ABSTRACT
The establishment of nucleic acid amplification technology and functional nucleic acid aptamers promote the development of detection techniques. In particular, the detection displayed promise for broader application, as well as improved specificity ad higher sensitivity when combined with immunoassays (IA). This review introduced the novel immuno-nucleic acid (INA) cooperative detection technology, which is currently applied in the field of food safety for the recognition of residues of veterinary drugs and pesticides, biological toxins, pathogenic microorganisms, genetically modified foods (GMF), environmental pollutants, and more. Ultimately, the prospects for future development prospects, as well as the potential advantages of this technology were presented.
Graphic. Magnifying glass, Taiji, Circular ring, Octagonal flowers, and Seven circular illustrations. Magnifying glass represents immunoassay (IA). Nucleic acid amplification and functional nucleic acid aptamers represent the yellow part and the green part of Taiji, respectively. The light blue circular ring is written “more sensitive, more specific, more rapid, low cost, high throughput” shows advantages of immunoassays combined nucleic acid amplification and functional nucleic acid aptamers. Each extended petals of octagonal flowers represent the application of immuno-nucleic acid cooperative detection technology for food safety in pesticide residues, veterinary drug residues, toxin, pathogenic microorganisms, genetically modified food, food additive, as well as allergen and harmful substance, respectively. Finally, the seven circular illustrations display the most representative elements in seven detection fields above intuitively.
GRAPHICAL ABSTRACT

1. Introduction
1.1. IA
An IA is a biochemical analysis method based on the efficient selection and specific binding ability between antibodies and antigens or haptens and plays a substantial role in food safety and clinical assay detection (Yang et al., Citation2018). However, the traditional methods used for the qualitative or quantitative detection of IA, such as the enzyme-linked immunosorbent assay (ELISA) (Choi et al., Citation2020), are only based on the reaction after the interaction between the enzyme-labeled antigen (antibody) and the known antibody (antigen) during the fixed phase, limiting the amplification effect of the signal and cannot be effectively applied for detection requiring high precision and sensitivity.
1.2. The nucleic acid amplification technology and functional nucleic acid aptamers
Nucleic acid amplification is an essential technique in the field of molecular biology and can be divided into two categories based on the temperature requirements of the reactions. Traditional nucleic acid amplification is primarily based on variable temperature amplification, such as the polymerase chain reaction (PCR), which uses DNA molecules as a template to provide the DNA polymerase, the dNTP, the primers of the fragment, and the temperature conditions required for the three stages of “derivation-gradual extension” in DNA replication in vivo. However, the dependence on professional equipment and the repeated temperature regulation lead to a prolonged detection time. Over the last decade, the development of nucleic acid isothermal amplification technologies further simplify requirements for instrumentation, and show the better application potential in fast detection (Tian et al., Citation2019). According to the principles and characteristics of isothermal amplification, the available technologies can be divided into rolling circle amplification (RCA), loop-mediated isothermal amplification (LAMP), nucleic acid sequence-based amplification (NASBA) and more (Ma, Citation2013).
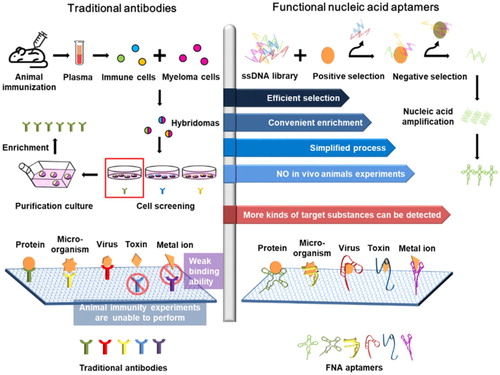
A nucleic acid aptamer is a short, single-stranded sequence composed of dozens of deoxyribonucleotides or ribonucleotides (Xiao et al., Citation2019). These aptamers can be folded to form the specific structures that bind with target molecules, such as small molecules, ions, and protein molecules (Xu et al., Citation2017). Owing to larger-scale production capacity, lower differences between production batches, lower immunogenicity, lower cytotoxicity, and stability during long-term storage in vitro, aptamers can replace or supplement natural antibody functionality and create new opportunities and breakthroughs in signal recognition and detection () (Cui et al., Citation2018).
1.3. The advantages and significance of INA cooperative detection in food safety
Food is necessary for human survival. Therefore, societal development, material production, processing, and transportation of food are continually evolving. However, since harmful factors and pollutants in food can enter the body to the detriment of human health, food safety is attracting significant attention in current research (Shen et al., Citation2010; Wei, Citation2018).
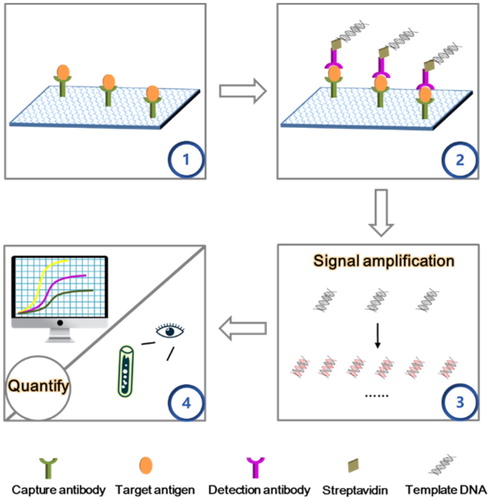
The importance of food safety necessitates the development of effective IAs. Various DNA amplification technologies and functional nucleic acids can improve the process of signal amplification () and specific recognition, meeting the requirements of IAs in various fields.
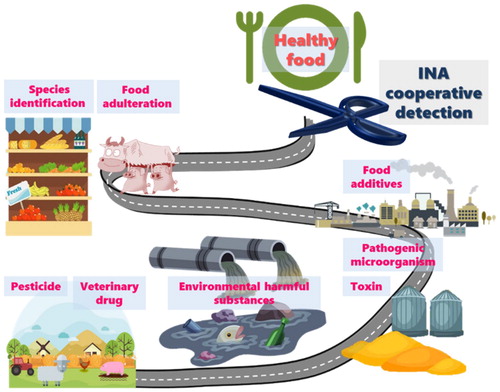
This study focuses on the application process of INA cooperative detection in recognising drug residues, foodborne toxins, pathogenic microorganisms, GMF, and environmental pollutants to selectively implement these detection techniques in the area of food safety in the future.
2. Application of INA cooperative detection
2.1. Pesticide residue
As the global population continues to grow, food demand is increasing daily. During the production of more crops to meet this demand, a variety of pesticides are widely used, leading to the problem of pesticide residue (Liao et al., Citation2019).
Here, organophosphorus (OP) and organic sulfur are the most commonly used pesticides in crops, but their high toxicity can severely damage human health. Therefore, Xiang et al. developed an aptamer-based ELISA to identify isocarbophos residues, effectively avoiding the restrictions posed by natural antibodies, such as strict storage and detection limitations with a limit of detection (LOD) of 0.05 ng/mL, and is more sensitive than the traditional ELISA (Xiang et al., Citation2019). For the assay in one sample of multifarious pesticide residues, Hua et al. isolated the phage-borne peptide from a cyclic eight-peptide phage library, using two simple and rapid immuno-LAMP without instrumental, LOD of detections for eight OP pesticides reached up to ng/mL (Hua et al., Citation2014).
2.2. Veterinary drug residue
The food requirements of consumers have gradually changed from quantity to quality and from plant-based to animal-based due to the improvement in living standards. Therefore, the detection of banned drugs such as β-agonist, antibiotics, and hormones used during the breeding and processing for animal-derived foods, has also become a problem that cannot be ignored (Chen et al., Citation2010).
For the detection of antibiotics, chloramphenicol (CAP) has become one of the most widely used drugs due to its low production cost and the high treatment efficiency of bacterial infections in livestock and poultry. Various countries have established a minimum required performance limit (MRPL) value or even banned the use for CAP to reduce the risk of its abuse as a drug. Therefore, Duan et al. developed a recognition element in the aptamer-based biosensors for the efficient detection of CAP residues in animal-sourced food, which eliminated the need for samples of high purity, as well as the high detection cost of the traditional ELISA. Furthermore, via CAP, the suitable aptamers provide an alternative rapid detection technology that is sensitive and economical (Duan et al., Citation2016). Tao et al. used immune magnetic beads to capture the anti-CAP monoclonal antibody linking a reporter DNA, and then amplified the reaction signal via real-time PCR. Using immuno-PCR (iPCR), sensitive detection and quantification of CAP were performed on a milk sample in a concentration range of 0.001 μg/L to 0.11 μg/L (Tao et al., Citation2014).
For the detection of β-agonist, Dai et al. established a real-time iPCR (RT-iPCR) technique to detect ractopamine via the signal amplification of quantitative fluorescence PCR, which increased the LOD to 0.0039 ng/mL and two orders of magnitude more sensitive than ELISA (Dai, Citation2015). Lei et al. proposed a novel pre-assembled DNA immunoprobe-based iPCR to assay salbutamol with a LOD of 21 fg/mL, exhibiting a 300-fold better LOD than ELISA using the same antibody (Lei et al., Citation2015).
However, most hormones are organic chemically-synthesized drugs, which can be adequately assessed using technologies, such as gas chromatography, liquid chromatography, and mass spectrometry, or merely a general IA. Consequently, cooperative detection with INA is rarely used currently.
2.3. Toxins
In recent years, the natural toxins in raw materials, as well as the accumulated toxins caused by microbial and pathogenic bacteria during the production, processing, and transportation of food, have increased the risk to human health and has become a substantial public health challenge.
Ricin is an extremely toxic natural protein with a tremendous ability to kill cells. He et al., used sandwich iPCR for the quantification of ricin in ground beef, liquid chicken egg, and milk, since the sensitive detection of this substance ensures food safety. The LOD of each sample was compared with those obtained from ELISA and found to have increased by 10-fold, to 1000-fold (He et al., Citation2010).
Staphylococcus enterotoxin (SE) is one of the most common causes of foodborne bacterial poisoning. Fischer et al. first combined the adaptability of an enzyme immunoassay (EIA) with real-time PCR for the detection of staphylococcal enterotoxins A (SEA) and B (SEB), offering versatile, highly sensitive, and specific tool for universal signal amplification via the stable antibody–DNA conjugate (Fischer et al., Citation2007). Therefore, an iPCR signal amplification (iPCR-SA) assay was created by Panneerseelan et al. The addition of magnetic beads allowed for the detection of the SEA and SEB in complex food samples, such as tryptic soy broth, milk, lemon cream pie, tuna salad, deli turkey, and ground turkey (Panneerseelan & Muriana, Citation2009). Kwon et al. established a quantitative RT-iPCR technique that performed the rapid detection of SEs in 1.5 h (Kwon et al., Citation2014). Artykov et al. used phage display mediated iPCR to increase the SEA LOD in milk to 100 pg/mL (Artykov et al., Citation2017). Besides SE, Mason et al. described an ultrasensitive iPCR based on the liposomes encapsulated in the DNA reporter, which increased the signal specificity of the substrate in the PCR amplification for detecting cholera toxin and botulinum neurotoxin type A (Mason et al., Citation2006). Rajkovic et al. used the same reaction to distinguish the presence and absence of botulinum neurotoxins A and B in semi-fat milk, employing multiplex iPCR (Rajkovic et al., Citation2012).
Among the mycotoxins, aflatoxins (AFT) are critical food pollutants that pose a significant health risk due to their carcinogenicity and mutagenicity (Gurban et al., Citation2017). Therefore, assay methods that are highly sensitive, specific, and simple to use are necessary to detect low concentrations of AFT in complex food samples. Babu and Muriana et al., successfully performed the sensitive detection and quantification of low natural AFT levels in complex food matrices and animal feed samples without requiring additional sample cleanup. Furthermore, based on quantitative RT-iPCR (Babu & Muriana, Citation2011; Babu & Muriana, Citation2014), and with the development of aptamer-based electrochemical immunosensors, the LOD of Aflatoxins M1 (AFM1) in milk and dairy products increased to ng/L (Gurban et al., Citation2017). Moreover, Wang et al. used anti-idiotypic VHH phage display-mediated iPCR to detect the mycotoxin, zearalenone, in cereals, indicating that the LOD (6.5 pg/mL) had improved 12-fold after iPCR-SA than when subjected to phage ELISA (Wang et al., Citation2016).
2.4. Pathogenic microorganisms
Pathogenic microorganisms represent the most common biological factors responsible for causing foodborne gastrointestinal diseases. However, traditional detection methods usually need to separate the culture first, then perform the biochemical or the serological test, leading to a prolonged sample processing cycle that cannot satisfy the actual application requirements (Cao et al., Citation2008; Dhama et al., Citation2013). Therefore, the proposed INA cooperative detection technology provides an alternative for pathogenic microorganism recognition.
Parasite infection often occurs from consuming food that is raw or not heated thoroughly, which is often missed or misdiagnosed by common etiology detection techniques, and a cause for significant concern in the field of food safety. Chye et al. used iPCR to detect circulating Angiostrongylus cantonensis antigens in serum samples (Chye et al., Citation2004), while Hua et al. used the same technology to detect toxoplasma circulating antigens in infected mice serum, providing a particular reference for the highly specific and sensitive diagnosis of parasites in food (Hua et al., Citation2005).
The detection of microorganisms has been a top priority in the field of food safety, and iPCR as a highly effective microdetection technique is widely used in this regard. Taban et al. detected Salmonella in chicken meat over a period of 16 h by combining two techniques, namely immunomagnetic separation and PCR (Taban & Aytac, Citation2009). Therefore, the introduction of immunocapture improves the capturing of high-volume samples with low target numbers, while simplifying the enrichment step. By using this technology combined with PCR, Waller, et al., and Morales-Rayas et al., achieved the isolation of Campylobacter cells in chicken skin, as well as the direct quantitative detection of Campylobacter jejuni in spiked milk samples and chicken skin washes (Morales-Rayas et al., Citation2008; Waller & Ogata, Citation2000). Peng et al. developed a reproducible way to detect 16S ribosomal RNA (rRNA) genes in Aeromonas hydrophila (Peng et al., Citation2002). Besides PCR, Zhang et al. a more sensitive, specific, and reliable detection than LAMP to identify Shigella in milk and clinical samples, namely immunocapture-LAMP, in which the LOD reached 8.7 CFU/mL and the overall analysis time was approximately 1 h (Zhang et al., Citation2018). Apart from the techniques mentioned above, the novel aptamer-IA was used for the detection of Salmonella typhimurium, Vibrio parahaemolyticus, and Listeria monocytogenes (Ohk et al., Citation2010; Teng et al., Citation2017; Wu et al., Citation2014). Combined with an amperometric or fiber-optic biosensor, the higher specificity of the antibody-aptamer achieved more than 10-fold sensitivity and more rapid detection than the classic ELISA.
Viruses in livestock or crops may cause zoonosis and a decline in the quality of grain production and is a food safety aspect requiring more attention. Van et al. attempted to diagnose the foot-and-mouth disease virus using immuno-RCA (iRCA) in 96-well ELISA plates. The false-positive results showed the non-specific conjugation with the DNA tail and the well surface, providing a reference for future improvement (Van Dessel et al., Citation2008). For the detection of plant viruses, researchers performed assays of the potato leafroll virus, raspberry bushy dwarf virus, and tomato spotted wilt virus using immunocapture, which was based on NASBA (Leone et al., Citation1997), RT–PCR (Kokko et al., Citation1996), and LAMP, respectively (Fukuta et al., Citation2004) to avoid the thermal and chemical disruption of the virus particles that relied on instruments and reagents. The Citrus tristeza virus (CTV) was isolated using PCR-ELISA (Silva & Nolasco, Citation2013).
2.5. Food additives
The excessive consumption of food additives increases the burden on the human metabolic organs and can even lead to the occurrence of cancer. Therefore, the number of food additives should be strictly controlled and effectively detected. Sun et al. first used the direct competitive biotin–streptavidin-amplified system based on RT-iPCR for detecting the plasticizer of dimethyl phthalate in beverage and drinking water samples. The linearity ranged from 10 ng/L to 100 ng/L, and the LOD was 1.98 pg/L while using the sensitive and high-throughput method (Sun et al., Citation2015). Furthermore, the detection of diethyl phthalate (DEP) in food samples was also achieved using a direct, high-throughput competitive RT-iPCR technique that was ultrasensitive and improved with gold nanoparticles, where the low linearity was achieved within a range of 4 pg/L to 40 ng/L, and the LOD was enhanced to 1.06 pg/L (Sun & Zhuang, Citation2015). The polychlorinated biphenyl (PCBs) in soil is often used as an additive in oily materials, Chen et al. developed a fast, powerful assay based on real-time ELISA PCR for the detection of PCBs in soil samples, where the linear sensitivity range of the assay covered more than six orders of magnitude, and the minimum LOD reached 5 fg/ml antigen (Chen & Zhuang, Citation2009). The same method was used for the assay of 3,4-dichlorinated biphenyl, where the linear range for the determination was between 10.0 fg/mL and 1.0 ng/mL, while the LOD was 1.53 fg/mL (Yu & Chen, Citation2013).
2.6. GMF
GMF is processed from crops that carried exogenous genes via genetic engineering technologies. Due to the uncertainty regarding its impact on public health, it is necessary to diagnose and label GMF. The common methods used for product quality control is performing qualitative analysis on specific transgenic protein markers in raw materials or semi-finished products, to determine whether the food is derived from genetically modified organisms or contains genetically modified ingredients. By using RT-iPCR, Kumar et al. overcame the low sensitivity of the methods only used for highly specific antibodies and achieved ppb level detection for vegetative insecticidal protein (Vip) and Cry1Ab protein (Kumar, Citation2011; Kumar & Kumar, Citation2012). Liu et al., developed a new phage-mediated iPCR for the ultrasensitive determination of Cry1Ac proteins based on the nanobodies, showing a dynamic range of 0.001 ng/mL to 100 ng/mL, while the LOD improved to 0.1 pg/mL (Liu et al., Citation2016). Cheng et al. was inspired by the convenient detectability of LAMP and introduced a cooperative IA based on LAMP and a lateral flow biosensor capable of rapid detection, which completed the assay for the in-field supervision and on-site screening of stacked GM soybean in 120 min without requiring large instruments (Cheng et al., Citation2017). Moreover, Liu et al. established an in situ liquid-phase hybridization-mediated PCR-ELISA method that enabled the fast, specific, and accurate detection of GMOs components in food products, establishing a reference for detection in large raw and processed food product samples using a full-automatic IA (Liu et al., Citation2004).
2.7. Allergens and harmful substances
Allergens and harmful substances in food or the environment cause food pollution during the process of breeding livestock, crop cultivation, raw material processing, and transportation. The ovalbumin (OVA) in animal eggs is an important source of protein, as well as the major allergens in food. Kobori et al. used iRCA to achieve the real-time detection of OVA at concentrations ranging from 10−12 g/mL to 10−7 g/mL. Combined with a fluorescent dye and a circular DNA probe, the effective signal enhancement of RCA contributed to the sensitive and effortless IA of food allergens (Kobori et al., Citation2009). As an additive, tetrabromobisphenol A (TBBPA) is an endocrine-disrupting chemical with characteristics denoting persistence, bioaccumulation, and biotoxicity in the environment. Bu et al. established an indirect competitive biotin–streptavidin IA, based on reliable and ultra-sensitive real-time PCR for the determination of TBBPA. The LOD could reach up to 2 pg/L with a detection range from 10 pg/L to 10 ng/L (Bu et al., Citation2015). Similarly, Ma et al. detected Benzo[a]pyrene (BaP) in vegetables and barbecued food, presenting significant potential hazards to the ecosystem and human health. The LOD of Bap was 2.85 pg/L with a concentration range from 5 pg/L to 10 ng/L (Ma et al., Citation2018).
3. Summary and outline
Human health and social stability are related to food safety. However, with the rapid development in food processing and manufacturing, factors such as temperature abuse, unsafe formulations, inadequate fermentation, insufficient thermal processing, post-process contamination, and a lack of adequate quality control for adjunct ingredients (Rajkovic et al., Citation2012), can cause serious food hygiene problems. Therefore, to minimise health risks from farm to table, it is important to detect and supervise contaminants and hazardous materials in food, which requires more rapid, more accurate, and more sensitive detection technologies. ()
Although IAs based on specific recognition reactions of antigens and antibodies (such as ELISA) play a significant role in the initial qualitative screening and real-time detection of food safety risk factors, they still exhibit some defects (Cui et al., Citation2018). These limitations include the fact that the detection sensitivity cannot adequately meet the international limit standard, the preparation cycle of the natural antibodies is long, the procedure is tedious and complex, the repeatability is poor, the detection for unknown complex samples, while the multi-component is blind, restricted and unsuitable. Moreover, these factors do not adequately meet the requirements of a modern food safety assay.
The nucleic acid amplification technology is continuously improving, breaking through the bottleneck of the IA. By amplifying the signal probe, iPCR achieves the quantitative ultrasensitive detection of allergens, contaminants, and drug residues in food. Combined with NASBA, the IA for detecting the short-stranded RNA virus in crops exhibits higher efficacy. The introduction of immuno-LAMP promotes the on-site, naked eye diagnosis and rapid qualitative detection of microorganisms, as well as the separation of GMF. Furthermore, it is also expected that the addition and creation of new nucleic acid amplification technologies will provide new inspiration for food safety diagnosis.
The pure synthesis in vitro of highly stable aptamers is easy to achieve, and they are significantly superior to antibodies regarding their applicability to various targets (Song et al., Citation2019). During toxin detection, the weak chemical conjugation between a toxin and antibody proteins can be offset by the introduction of specific nucleic acid aptamers. Moreover, compared with conventional methods, antibody-aptamer cooperative diagnosis in microorganisms displays higher efficacy, screening of additional useable aptamers still needs to be performed to resolve the limitations during the detection of pesticides and hormone drug residues.
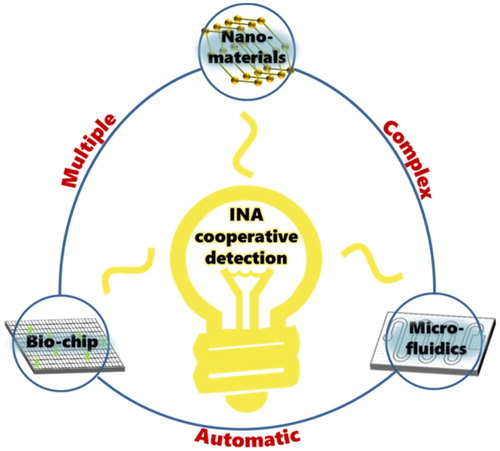
In conclusion, the cooperative IA based on nucleic acids and aptamers has displays potential for substantial application in food safety. Advances in nanotechnology (He et al., Citation2019; Zhou et al., Citation2019), biochip technology (Ha et al., Citation2016), and microfluidics (Asal et al., Citation2018; Chen et al., Citation2019; Hao et al., Citation2018) will allow immunosensors to assist during INA cooperative detection to better capture the target material and transform the reaction signal, leading to more rapid and simplified detection. () This will provide a foundation for the automatic detection of large samples and complex substances in the field of food safety.
Therefore, researchers believe that the novel INA cooperative detection technology can provide diverse options for different detection techniques while increasing their economic and social application value.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Artykov, A. A., Fursova, K. K., Ryazantsev, D. Y., Shchannikova, M. P., Loskutova, I. V., Shepelyakovskaya, A. O., Laman, A. G., Zavriev, S. K., & Brovko, F. A. (2017). Detection of staphylococcal enterotoxin a by phage display mediated immuno-PCR method. Russian Journal of Bioorganic Chemistry, 43(5), 540–543. https://doi.org/10.1134/S1068162017050028
- Asal, M., Ozen, O., Sahinler, M., & Polatoglu, I. (2018). Recent Developments in enzyme, DNA and immuno-based biosensors. SENSORS, 18(6), 1924. https://doi.org/10.3390/s18061924
- Babu, D., & Muriana, P. M. (2011). Immunomagnetic bead-based recovery and real time quantitative PCR (RT iq-PCR) for sensitive quantification of aflatoxin B(1). Journal of Microbiological Methods, 86(2), 188–194. https://doi.org/10.1016/j.mimet.2011.05.002
- Babu, D., & Muriana, P. M. (2014). Sensitive quantification of aflatoxin B1 in animal feeds, corn feed grain, and yellow corn meal using immunomagnetic bead-based recovery and real-time immunoquantitative-PCR. Toxins, 6(12), 3223–3237. https://doi.org/10.3390/toxins6123223
- Bu, D., Zhuang, H., Yang, G., & Ping, X. (2015). A real-time immuno-PCR assay for the detection of tetrabromobisphenol A. Analytical Methods, 7(1), 99–106. https://doi.org/10.1039/C4AY01343C
- Cao, W., Wang, M., Wang, X., & Liu, X. (2008). Studies on rapid detection of food-borne pathogenic bacteria by nucleic acid testing and related technology. Journal of Hygiene Research, 37(2), 245–248. https://doi.org/10.1016/S1005-8885
- Chen, A., Ding, X., & Xiong, Y. (2010). Application of monoclonal antibody in food safety and clinic. Labeled Immunoassays and Clinical Medicine, 17(5), 338–341. https://doi.org/10.3969/j.issn.1006-1703.2010.05.027
- Chen, Z., Zhou, K., Ha, W., Chen, P., Fu, H., Shen, Y., Sun, Y., & Xu, Z. (2019). Development of a low-cost, simple, fast and quantitative lateral-flow immunochromatographic assay (ICA) strip for melatonin in health foods. Food and Agricultural Immunology, 30(1), 497–509. https://doi.org/10.1080/09540105.2019.1602112
- Chen, H. Y., & Zhuang, H. S. (2009). Real-time immuno-PCR assay for detecting PCBs in soil samples. Analytical and Bioanalytical Chemistry, 394(4), 1205–1211. https://doi.org/10.1007/s00216-009-2787-8
- Cheng, N., Shang, Y., Xu, Y., Zhang, L., Luo, Y., Huang, K., & Xu, W. (2017). On-site detection of stacked genetically modified soybean based on event-specific TM-LAMP and a DNAzyme-lateral flow biosensor. Biosensors and Bioelectronics, 91, 408–416. https://doi.org/10.1016/j.bios.2016.12.066
- Choi, S., Rhie, G., & Jeon, J. H. (2020). Development of a double-antibody sandwich ELISA for sensitive detection of Yersinia pestis. Microbiology and Immunology, 64(1), 72–75. https://doi.org/10.1111/1348-0421.12751
- Chye, S. M., Lin, S. R., Chen, Y. L., Chung, L.-Y., & Yen, C.-M. (2004). Immuno-PCR for detection of antigen to angiostrongylus cantonensis circulating fifth-stage worms. Clinical Chemistry, 50(1), 51–57. https://doi.org/10.1373/clinchem.2003.020867
- Cui, X., Jin, M., Du, P., Chen, G., Zhang, C., Zhang, Y., Shao, Y., & Wang, J. (2018). Development of immunoassays for multi-residue detection of small molecule compounds. Food and Agricultural Immunology, 29(1), 638–652. https://doi.org/10.1080/09540105.2018.1428284
- Dai, M. (2015). Study on the methods of real-time immuno-PCR for detection ractopamine. China Jiliang University, 1–77.
- Dhama, K., Rajagunalan, S., Chakraborty, S., Verma, A. K., Kumar, A., Tiwari, R., & Kapoor, S. (2013). Food-borne pathogens of animal origin-diagnosis, prevention, control and their zoonotic significance: A review. Pakistan Journal of Biological Sciences: PJBS, 16(20), 1076–1085. https://doi.org/10.3923/pjbs.2013.1076.1085
- Duan, Y., Gao, Z., Wang, L., Wang, H., Zhang, H., & Li, H. (2016). Selection and identification of chloramphenicol-specific DNA aptamers by Mag-SELEX. Applied Biochemistry and Biotechnology, 180(8), 1644–1656. https://doi.org/10.1007/s12010-016-2193-6
- Fischer, A., von Eiff, C., Kuczius, T., Omoe, K., Peters, G., & Becker, K. (2007). A quantitative real-time immuno-PCR approach for detection of staphylococcal enterotoxins. Journal of Molecular Medicine, 85(5), 461–469. https://doi.org/10.1007/s00109-006-0142-5
- Fukuta, S., Ohishi, K., Yoshida, K., Mizukami, Y., Ishida, A., & Kanbe, M. (2004). Development of immunocapture reverse transcription loop-mediated isothermal amplification for the detection of tomato spotted wilt virus from chrysanthemum. Journal of Virological Methods, 121(1), 49–55. https://doi.org/10.1016/j.jviromet.2004.05.016
- Gurban, A., Epure, P., Oancea, F., & Doni, M. (2017). Achievements and prospects in electrochemical-based biosensing platforms for aflatoxin M₁ detection in milk and dairy products. Sensors (Basel, Switzerland), 17(12), 2951. https://doi.org/10.3390/s17122951
- Ha, M., Chung, M., & Bae, D. (2016). Surface modification techniques and competitive immunoassay to detect residual ciprofloxacin in foods. Food and Agricultural Immunology, 27(6), 886–896. https://doi.org/10.1080/09540105.2016.1202212
- Hao, K., Suryoprabowo, S., Song, S., Liu, L., & Kuang, H. (2018). Rapid detection of zearalenone and its metabolite in corn flour with the immunochromatographic test strip. Food and Agricultural Immunology, 29(1), 498–510. https://doi.org/10.1080/09540105.2017.1406461
- He, X., McMahon, S., McKeon, T., & Brandon, D. (2010). Development of a novel immuno-PCR assay for detection of ricin in ground beef, liquid chicken egg, and milk. Journal of Food Protection, 73(4), 695–700. https://doi.org/10.4315/0362-028X-73.4.695
- He, F., Zou, T., Yang, J., Wang, H., Deng, L., Tian, Y., Xu, Z., Sun, Y., Lei, H., Tan, X., & Shen, Y. (2019). Development of a skeleton-specific antibody and Au nanoparticle-based immunochromatographic sensor for simultaneous detection of various tadalafil adulterants in health food. Food and Agricultural Immunology, 30(1), 349–368. https://doi.org/10.1080/09540105.2019.1585417
- Hua, X., Yin, W., Shi, H., Li, M., Wang, Y., Wang, H., Ye, Y., Kim, H. J., Gee, S. J., Wang, M., Liu, F., & Hammock, B. D. (2014). Development of phage immuno-loop-mediated isothermal amplification assays for organophosphorus pesticides in agro-products. Analytical Chemistry, 86(16), 8441–8447. https://doi.org/10.1021/ac5020657
- Hua, H., Zhou, Y., & Xue, Z. (2005). Study on the detection of toxoplasma circulating antigen with double antibodies immune-PCR. Modern Medicine Journal of China, 7(6), 1–2. https://doi.org/10.3969/j.issn.1672-9463.2005.06.001
- Kobori, T., Matsumoto, A., Takahashi, H., & Sugiyama, S. (2009). Rolling circle amplification for signal enhancement in ovalbumin detection. Analytical Sciences, 25(12), 1381–1383. https://doi.org/10.2116/analsci.25.1381
- Kokko, H. I., Kivineva, M., & Kärenlampi, S. O. (1996). Single-step immunocapture RT-PCR in the detection of raspberry bushy dwarf virus. Biotechniques, 20(5), 842–846. https://doi.org/10.2144/96205st03
- Kumar, R. (2011). A quantitative Immunopolymerase chain reaction method for detection of vegetative insecticidal protein in genetically modified crops. Journal of Agricultural and Food Chemistry, 59(19), 10448–10453. https://doi.org/10.1021/jf202453v
- Kumar, R., & Kumar, R. (2012). A real-time immuno-PCR assay for the detection of transgenic Cry1Ab protein. European Food Research and Technology, 234(1), 101–108. https://doi.org/10.1007/s00217-011-1618-2
- Kwon, K. H., Hwang, S. Y., Park, Y. K., Yoon, J. W., Kim, S., & Hong, J. (2014). A quantitative real-time immuno-PCR assay for detection of staphylococcus aureus enterotoxin H. Journal of Food Safety, 34(3), 249–256. https://doi.org/10.1111/jfs.12120
- Lei, Y., Li, X., Akash, M. S. H., Zhou, L., Tang, X., Shi, W., Liu, Z., & Chen, S. (2015). Development of analytical method for ultrasensitive detection of salbutamol utilizing DNA labeled-immunoprobe. Journal of Pharmaceutical and Biomedical Analysis, 107, 204–208. https://doi.org/10.1016/j.jpba.2014.12.027
- Leone, G., Van Schijndel H, B., van Genien, B., & Schoen, C. D. (1997). Direct detection of potato leafroll virus in potato tubers by immunocapture and the isothermal nucleic acid amplification method NASBA. Journal of Virological Methods, 66(1), 19–27. https://doi.org/10.1016/S0166-0934(97)02203-9
- Liao, Y., Cui, X., Chen, G., Wang, Y., Qin, G., Li, M., Zhang, X., Zhang, Y., Zhang, C., Du, P., Yan, F., El-Aty, A. M. A., Wang, J., Zhang, C., & Jin, M. (2019). Simple and sensitive detection of triazophos pesticide by using quantum dots nanobeads based on immunoassay. Food and Agricultural Immunology, 30(1), 522–532. https://doi.org/10.1080/09540105.2019.1597022
- Liu, Y., Jiang, D., Lu, X., Wang, W., Xu, Y., & He, Q. (2016). Phage-mediated immuno-PCR for ultrasensitive detection of Cry1Ac protein based on nanobody. Journal of Agricultural and Food Chemistry, 64(41), 7882–7889. https://doi.org/10.1021/acs.jafc.6b02978
- Liu, G., Su, W., Xu, Q., Long, M., Zhou, J., & Song, S. (2004). Liquid-phase hybridization based PCR-ELISA for detection of genetically modified organisms in food. Food Control, 15(4), 303–306. https://doi.org/10.1016/S0956-7135(03)00081-1
- Ma, L. (2013). Advances in nucleic acid isothermal amplification technology. Zhejiang Journal of Preventive Medicine, 25(1), 24–27. https://doi.org/10.3969/j.issn.1007-0931.2013.01.007
- Ma, Z., Ma, Z., Zhuang, H., & Zhuang, H. (2018). A highly sensitive real-time immuno-PCR assay for detecting benzo[a]pyrene in food samples by application of biotin-streptavidin system. Food Analytical Methods, 11(3), 862–872. https://doi.org/10.1007/s12161-017-1046-4
- Mason, J., Xu, L., Sheng, Z., He, J., & O'Leary, T. (2006). Liposome polymerase chain reaction assay for the sub-attomolar detection of cholera toxin and botulinum neurotoxin type A. Nature Protocols, 1(4), 2003–2011. https://doi.org/10.1038/nprot.2006.331
- Morales-Rayas, R., Wolffs, P. F. G., & Griffiths, M. W. (2008). Immunocapture and real-time PCR to detect Campylobacter spp. Journal of Food Protection, 71(12), 2543–2547. https://doi.org/10.4315/0362-028X-71.12.2543
- Ohk, S. H., Koo, O. K., Sen, T., Yamamoto, C. M., & Bhunia, A. K. (2010). Antibody-aptamer functionalized fibre-optic biosensor for specific detection of Listeria monocytogenes from food. Journal of Applied Microbiology, 109(3), 808–817. https://doi.org/10.1111/j.1365-2672.2010.04709.x
- Panneerseelan, L., & Muriana, P. (2009). An immunomagnetic PCR signal amplification assay for sensitive detection of staphylococcus aureus enterotoxins in foods. Journal of Food Protection, 72(12), 2538–2546. https://doi.org/10.4315/0362-028X-72.12.2538
- Peng, X., Zhang, J., Wang, S., Lin, Z., & Zhang, W. (2002). Immuno-capture PCR for detection of aeromonas hydrophila. Journal of Microbiological Methods, 49(3), 335–338. https://doi.org/10.1016/S0167-7012(02)00010-6
- Rajkovic, A., El Moualij, B., Fikri, Y., Dierick, K., Zorzi, W., Heinen, E., Uner, A., & Uyttendaele, M. (2012). Detection of Clostridium botulinum neurotoxins A and B in milk by ELISA and immuno-PCR at higher sensitivity than mouse bio-assay. Food Analytical Methods, 5(3), 319–326. https://doi.org/10.1007/s12161-011-9300-7
- Shen, W., Huang, M., Xiao, X., Xu, X., Zhou, X., & Zhou, G. (2010). Immuno-PCR technique and its application in food safety. Journal of Nanjing Agricultural University, 33(6), 119–124. https://doi.org/10.7685/j.issn.1000-2030.2010.06.023
- Silva, G., & Nolasco, G. (2013). Epidemiological situation of Citrus tristeza virus in mainland Portugal. Phytopathologia Mediterranea, 52(3), 472–477. https://doi.org/10.1071/FP12392
- Song, S., Gao, Z., Guo, X., & Chen, G. (2019). Aptamer-based detection methodology studies in food safety. Food Analytical Methods, 12(4), 966–990. https://doi.org/10.1007/s12161-019-01437-3
- Sun, R., Sun, R., Zhuang, H., & Zhuang, H. (2015). Biotin-streptavidin-amplified real-time immune-PCR assay for detecting dimethyl phthalate in beverage and drinking water samples. Analytical and Bioanalytical Chemistry, 407(4), 1261–1265. https://doi.org/10.1007/s00216-014-8329-z
- Sun, R., & Zhuang, H. (2015). An ultrasensitive gold nanoparticles improved real-time immuno-PCR assay for detecting diethyl phthalate in foodstuff samples. Analytical Biochemistry, 480, 49–57. https://doi.org/10.1016/j.ab.2015.04.010
- Taban, B., & Aytac, S. (2009). Application of magnetic immuno-polymerase chain reaction assay for detection of Salmonella spp. in chicken meats. European Food Research and Technology, 229(4), 623–628. https://doi.org/10.1007/s00217-009-1091-3
- Tao, X., He, Z., Cao, X., Shen, J., & Li, H. (2014). Development of a highly sensitive real-time immuno-PCR for the measurement of chloramphenicol in milk based on magnetic bead capturing. Analytical Methods, 6(23), 9340–9347. https://doi.org/10.1039/C4AY02158D
- Teng, J., Ye, Y., Yao, L., Yan, C., Cheng, K., Xue, F., Pan, D., Li, B., & Chen, W. (2017). Rolling circle amplification based amperometric aptamer/immuno hybrid biosensor for ultrasensitive detection of Vibrio parahaemolyticus. Microchimica Acta, 184(9), 3477–3485. https://doi.org/10.1007/s00604-017-2383-0
- Tian, J., Chu, H., Zhang, Y., Li, K., Tian, H., Zhang, X., & Xu, W. (2019). Tio2 nanoparticle-enhanced linker recombinant strand displacement amplification (LRSDA) for universal label-free visual bioassays. ACS Applied Materials & Interfaces, 11(50), 46504–46514. https://doi.org/10.1021/acsami.9b16314
- Van Dessel, W., Vandenbussche, F., Staes, M., Goris, N., & De Clercq, K. (2008). Assessment of the diagnostic potential of immuno-RCA in 96-well ELISA plates for foot-and-mouth disease virus. Journal of Virological Methods, 147(1), 151–156. https://doi.org/10.1016/j.jviromet.2007.08.020
- Waller, D. F., & Ogata, S. A. (2000). Quantitative immunocapture PCR assay for detection of campylobacter jejuni in foods. Applied and Environmental Microbiology, 66(9), 4115–4118. https://doi.org/10.1128/AEM.66.9.4115-4118.2000
- Wang, X., He, Q., Xu, Y., Liu, X., Shu, M., Tu, Z., Li, Y., Wang, W., & Cao, D. (2016). Anti-idiotypic VHH phage display-mediated immuno-PCR for ultrasensitive determination of mycotoxin zearalenone in cereals. Talanta, 147, 410–415. https://doi.org/10.1016/j.talanta.2015.09.072
- Wei, X. (2018). The application of ELISA in analysis of food safety. Journal of Liuzhou Vocational & Technical College, 18(1), 121–125. https://doi.org/10.16221/j.cnki.issn1671-1084.2018.01.026
- Wu, W., Li, J., Pan, D., Li, J., Song, S., Rong, M., Li, Z., Gao, J., & Lu, J. (2014). Gold nanoparticle-based enzyme-linked antibody-aptamer sandwich assay for detection of Salmonella typhimurium. ACS Applied Materials & Interfaces, 6(19), 16974–16981. https://doi.org/10.1021/am5045828
- Xiang, L., Wu, H., Cui, Z., & Tang, J. (2019). Indirect competitive aptamer-based enzyme-linked immunosorbent assay (apt-ELISA) for the specific and sensitive detection of isocarbophos residues. Analytical Letters, 1–10. https://doi.org/10.1080/00032719.2019.1587446
- Xiao, X., Zhu, L., He, W., Luo, Y., & Xu, W. (2019). Functional nucleic acids tailoring and its application. Trends in Analytical Chemistry, 118, 138–157. https://doi.org/10.1016/j.trac.2019.05.027
- Xu, W., Tian, J., Shao, X., Zhu, L., Huang, K., & Luo, Y. (2017). A rapid and visual aptasensor for Lipopolysaccharides detection based on the bulb-like triplex turn-on switch coupled with HCR-HRP nanostructures. Biosensors and Bioelectronics, 89(Pt 2), 795–801. https://doi.org/10.1016/j.bios.2016.10.012
- Yang, S., Yi, X., Mao, X., Liu, Y., Zhang, S., & Li, Y. (2018). Integrated immunoassay-based broad detection of multi-class mycotoxins. Food and Agricultural Immunology, 29(1), 615–624. https://doi.org/10.1080/09540105.2018.1424121
- Yu, H., & Chen, S. H. (2013). Analysis of polychlorinated Biphenyls congeners in soil samples by real-time immuno-PCR assay. The 17th National Symposium on Organic Analysis and Biological Analysis of Chinese Chemical Society.
- Zhang, L., Wei, Q., Han, Q., Chen, Q., Tai, W., Zhang, J., Song, Y., & Xia, X. (2018). Detection of Shigella in milk and clinical samples by magnetic immunecaptured-loop-mediated isothermal amplification assay. Frontiers in Microbiology, 9, 94. https://doi.org/10.3389/fmicb.2018.00094
- Zhou, B., Yuan, X., Hu, Y., Fan, J., Yang, W., Guo, M., Zhang, Y., Li, W., & Zhang, J. (2019). Detection of deoxynivalenol (DON) using europium chelates and magnetic nanoparticles. Food and Agricultural Immunology, 30(1), 87–94. https://doi.org/10.1080/09540105.2018.1548577