Abstract
Kawasaki disease is a vasculitis of medium-sized vessels and the most common cause of acquired heart defects in the United States. Although its etiology is unclear, an infectious trigger has been theorized, which has been highlighted by the recent pandemic. We present a case of a 17-month-old-girl with concurrent Kawasaki disease and non–SARS-CoV2 coronavirus infection and a sequela of onychomadesis.
The etiology of Kawasaki disease (KD) is largely unknown; however, several theories exist such as the environmental toxin theory, autoimmune pathogenesis theory, superantigen theory, and others.Citation1 The theory of an infectious etiology is supported by KD’s seasonality, geographic spikes, array of clinical findings that may resolve spontaneously, and often a preceding respiratory illness.Citation1 Furthermore, intracytoplasmic inclusion bodies associated with viral-like particles were identified throughout varying cell types in KD patients on postmortem examination, suggesting an RNA viral etiology.Citation2 Another study comparing children with KD to age- and sex-matched healthy controls found overall increased positivity of viral polymerase chain reaction testing.Citation3 Of the viruses tested, enterovirus, adenovirus, human rhinovirus, and coronavirus all occurred at higher rates in children with KD.Citation3 Through this case report, we explore coronavirus’ association with KD and a unique sequela of onychomadesis.
CASE DESCRIPTION
A 17-month-old previously healthy Hispanic girl presented to the emergency department with 4 days of fever and a widespread exanthematous eruption. The patient’s mother denied sick contacts, recent travel, or animal exposure. Her vaccinations were up to date. At the time of presentation, her temperature was 39.2°C. She had erythematous, blanchable plaques on the trunk and extremities. Her cheeks were erythematous with desquamative changes. Her lips had erosions with overlying hemorrhagic crust. Trace erythema without edema was noted on the palms and soles. The conjunctivae were injected. Cervical lymphadenopathy was noted on the right, but no node was larger than 1.5 cm. The complete blood count was significant for a hemoglobin of 10.7 g/dL. C-reactive protein and erythrocyte sedimentation rate were both elevated. Microscopic urinalysis showed pyuria. The urine culture was negative. A respiratory viral panel detected coronavirus. A chest radiograph showed bronchial wall thickening consistent with a viral respiratory infection.
The differential diagnosis included incomplete KD, Mycoplasma pneumonia-induced rash and mucositis, and a viral exanthem. With persistent fever, the patient met American Heart Association criteria () for KD on day 2 of hospitalization and promptly received intravenous immunoglobulin and high-dose aspirin. Echocardiography was pursued on day 4, which showed a small left anterior descending artery aneurysm (2.3 mm, Z-score = 2.77) and right coronary artery dilation (2.3 mm, Z-score = 2.16). Two days later, echocardiography showed worsening of the affected arteries (). She received one infusion of infliximab on day 7 and was discharged on day 10 on enoxaparin and aspirin.
Figure 1. Echocardiogram demonstrating aneurysms (arrows). (a) Left anterior descending artery (3.2 mm, Z-score = 6.91). (b) Right coronary artery (3.1 mm, Z-score = 4.93).
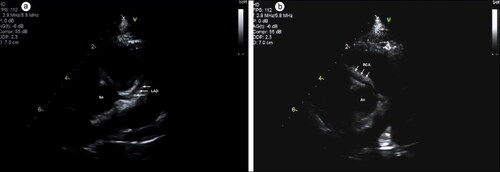
Table 1. American Heart Association diagnostic criteria for Kawasaki diseaseCitation4
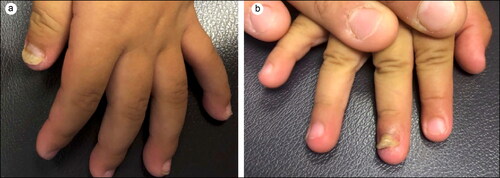
At 4-month follow-up, nail plate separation affecting the left first digit and right third digit of the hands was noted (). Echocardiography showed regression of the affected arteries to mild dilation. She will require lifelong aspirin and assessment for ischemia every 2 to 5 years based on American Heart Association 2017 KD guidelines.Citation4
DISCUSSION
There has been renewed interest in KD’s proposed link to coronavirus infection since the emergence of SARS-CoV2. A French study found a 497% increase of KD hospitalizations in April 2020, 2 weeks following the region’s first peak of the SARS-CoV2 epidemic.Citation5 Similar findings were found in New York, Italy, and the United Kingdom that all described an inflammatory syndrome with overlapping features of KD following spikes of SARS-CoV2.Citation6–8 In response to this, in May 2020, the US Centers for Disease Control and Prevention issued a health advisory for a multisystem inflammatory syndrome in children related to the pandemic.Citation9 Our patient was not tested for SARS-CoV2, since it was not believed to be in circulation in the region at the time of presentation. Retrospective verification of her strain was not possible, and SARS-CoV2 antibody testing was not sought. Ultimately, coronavirus’ association with KD is not well understood.
Although onychomadesis has been previously reported following KD,Citation10 this is the first time onychomadesis has been reported in association with coronavirus-associated KD. Onychomadesis results from complete toxicity of the nail matrix and presents with shedding of the nail or a proximal sulcus that splits the nail plate into two parts.Citation11 Onychomadesis has been associated with hand, foot, and mouth disease and typically occurs within 2 months of the infection.Citation11 A recent report of a child who presented with onychomadesis was found to have a previously missed KD diagnosis upon further investigation.Citation12 Our report suggests that physicians should be suspicious of undiagnosed KD and potentially coronavirus in a patient presenting with onychomadesis of unknown cause.
Speculation has been made on a potential surge of delayed or missed KD diagnoses due to the recent pandemic and fear of seeking in-person care.Citation13 Our report adds to the current body of evidence suggesting a link between coronavirus and KD and highlights the sequela of onychomadesis that could be used to identify a child who went undiagnosed.
- Rowley AH. Kawasaki disease: novel insights into etiology and genetic susceptibility. Annu Rev Med. 2011;62:69–77. doi:https://doi.org/10.1146/annurev-med-042409-151944.
- Rowley AH, Baker SC, Shulman ST, et al. Ultrastructural, immunofluorescence, and RNA evidence support the hypothesis of a “new” virus associated with Kawasaki disease. J Infect Dis. 2011;203(7):1021–1030. doi:https://doi.org/10.1093/infdis/jiq136.
- Chang L-Y, Lu C-Y, Shao P-L, et al. Viral infections associated with Kawasaki disease. J Formos Med Assoc. 2014;113(3):148–154. doi:https://doi.org/10.1016/j.jfma.2013.12.008.
- McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation. 2017;135(17):e927–e999. doi:https://doi.org/10.1161/CIR.0000000000000484.
- Ouldali N, Pouletty M, Mariani P, et al. Emergence of Kawasaki disease related to SARS-CoV-2 infection in an epicentre of the French COVID-19 epidemic: a time-series analysis. Lancet Child Adolesc Health. 2020;4(9):662–668. doi:https://doi.org/10.1016/S2352-4642(20)30175-9.
- Capone CA, Subramony A, Sweberg T, et al. Characteristics, cardiac involvement, and outcomes of multisystem inflammatory syndrome of childhood associated with severe acute respiratory syndrome coronavirus 2 infection. J Pediatr. 2020;224:141–145. doi:https://doi.org/10.1016/j.jpeds.2020.06.044.
- Riphagen S, Gomez X, Gonzalez-Martinez C, Wilkinson N, Theocharis P. Hyperinflammatory shock in children during COVID-19 pandemic. Lancet. 2020;395(10237):1607–1608. doi:https://doi.org/10.1016/S0140-6736(20)31094-1.
- Verdoni L, Mazza A, Gervasoni A, et al. An outbreak of severe Kawasaki-like disease at the Italian epicentre of the SARS-CoV-2 epidemic: an observational cohort study. Lancet. 2020;395(10239):1771–1778. doi:https://doi.org/10.1016/S0140-6736(20)31103-X.
- Centers for Disease Control and Prevention. Multisystem inflammatory syndrome in children (MIS-C) associated with coronavirus disease 2019 (COVID-19). https://emergency.cdc.gov/han/2020/han00432.asp. Published May 14, 2020. Accessed August 15, 2020.
- Pilapil VR, Quizon DF. Nail shedding in Kawasaki syndrome. Am J Dis Child. 1990;144(2):142–143. doi:https://doi.org/10.1001/archpedi.1990.02150260020012.
- Tosti A. Nail Disorders. St. Louis, MO: Elsevier; 2018.
- Banday AZ, Neelam H, Jindal AK. Spatio-temporal evolution of onychomadesis in a child with missed Kawasaki disease. Clin Rheumatol. 2020;39(7):2237–2238. doi:https://doi.org/10.1007/s10067-020-05100-9.
- Harahsheh AS, Dahdah N, Newburger JW, et al. Missed or delayed diagnosis of Kawasaki disease during the 2019 novel coronavirus disease (COVID-19) pandemic. J Pediatr. 2020;222:261–262. doi:https://doi.org/10.1016/j.jpeds.2020.04.052.