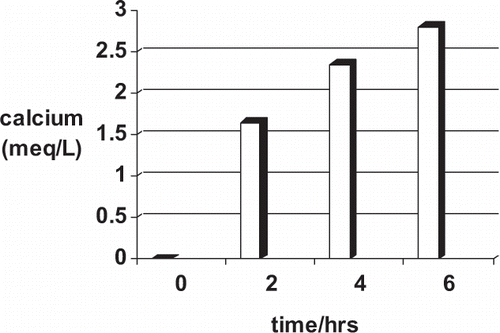
From our measurements, i.p. sodium thiosulfate increased calcium removal by an average of 0.65 meql/L (13 mg/L) as opposed to 0.05 meq/L (1 mg/L) with low-calcium Dianeal alone (). We also introduced a 6-hour dwell with 0.9% normal saline in order to maximize the calcium concentration gradient across the peritoneal membrane and thus increase calcium removal (). Calcium concentration in i.p. saline increased as a function of dwell time.
DISCUSSION
One of the most recent advances in the treatment of CUA is the use of inorganic salt sodium thiosulfate pentahydrate. This compound has been used in the treatment of recurrent calcium stones, and in hemodialysis patients with tumoral calcinosis.Citation[5],Citation[6] Sodium thiosulfate binds to calcium to form calcium thiosulfate salt, which is 250–100,000 times more soluble than other calcium salts such as oxalate or phosphate.Citation[5] It is thought that the formation of calcium thiosulfate promotes dissolution of calcium deposits and inhibits precipitation of calcium salts. Cicone et al.Citation[7] described the efficacy of intravenous sodium thiosulfate in a patient with CUA on continuous ambulatory peritoneal dialysis. Guerra et al.Citation[8] reported a case of CUA in a pre-dialysis patient successfully treated with intravenous sodium thiosulfate in conjunction with continuous venovenous hemofiltration to allow for continuous removal of solubilized calcium deposits.
To our knowledge, sodium thiosulfate has not been administered i.p. for the treatment of CUA. It has potential advantages over the intravenous administration for patients on PD. First, it obviates the need for placement of a central indwelling catheter, thus preventing potential complications such as infection and venous thrombosis. Second, it can be self-administered with the long day dwell, thus obviating additional visits to the chronic dialysis center. Moreover, it did not produce any precipitate when mixed with PD solution, and its instillation into the peritoneum was well tolerated. The long-term effect of i.p. sodium thiosulfate on transport characteristics of the peritoneal membrane are not known.
CONCLUSION
In summary, we report a patient on peritoneal dialysis with severe calciphylaxis and difficult intravenous access who we treated with i.p. administered sodium thiosulfate. To our knowledge, this is the first report on i.p. administration of sodium thiosulfate. This approach was well tolerated and led to 0.65 meq/L (13 mg/L) of extra calcium removed with low-calcium Dianeal. This represents a small, but significant route of calcium removal in calciphylaxis.
REFERENCES
- Llach F. Calcific uremic arteriolopathy (calciphylaxis): an evolving entity?. Am. J. Kidney Dis 1998; 32: 514–518, [PUBMED], [INFOTRIEVE], [CSA]
- Llach F. The evolving clinical features of calciphylaxis. Kidney Int 2003; 63(Suppl 85)S122–S124, [CSA], [CROSSREF]
- Don BR, Chin AI. A strategy for the treatment of calcific uremic arteriolopathy (calciphylaxis) employing a combination of therapies. Clin. Nephrol 2003; 69: 463–470, [CSA]
- Wilmer WA, Magro CM. Calciphylaxis: emerging concepts in prevention, diagnosis and treatment. Sem. in Dial 2002; 15: 172–186, [CSA], [CROSSREF]
- Yatzidis H. Successful sodium thiosulphate treatment for recurrent calcium urolithiasis. Clin. Nephrol 1985; 23: 63–67, [INFOTRIEVE], [CSA]
- Kyriakopoulos G, Kontogiann K. Sodium thiosulfate treatment of tumoral calcinosis in patients with end-stage renal disease. Renal Fail 1990; 2: 213–219, [CSA]
- Cicone JS, Petronis JB, Embert CD, Spector DA. Successful treatment of calciphylaxis with intravenous sodium thiosulfate. Am. J. Kidney Dis 2004; 43: 1104–1108, [INFOTRIEVE], [CSA]
- Guerra G, Shah RC, Ross EA. Rapid resolution of calciphylaxis with intravenous sodium thiosulfate and continuous venovenous hemofiltration using low calcium replacement fluid: case report. Nephrol. Dial. Transplant 2005; 20: 1260–1262, [INFOTRIEVE], [CSA], [CROSSREF]