ABSTRACT
Herbal formulas supplemented have improved the health of ruminants. Our objectives were to determine the effects of supplemental Indian polyherbal phytogenic sources (IndPPs) on the growth performance, blood chemistry, blood cells, and antibody amounts of dairy calves. Forty Holstein calves (Initial BW 45.8 ± 7.2 kg) were randomly assigned to treatments: 0, 1, 2, and 3 gd-1 of IndPPs. The herbal supplement was characterized by GC-MS analysis. The calves treated with IndPPs additive did not affect the average productive variable involved in growth. The use of IndPPs linearly reduces the blood serum biochemistry glucose (P < 0.01) but linearly increases the blood serum concentration of protein, bilirubin, alkaline phosphatase, and antibody concentrations (P < 0.05). On blood cells from calves, supplementation with IndPPs linearly increases the concentrations of hematocrit, hemoglobin, and erythrocytes (P < 0.05). These results were attributed to the IndPPs containing bioactive substances with phytogenic properties.
KEYWORDS:
Introduction
Rearing calves are among the most critical and sensitive management plans in cattle farms as calves are considered significant sources of profits for dairy farms (Seifzadeh et al., Citation2017). During calf rearing, respiratory and digestive problems occur, and the incidence may cause high morbidity, mortality, and economic loss (Cho & Yoon, Citation2014). These diseases require antibiotics, which have been banned as growth promotors in several countries (McGuirk, Citation2008; Björkman et al., Citation2021). Also, there is an increase in public pressure for the usage of these antibiotics in animals (Björkman et al., Citation2021).
Thus, alternative sources of feedstuff should be evaluated to maximize the livestock production systems (Sánchez et al., Citation2021). In recent years, research in the use of herbal feed additives has reported beneficial effects during the suckling period in sheep (Crosby et al., Citation2017; Roque-Jiménez et al., Citation2020). The herbal feed additives have been reported with different bioactive compounds such as alkaloids, polyphenols, isothiocyanates, tannins, saponins, and terpenoids, all of which can stimulate the immune system (Frankič et al., Citation2009). There is interest in using herbal feed additives as alternatives to antibiotics in dairy and beef production systems during diseases related to respiratory and digestive systems. (Seifzadeh et al., Citation2017; Cho & Yoon, Citation2014). However, the properties of herbal feed additives have mainly aimed to modify rumen fermentation (Frankič et al., Citation2009). Currently, some standardized herbal formulas as antibiotics sources can contain Acacia concinna, Saccharum officinarum, Adhatoda vasica, Solanum xanthocarpum, Curcuma longa, Hedychium spicatum, Boerrhavia diffusa, Piper longum, Albezia lebbek, and other varieties (Sánchez et al., Citation2021). These plants have been related to higher contains phytogenic, with positive effects on ruminant production, health, and immune response (Sánchez et al., Citation2021; Mendoza et al., Citation2019). Herbal formulas differ from extracts or essential oils (Frankič et al., Citation2009). It usually contains several molecules with one or more predominant active substances or nutrients responsible for positive biological effects compromised of diverse metabolites and mechanisms of action (Seifzadeh et al., Citation2017; Frankič et al., Citation2009). Assan et al. (Assan, Citation2018) proposed that herbal formulas could be a source of antibiotics in mammals’ suckling period. However, the research is still missing about indicators of health and production in the offspring supplemented with different types of bioactive compounds in the herbal formulas. (Lv et al., Citation2021; Quigley, Citation2011).
Based on the previous background, we hypothesized that the herbal compounds of Indian polyherbal phytogenic sources (IndPPs) have beneficial effects on dairy calves’ immune system, growth performance, blood biochemistry, and biometry. Therefore, the objective of this experiment was to characterize the natural compounds in IndPPs and determine their effect on the immune system, growth performance, blood biochemistry, and biometry of dairy calves.
Materials and methods
Ethics
The animal procedures were reviewed and approved by the Committee for the Ethical Use of Animals in Experiments of Universidad Autónoma Metropolitana, and Universidad Autonoma de San Luis Potosi, according to the regulations and standards that are required by the Mexican Government for the use of animals for a number of diverse activities. Federal law on technical specifications for the care and use of laboratory animals and for livestock farms; farms; centres of production, reproduction, and breeding; zoos; and exhibition halls must meet the basic principles of animal welfare (NOM-062-ZOO-1995).
Location
The current experiment was conducted at the facilities of a Mexican dairy farm located on Torreon, Coahuila (Latitude 25°39'14.4"N; Longitude 103°27'27.8"W; altitude 1,139 metres) in semi-dry climate (mean temperature of 20.21 °C).
Indian polyherbal phytogenic source characterization
The herbal formula used in the current experimental study (IndPPs; PeptaSan®, Nuproxa, LTD, Switzerland) is a commercial standardized herbal formula labelled that contain Acacia concinna and Saccharum officinarum. The extraction of bioactive compounds was performed using an ultrasonic processor (GEX130, 115 V 50/60 Hz) equipped with a 3 mm titanium tip and mechanical stirrers (Cole-Parmer, IL, USA). One gram of IndPPs was mixed with 10 mL of hexane. Subsequently, the organic phase was separated, concentrated to 1 mL of extracted mixture, and evaporated (Zymark, Turbovap LV Concentration Evapotarot, NB, USA) for the final analysis.
The characterization of IndPPs was performed with gas chromatography (GC-HP 6890) coupled with mass spectrophotometry (MSHP 5973), equipped with a capillary column 60 m length, 0.255 mm diameter, and 0.25 µm film thickness (HP 5MS, Agilent). The temperature programme was 70 °C for 2 min, which was then increased to 250 °C at the rate of 20 °C/min, then to 290 °C at the rate of 5 °C/min, then increased to 300 °C at the rate of 1 °C/min, then to 310 °C at the rate of 5 °C/min and kept there for 36 min. The injector temperature was 250 °C in splitless mode. The helium flow rate was 1 mL/min. The mass spectrophotometry was programmed in SCAN mode (50–500 m/z) to identify compounds.
Animals, treatments, and sampling
Forty female Holstein calves (initial body weight (IBW) 45.8 ± 7.2 Kg) with 20.4 ± 2.9 days post-birth were randomly assigned to one of four treatments. The treatments (10 calves for treatment) consisted of orally dosed that contained: 0, 1, 2, or 3 gd−1 of IndPPs. Treatments were top-dressed on the feed using gelatin as the medium. Gelatin was made similarly to the methodology described by Sánchez et al. (Citation2021). The calves were housed in individual pens equipped with steel buckets for feed and milk. All calves received colostrum after birth and the initial total serum protein was confirmed with a refractometer (Quigley, Citation2011).
Calves were fed twice a day (07:00–17.00) with milk (56%) and milk replacer (44% Milk replacer Nu-3 Group, México), reconstituted in hot water (65°C; 130 g/L). The milk mix was served at a temperature of 39°C in the buckets. Milk replacer was offered twice per day (4 L/feeding) and reduced to one meal on day 25 as starter diet intake was augmented. The starter diet concentrate was offered since the third day of age and was gradually increased (Started Premium, Nuplen, México).
Experimental procedures
Calves were weighed on days 1, and 90 of the experiment to calculate the average daily gain. Also, on day 1 and day 10, all the calves received a bovine rhinotracheitis vaccine (Pyramyd-5; Boehringer Ingelheim). Later, on day 50, all the calves were vaccinated with a clostridial bacterin-toxoid (Covexin 10; MSD). On day 90, hip, wither height, and thoracic girth were measured to evaluate growth and development (Parish et al., Citation2012; Maciel et al., Citation2016). Feed intake (milk and starter diet concentrate) was recorded daily and based on the dry matter content of the starter diet and milk replacer. The feed efficiency ratio was estimated (Kazemi-Bonchenari et al., Citation2018); and the variation coefficient of dry matter intake, between days calculated as a stability indicator (Britton et al., Citation1990). Calves were evaluated daily for diseases and veterinary treatments (Pardon et al., Citation2013). On day 90, all the calves were sampled for blood obtain. Three blood samples (5 mL per sample) were collected from the jugular vein by a puncture and immediately transferred to blood collection tubes. The first sample without anticoagulant were centrifuged to obtain blood serum and later were used for the analysis of cholesterol, glucose, total protein, urea, uric acid, albumin, globulin, bilirubin, alkaline phosphatase (ALP), lactate dehydrogenase (LDH), aspartate aminotransferase (AST), Ca, P and creatinine using an autoanalyzer (Kontrolab QS EasyVet, Italy) as described Sánchez et al. (Citation2021). For the second sample without anticoagulant, the blood was centrifuged at 1500 rpm for five minutes to obtain blood serum, later the serum was collected in vials and put at −80°C until the ELISA analysis. Finally, the last sample of blood collected in tubes with anticoagulant was used for complete blood count, hematocrit, and leukocyte differential count in a hematology analyzer (Kontrolab QS EasyVet, Italy).
Quantification of immunoglobulins (IgG) by elisa
An ELISA method was performed using plates with antigen (Clostridium spp) obtained from the Covexin 10. The antigen was incubated for 24 hours at room temperature, then 200 µL was added to each well to decrease non-specific binding sites. Later, 100 µL of the serum sample was added to each well and incubated for 1 hour at 37°C. The plates’ contents were decanted, and 100 µL of the bovine IgG specific secondary antibody were added and allowed to incubate for 1 hour at 37°C. Subsequently, 100 of TMB (3,3’, 5,5'-liquid tetramethylbenzidine) was added to each well. Finally, 100 µL of stopping solution (H2SO4) was added to each well with the aim not to continue the reaction. The plate was read at 540 nm.
Statistical analysis
Shapiro–Wilk tests were used to ascertain normal distribution, and data were analyzed under a completely randomized design with R software, testing for linear and quadratic effects of IndPPs supplementation dose by orthogonal polynomials (Mirman, Citation2014). The initial body was tested as a covariate. For main effects, a significant difference was established at p ≤ 0.05, trends were determined at p ≥ 0.05 and p ≤ 0.10.
Results
Indian polyherbal phytogenic source (IndPPs) characterization
Gas chromatography coupled with mass spectrophotometry revealed various chemical compounds present in the IndPPs formula (). The peak with the number 4 is the solvent used in the standard mixture. The main components detected were a variety of alkanes such as toluene, tetracosane, hexacosane, and eicosane. Alkaloids, such as a type of allylbenzene called eugenol, terpenes such as thymol and vitamin E, and other components, were also detected.
Figure 1. Total ion chromatogram of the volatile components and chemical composition in IndPPs. Chemical composition of Indian polyherbal phytogenic source (IndPPs) by CG-MS with retention time(rt): 1.- Toluene; 2.- Cyclotrisiloxane, hexamethyl-; 3.- 4,7-Methanoindan-2-one, 3a,4,7,7a- tetrahydro-; 4.- 1-Hexene,3,3,5-trimethyl; 5.- Cyclopentasiloxane, decamethyl-; 6.- 2-Decenal, (E)-; 7.- Thymol; 8.- Phenol, 2-methoxy-3-(2-propenyl)-; 9.- Benzene, 1-(1,5-dimethyl-4-hexenyl)−4-methyl-; 10.- Benzene, 1-(5,5-dimethyl-1-cyclopenten-1-yl)−2-methoxy-; 11.- Caryophyllene oxide; 12.- 2-Cyclohexen-1-one, 5-methyl-2-(1-methylethyl)-; 13.- 1,5-Heptadien-4-one, 3,3,6-trimethyl-; 14.- Cyclopropyl phenyl carbinol; 15.- Naphthalene, 1,2,3,4-tetrahydro-1,1,6-trimethyl-; 16.- 5-Hydroxymethyl-1,1,4a-trimethyl-6; 17.- Hexadecanoic acid, methyl ester; 18.- n-Hexadecanoic acid; 19.- 8-Octadecenoic acid, methyl ester; 20.- Phenol, 4-(3,7-dimethyl-3-ethenylo cta-1,6-dienyl)-; 21.- Oleic Acid; 22.- 9-Octadecenoic acid, (E)-; 23.- Hexadecanoic acid, 1,1-dimethylethyl ester; 24.- 11-Eicosenoic acid, methyl ester; 25.- Octadec-9-enoic acid; 26.- Cyclohexane, 1-(1,5-dimethylhexyl)−4-(4-methylpentyl)-; 27.- Tetracosane; 28.- 9,17-Octadecadienal, (Z)-; 29.- Hexacosane; 31.- Cinnamyl cinnamate; 32.- Heptacosane; 33.- Docosane; 34.- Triacontane; 35.- Nonadecane; 36.- Dotriacontane; 37.- D,.alpha.-Tocopherol; 38.- Silane, trimethyl[5-methyl-2-(1-methylethyl)phenoxy]-; 39.- Heneicosane, 11-decyl-; 40.- Silane, trimethyl[5-methyl-2-(1-methylethyl)phenoxy]-; 41.- Acetamide, N-[4-(trimethylsilyl)phenyl]-; 42.- Eicosane; 43.- Demecolcine.
![Figure 1. Total ion chromatogram of the volatile components and chemical composition in IndPPs. Chemical composition of Indian polyherbal phytogenic source (IndPPs) by CG-MS with retention time(rt): 1.- Toluene; 2.- Cyclotrisiloxane, hexamethyl-; 3.- 4,7-Methanoindan-2-one, 3a,4,7,7a- tetrahydro-; 4.- 1-Hexene,3,3,5-trimethyl; 5.- Cyclopentasiloxane, decamethyl-; 6.- 2-Decenal, (E)-; 7.- Thymol; 8.- Phenol, 2-methoxy-3-(2-propenyl)-; 9.- Benzene, 1-(1,5-dimethyl-4-hexenyl)−4-methyl-; 10.- Benzene, 1-(5,5-dimethyl-1-cyclopenten-1-yl)−2-methoxy-; 11.- Caryophyllene oxide; 12.- 2-Cyclohexen-1-one, 5-methyl-2-(1-methylethyl)-; 13.- 1,5-Heptadien-4-one, 3,3,6-trimethyl-; 14.- Cyclopropyl phenyl carbinol; 15.- Naphthalene, 1,2,3,4-tetrahydro-1,1,6-trimethyl-; 16.- 5-Hydroxymethyl-1,1,4a-trimethyl-6; 17.- Hexadecanoic acid, methyl ester; 18.- n-Hexadecanoic acid; 19.- 8-Octadecenoic acid, methyl ester; 20.- Phenol, 4-(3,7-dimethyl-3-ethenylo cta-1,6-dienyl)-; 21.- Oleic Acid; 22.- 9-Octadecenoic acid, (E)-; 23.- Hexadecanoic acid, 1,1-dimethylethyl ester; 24.- 11-Eicosenoic acid, methyl ester; 25.- Octadec-9-enoic acid; 26.- Cyclohexane, 1-(1,5-dimethylhexyl)−4-(4-methylpentyl)-; 27.- Tetracosane; 28.- 9,17-Octadecadienal, (Z)-; 29.- Hexacosane; 31.- Cinnamyl cinnamate; 32.- Heptacosane; 33.- Docosane; 34.- Triacontane; 35.- Nonadecane; 36.- Dotriacontane; 37.- D,.alpha.-Tocopherol; 38.- Silane, trimethyl[5-methyl-2-(1-methylethyl)phenoxy]-; 39.- Heneicosane, 11-decyl-; 40.- Silane, trimethyl[5-methyl-2-(1-methylethyl)phenoxy]-; 41.- Acetamide, N-[4-(trimethylsilyl)phenyl]-; 42.- Eicosane; 43.- Demecolcine.](/cms/asset/58d3128e-6658-4b82-bc93-29d832285da2/cfai_a_2024150_f0001_ob.jpg)
Effects of an Indian polyherbal phytogenic source (IndPPs) on calves productive performance and health.
There was not difference among the treatments for the average daily gain, the final body weight, the final wither height, and the final hip height. The final thoracic girth showed a quadratic tend to be greater in the calves with 3 gd−1 of IndPPs supplementation (P = 0.09) (). The calves with supplementation using IndPPs reduce linearly (P < 0.01) the starter intake gd−1 (). Also, the calves without supplementation using IndPPs reduce the feed intake variation compared with the calves that received the supplementation with IndPPs (linear P < 0.01; quadratic P < 0.01). However, the calves with 2 gd−1 of IndPPs supplementation shown the greatest milk + replacer intake compared with the other treatments (linear P < 0.01; quadratic P < 0.01) (). Interestingly, there was no effect on calves’ health. The calves that received supplementation with IndPPs had not reduced the cases of diarrheas observed during the experimental period (linear P < 0.01). Nevertheless, the supplementation with 3 gd−1 IndPPs trend to reduce the antibiotic dose used on the calves (linear p = 0.09).
Table 1. Effect of an Indian polyherbal phytogenic source (IndPPs) on calves productive performance and health.
Effects of an Indian polyherbal phytogenic source (IndPPs) on blood serum biochemistry of calves.
Serum glucose concentration showed a greater concentration on calves without IndPPs supplementation (linear P < 0.01; quadratic P < 0.01). The supplementation with 3 gd−1 of IndPPs presented a quadratic effect on calves, increasing the concentration of B-OH butyrate on blood serum. (). Serum creatinine concentration trend to be greater on calves supplemented with IndPPs (linear P = 0.10). A significant effect and a quadratic trend were observed for a greater serum protein concentration on calves that received 2 gd−1 of IndPPs compared with the other treatments (linear P = 0.04; quadratic P = 0.09) (). Similar effects were observed using 2 gd−1 of IndPPs for serum bilirubin concentration (linear p = 0.03) and alkaline phosphatase (ALP; linear P < 0.01), where the calves increase the concentrations more than the other treatments. Additionally, the supplementation with 1 gd−1 showed a quadratic response (P = 0.01), increasing the albumin serum concentration compared to the calves’ of the other treatments. Finally, the serum phosphorus concentration trend to be greater on calves with 2 gd−1 of IndPPs supplementation (Quadratic P = 0.10) ().
Table 2. Effect of an Indian polyherbal phytogenic source (IndPPs) on blood serum biochemistry of calves.
Effects of an Indian polyherbal phytogenic source (IndPPs) on blood cells of calves.
The hematocrit percent in blood cells from calves supplemented with 2 gd−1 of IndPPs was greater linearly (P < 0.01) compared with the other treatments (). Similarly, there was a linear effect for hemoglobin concentration (P < 0.01) and erythrocytes (P = 0.01) in blood cells from calves supplemented with 2 gd−1 of IndPPs that were greater com-pared with the other treatments (). A quadratic effect (P = 0.04) was observed in the increase of the medium corpuscular hemoglobin (MCH) concentration in blood cells from calves supplemented with 1 gd−1 of IndPPS. Also, medium corpuscular hemoglobin concentration in blood cells showed a quadratic response (P = 0.02) on calves supplemented with 1 gd−1 of IndPPs (). Lymphocytes (P = 0.01) and neutrophils (P = 0.04) showed a quadratic effect with the greater concentrations using 2 gd−1 of supplementation with IndPPs on calves. Finally, there was a quadratic response to IndPPs in segmented neutrophils (P = 0.03) with the greatest concentration using 1 gd−1 on calves ().
Table 3. Effect of an Indian polyherbal phytogenic source (IndPPs) on blood cells of calves.
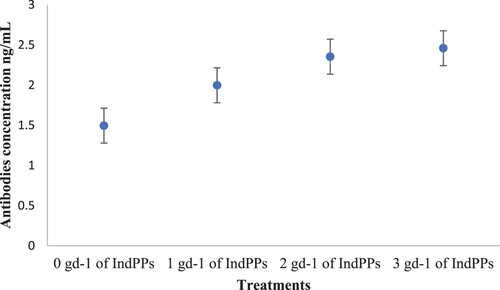
Effects of an Indian polyherbal phytogenic source (IndPPs) on antibodies on plasma blood from calves.
The antibody concentration in plasma increase on the calves that received supplementation using IndPPs compared with the calves without herbal supplementation. Specifically, the concentration of antibodies increase linearly (P < 0.01) as increase the amount of gd−1 of IndPPs supplementation on the calves ().
Discussion
The IndPPS is an herbal extract composed of different medicinal plants native to India and East Asia (Wall et al., Citation2014). In the present study, forty-three organic compounds were detected in IndPPs, some of which contain thymol, cinnamon, caryophyllene, phenols, oleic acid, polyunsaturated fatty acids, and vitamin E (). Previous studies (Sánchez et al., Citation2021; Wall et al., Citation2014) provide solid evidence several of the biocompounds in IndPPs improve growth performance, feed intake, and increase livestock immune response.
Results from in vitro (Castillejos et al., Citation2006; Fraser et al., Citation2007) and in vivo (Cardozo et al., Citation2006; Benchaar et al., Citation2008) studies on effects of the active component of thymol have been contradictory, and more research is needed to understand its effects on rumen fermentation and metabolism in dairy cattle (Benchaar et al., Citation2008). However, thymol has been found to have antimicrobial effects. Thymol has been attributed to disruption of the plasma membrane of bacteria and reduction in glucose uptake (Benchaar et al., Citation2008; Calsamiglia et al., Citation2007). It has been suggested that this occurs through the interaction of its carbonyl group with proteins in the periplasm, inactivating microbial enzymes (Calsamiglia et al., Citation2007; Vakili et al., Citation2013).
Studies reported using cinnamaldehyde (one of the main components of cinnamon essential oil) on ruminants are limited (Wall et al., Citation2014; Castillejos et al., Citation2006). Wall et al. (Wall et al., Citation2014) concluded that cinnamaldehyde supplementation in dairy cows, alone or with other plant secondary metabolites, could affect the feed intake and production performance. Nevertheless, the same authors (Wall et al., Citation2014) suggested that the contrasting results are likely a result of differences in the source and dose of supplement provided, feeding management, parity, and physiological status of the animal. Among the essential oils, eugenol has attracted much attention because of its potential antimicrobial activity against rumen microbes (Benchaar et al., Citation2008). Also, it has been reported in dairy cows that eugenol from herbal sources can increase dry matter intake and milk production (Wall et al., Citation2014). The vitamin E (αtocopherol) status of dairy cattle is an important component of a well-functioning immune system of cows and young dairy calves because of its antioxidant effects (Nehme et al., Citation2021). Experimental reports of vitamin E from herbal formulas are few. Vitamin E content is high in fresh grass, but it decreases markedly during forage storage and conservation. For this reason, NRC (NRC, Citation2001) recommends that the entire vitamin E requirement be given via dietary supplements when conserved forages are fed. Extra supplementation may be useful during immune suppression periods, such as around calving (Nehme et al., Citation2021). Other organic compounds found in the CG-MS analysis of IndPPs were different polyunsaturated fatty acids, oleic acid, phenols, and phytogenics. The presence of polyunsaturated fatty acids in herbs is common (Orzuna-Orzuna et al., Citation2019). However, Nehme et al. (Nehme et al., Citation2021) suggested some benefits of phytogenics interaction with polyunsaturated fatty acids.
There was no significant effect of IndPPs supplementation on average daily gain, final body weight, or final wither height in our study (). Previous studies indicated positive effects on different parameters of production in ruminants. Hernández-Reyes et al. (Hernández-Reyes et al., Citation2017) shown that daily gain in feedlot lambs could be improved by dietary supplementation with polyherbal source. Similarly, in sheep, Dorantes-Iturbide et al. (Dorantes-Iturbide et al., Citation2019) and Orzuna-Orzuna et al. (Orzuna-Orzuna et al., Citation2019) presented results of the use of an herbal formula that contained Asian medicinal plants increase the growth up to 11.1% using 1g−1 by Kg−1 of dry matter intake. However, in the two experimental reports, the supplementation with 2g−1 by Kg−1 of dry matter intake reduces the beneficial effects on different performance variables. This could indicate that the concentration of plant metabolites, such as terpenes, phenols, polyphenols, carotenoids, oligosaccharides, and vitamins, must be precise to benefit from these phytogenic growth promoters (Assan, Citation2018; Lv et al., Citation2021; Quigley, Citation2011). However, Jouany and Morgavi (Jouany & Morgavi, Citation2007) concluded that these metabolites in greater amounts could cause negative effects in the organism. Some of the plants that constitutive IndPPs have been evaluated individually with positive response in broilers (Sánchez et al., Citation2021).
There is no clear explanation of why IndPPs decreased starter diet intake and increased milk consumption, as IndPPs supplementation was offered through gelatins. In lambs, Hernández-Reyes et al. (Hernández-Reyes et al., Citation2017) did not reported changes in DMI using 1.5 gd−1 of an herbal formula, while Orzuna-Orzuna et al. (Orzuna-Orzuna et al., Citation2019) reported an increment of 8% in dry matter intake in lambs supplemented with 1 and 2g−1 by Kg−1 of dry matter. However, Sánchez et al. (Citation2021) obtained similar results to our study. In dairy calves the supplementation with 3, 4 and 5 gd−1 of an herbal formula reduced the started diet intake but increased the milk consumption. Sánchez et al. (Citation2021) described that the difference in the results could be related to the flavor and palatability which could depend on the type and dose of the phytogenic source (Jouany & Morgavi, Citation2007). The number of diarrheas presented a quadratic effect. The explanation may be associated with the effects of the saponins in gut motility. Although Mendel et al. (Mendel et al., Citation2017) indicate that it is not possible to generalize the effect of saponins in ruminants, it appears that the myorelaxant effect and the antispasmodic effects of saponins have some maximum dose that may cause a disequilibrium in calves that changes conditions in their gut and its microbiota that results in a higher incidence of diarrhea. Calves’ gut in early stages will behave as pre-ruminant, but in later stages, as the rumen is developed, saponins could affect their bacteria and protozoa (Mendel et al., Citation2017).
Several medicinal plants from India have shown hypoglycemic effects (Khosla et al., Citation2000) similar to those of other herbal products evaluated in calves (Sánchez et al., Citation2021). El Barky et al. (El-Barky et al., Citation2017) reviewed the different hypoglycemic effects of different levels of plant saponins on insulin (modifying signaling, rejuvenation, release), glycogen synthesis, gluconeogenesis inhibition and reduction of mRNA gene expression of glycogen phosphorylase and glucose 6-phosphatase and increased the relative mRNA expression of insulin-regulated glucose transporter 4 (GLUT4). Other researchers interpret the lower glucose values in calves supplemented with phytogenics as an improved capacity to metabolize glucose (Sánchez et al., Citation2021; El-Barky et al., Citation2017). Like these experimental studies, our results showed lower serum glucose concentration in calves that received supplementation with IndPPs, relative to calves without the supplement (). The quadratic response in blood butyrate suggests that phytogenic secondary metabolites could affect rumen microbiota development or have an indirect effect by reducing solid intake and causing slow rumen development as observed by Sánchez et al. (Citation2021) in dairy calves supplemented with an herbal formula. It has been recognized that volatile fatty acids, particularly butyrate, affect papillary development (Mentschel et al., Citation2001). Heinrichs and Lesmeister (Heinrichs & Lesmeister, Citation2004) documented the differences in rumen development when calves received only milk versus milk supplemented with forage or a concentrated diet. Using a microarray approach, Connor et al. (Connor et al., Citation2013) described that feeding the growing calf could affect several gene networks involved in the metabolism and morphology of rumen epithelium. Additionally, Nakamura et al. (Nakamura et al., Citation2018) reported that secondary metabolites from the phytogenic could affect gene expression of proton-linked monocarboxylate transporters 1 (MCT1), which can alter the rumen the increased butyrate in the bloodstream. There is no clear explanation of why the supplementation with IndPPS increased the total protein without changes in globulins or albumins or plasma proteins (). A study in Boer goats with graded levels of infusion tea using similar types of phytogenic showed changes in serum total protein (Hu et al., Citation2006). Zhang et al. (Zhang et al., Citation2013) also reported that the supplementation with a Chinese herbal formula based on Astragalus to the basal diet could enhance protein metabolism (increased serum protein) in broiler chickens. Their findings are in good agreement with our present observation in dairy calves. Some secondary metabolites could explain the changes observed in bilirubin from herbal supplements containing polyphenolic compounds caused inhibitory effects on UDP Glucuronosyltransferase Family 1 Member A9 (UGT1A9), the gene responsible for glucuronidation of bilirubin (Mohamed & Frye, Citation2011), which may alter the bilirubin concentrations, and other secondary metabolites could cause cross-reactions responsible for increasing bilirubin, preventing liver injury (Dufour et al., Citation2000). The values of ALP increased linearly in the calves supplemented with IndPPs. An ALP value between 17.5 and 387 U/L is considered normal (Hatate et al., Citation2016). During the growth of newborn calves, the ALP value has been described as such a bone marker, suggesting that it indicates bone metabolic and skeleton formation (Hatate et al., Citation2016). It is not clear at which level the phytogenic affected the synthesis of ALP, but it could have been at the intestinal level (Riaz et al., Citation2014). Exist few scientific reports that described the ALP response in livestock, so that further studies should examine the clinical utility of bone metabolic in neonatal calves.
The supplementation with IndPPs linearly increased hematocrit, hemoglobin, and erythrocytes on the blood cells of the calves (). Panousis et al. (Hashemzadeh-Cigari et al., Citation2014) described the hematology reference intervals for neonatal Holstein calves. A hematocrit value between 12.40% and 42.8% is considered normal, while a hemoglobin value is considered normal with a minimum of 4.3–15.4 g/dl as maximum. The concentration of erythrocytes was established normal between 4.46 and 11.90 × 106/ml. Our results fall within the normal range; however, it is interesting to observe a greater concentrations of these values in the calves with 2 gd−1 of IndPPs. These suggest that IndPPs have a greater effect on the liver, likely indicating liver changes in response to IndPPs metabolites as observed in other plants and herbal formulas (Sánchez et al., Citation2021; Riaz et al., Citation2014). Quadratic responses were observed in lymphocytes, and segmented neutrophils in blood cells from calves supplemented with 1 and 2 gd−1 of IndPPs (). These changes are within the reference limits for calves (Panousis et al., Citation2018). Therefore, our results do not indicate a pathologic process, bone marrow disorder, or autoimmune disease (Roland et al., Citation2014), but rather suggest that some metabolites of the herbal mixture send stimuli to the immune system that is being interpreted as stress (Roland et al., Citation2014; Kampen et al., Citation2006). This could be considered positive since cattle have a small bone marrow reserve for granulocytes (Kampen et al., Citation2006). Thus, subpopulations of the lymphocytes and neutrophils may be stimulated, reducing the neutrophils in the band (Roland et al., Citation2014; Kampen et al., Citation2006).
In most of the little research on the effects of herbal formulas on dairy calves, attention is focused on secondary metabolites (Sánchez et al., Citation2021; Frankič et al., Citation2009; Mendel et al., Citation2017) but herbal formulas also contain volatile compounds (Sánchez et al., Citation2021; Mendoza et al., Citation2019). In any case, herbal formulas are nutraceutical mixes that can positively stimulate the immune system (Sánchez et al., Citation2021). Another study reported a decrease in other diseases that most commonly occur during calving and calf growth (Gutiérrez et al., Citation2019). It may be that the herbal formulas act in multiple ways through nonspecific mechanisms of the phytogenic on antioxidants, immunomodulation, anti-inflammatories, antimicrobial compounds, intestinal microbiota modulation, and others that are being detected in gene expression modulation (Roque-Jiménez et al., Citation2020; Frankič et al., Citation2009; Vakili et al., Citation2013). Combining plants has additive effects since evaluations of individual plants do not show changes in hematological parameters (Bharti et al., Citation2004) or immune response (Chand et al., Citation2014).
Finally, the increase in antibodies by IndPPs supplementation could be related to immune-stimulation through many pathways (). In contrast to antibiotics, herbal formulas and essential oils contain different antibacterial agents that possibly employ several inhibitory mechanisms, making it difficult for pathogens to initiate resistance (Aljarallah, Citation2017). In non-ruminants, thymol has been reported to suppress pathogens in the small intestine without damaging beneficial commensal colonic bacteria (Aljarallah, Citation2017). Saleh et al. (Saleh et al., Citation2018) reported that thymol negatively affected Salmonella typhimurium, Escherichia coli, Staphylococcus aureus, and Clostridium perfingers. These results suggest that the combination of antimicrobials in herb mixtures controls microflora in the gastrointestinal tract, enhancing feed digestibility and growth performance (Frankič et al., Citation2009).
Conclusion
We evaluated the effects of including a Indian polyherbal phytogenic sources in the early-life diet of dairy calves on productive performance, health, blood serum biochemistry, blood cells, and antibodies. Significant changes by Indian polyherbal phytogenic sources were observed in milk and started diet intake, blood serum protein, blood serum bilirubin, blood serum alkaline phosphatase, hematocrit, hemoglobin, erythrocytes, lymphocytes, and segmented neutrophils. These results were attributed to the Indian polyherbal phytogenic sources containing bioactive substances with phytogenic properties, which have a possible role in stimulating the immune system. The supplementation with Indian polyherbal phytogenic sources had no impact on growth, but it increases antibody count. Future research should focus on elucidating the effect of varying amounts of individual bio-compounds from herbal formulas on calving and calf early life, their metabolomic effect, and the subsequent health outcomes in adult life.
Acknowledgements
We are grateful to the owners and management of the dairy farms where we conducted this study. We are also grateful to Nuproxa for providing the Indian polyherbal phytogenic sources.
Disclosure statement
No potential conflict of interest was reported by the author(s).
References
- Aljarallah, K. M. (2017). Conventional and alternative treatment approaches for Clostridium difficile infection. International Journal of Health Sciences, 11(1), 1–10.
- Assan, N. (2018). Plant based feed additives (phytogenic) as a primary solution to an antibiotic free nutritional program and feeding strategy in rabbit production. Journal of Animal Science, 7, 493–503. doi:10.14196/sjas.v7i3.2521
- Benchaar, C., Calsamiglia, S., Chaves, A. V., Fraser, G. R., Colombatto, D., McAllister, T. A., & Beauchemin, K. A. (2008). A review of plant-derived essential oils in ruminant nutrition and production. Animal Feed Science and Technology, 145, 209–228. doi:10.1016/j.anifeedsci.2007.04.014
- Bharti, M. K., Dhuria, R. K., Sharma, T., & Garg, D. D. (2004). Haematobiochemical profile of magra sheep during feeding of sares (Albizia lebbeck) leaves. Modern Veterinary Practice, 5(1), 48–50. https://scholar.google.com/scholar_lookup?hl=en&volume=5&publication_year=2004&pages=48-50&issue=1&author=M.+K.+Bharti&author=R.+K.+Dhuria&author=T.+Sharma&author=D.+D.+Garg&title=Haematobiochemical+profile+of+magra+sheep+during+feeding+of+sares+%28Albizia+lebbeck%29+leaves
- Björkman, I., Röing, M., Sternberg-Lewerin, S., Stålsby-Lundborg, C., & Eriksen, J. (2021). Animal Production with Restrictive Use of Antibiotics to Contain Antimicrobial Resistance in Sweden—A Qualitative Study. Frontiers in Veterinary Science, 7, 1197. doi:10.3389/fvets.2020.619030
- Britton, R., Stock, R., Sindt, M., Oliveros, B., & Parrott, C. (1990). New feed additive and technique to evaluate acidosis in cattle. MP-University of Nebraska, Agricultural Experiment Station. United States of America. https://scholar.google.com/scholar_lookup?hl=en&publication_year=1990&author=R.+Britton&author=R.+Stock&author=M.+Sindt&author=B.+Oliveros&author=C.+Parrott&title=New+feed+additive+and+technique+to+evaluate+acidosis+in+cattle
- Calsamiglia, S., Busquet, M., Cardozo, P. W., Castillejos, L., & Ferret, A. (2007). Invited review: Essential oils as modifiers of rumen microbial fermentation. Journal of Dairy Science, 90(6), 2580–2595. doi:10.3168/jds.2006-644
- Cardozo, P. W., Calsamiglia, S., Ferret, A., & Kamel, C. (2006). Effects of alfalfa extract, anise, capsicum and a mixture of cinnamaldehyde and eugenol on ruminal fermentation and protein degradation in beef heifers fed a high concentrate diet. Journal of Animal Science, 84, 2801–2808. doi:10.2527/jas.2005-593
- Castillejos, L., Calsamiglia, S., & Ferret, A. (2006). Effect of essential oil active compounds on rumen microbial fermentation and nutrient flow in in vitro systems. J. Dairy. Sci, 89(7), 2649–2658. doi:10.3168/jds.S0022-0302(06)72341-4
- Chand, N., Naz, S., Shah, Z., Khan, S., Ali, S. A., & Khan, R. U. (2014). Growth performance and immune status of broilers fed graded levels of Albizia lebbeck seeds. Pakistan Journal of Zoology, 46(2), 574–577. https://scholar.google.com/scholar_lookup?hl=en&volume=46&publication_year=2014&pages=574-577&issue=2&author=N.+Chand&author=S.+Naz&author=Z.+Shah&author=S.+Khan&author=S.+A.+Ali&author=R.+U.+Khan&title=Growth+performance+and+immune+status+of+broilers+fed+graded+levels+of+Albizia+lebbeck+seeds
- Cho, Y. I., & Yoon, K. J. (2014). An overview of calf diarrhea - infectious etiology, diagnosis, and intervention. Journal of Veterinary Science, 15(1), 1–17. doi:10.4142/jvs.2014.15.1.1
- Connor, E. E., Baldwin, V. I. R. L., Li, C., Li, R. W., & Chung, H. (2013). Gene expression in bovine rumen epithelium during weaning identifies molecular regulators of rumen development and growth. Functional & Integrative Genomics, 13(1), 133–142. doi:10.1007/s10142-012-0308-x
- Crosby, M., Mendoza-Martínez, G. D., Relling, A., Vázquez-Valladolid, A., Lee-Rangel, H. A., Martínez, J. A., & Oviedo, M. (2017). Influence of supplemental choline on milk yield, fatty acid profile, and postpartum weight changes in suckling ewes. Journal of Dairy Science, 100(125). https://scholar.google.com/scholar_lookup?hl=en&volume=100&publication_year=2017&pages=125&issue=Suppl+2&author=M.+Crosby&author=G.+D.+Mendoza-Mart%C3%ADnez&author=A.+Relling&author=V.+A.+V%C3%A1zquez&author=H.+A.+Lee-Rangel&author=J.+A.+Mart%C3%ADnez&author=M.+Oviedo&title=Influence+of+supplemental+choline+on+milk+yield%2C+fatty+acid+profile%2C+and+postpartum+weight+changes+in+suckling+ewes
- Dorantes-Iturbide, G., Lara-Bueno, A., Orzuna-Orzuna, J. F., Mendoza-Martínez, G. D., Miranda-Romero, L. A., & Hernández-García, P. A. (2019). Efecto de la mezcla herbal Animunin en las características de la canal de corderos en finalización. Rev Acad Ciênc Anim, 17(1), 183–185.
- Dufour, D. R., Lott, J. A., Nolte, F. S., Gretch, D. R., Koff, R. S., & Seeff, L. B. (2000). Diagnosis and monitoring of hepatic injury. I. Performance characteristics of laboratory tests. Clin Chem, 46(12), 2027–2049. doi:10.1093/clinchem/46.12.2027
- El-Barky, A. R., Hussein, S. A., Alm-Eldeen, A. A., Hafez, A., & Mohamed, T. M. (2017). Saponins and their potential role in diabetes mellitus. Diabetes Management, 7(1), 148–158. https://scholar.google.com/scholar_lookup?hl=es-US&volume=7&publication_year=2017&pages=148-158&issue=1&author=A.+R.+El+Barky&author=S.+A.+Hussein&author=A.+A.+Alm-Eldeen&author=A.+Hafez&author=T.+M.+Mohamed&title=Saponins+and+their+potential+role+in+diabetes+mellitus
- Frankič, T., Voljč, M., Salobir, J., & Rezar, V. (2009). Use of herbs and spices and their extracts in animal nutrition. Acta Agric Slov, 94, 95–102. https://scholar.google.com/scholar_lookup?hl=es-US&volume=94&publication_year=2009&pages=95-102&issue=2&author=T.+Franki%C4%8D&author=M.+Volj%C4%8D&author=J.+Salobir&author=V.+Rezar&title=Use+of+herbs+and+spices+and+their+extracts+in+animal+nutrition
- Fraser, G. R., Chaves, A. V., Wang, Y., McAllister, T. A., Beauchemin, K. A., & Benchaar, C. (2007). Assessment of the effects of cinnamon leaf oil on rumen microbial fermentation using two continuous culture systems. Journal of Dairy Science, 90(5), 2315–2328. doi:10.3168/jds.2006-688.
- Gutiérrez, A. R., Gutiérrez, A., Sánchez, C., & Mendoza, G. D. (2019). Effect of including herbal choline in the diet of a dairy herd; a multiyear evaluation. Emir J Food Agric, 31, 477–481. doi:10.9755/ejfa.2019.v31.i6.1971
- Hashemzadeh-Cigari, F., Khorvash, M., Ghorbani, G. R., Kadivar, M., Riasi, A., & Zebeli, Q. (2014). Effects of supplementation with a phytobiotics-rich herbal mixture on performance, udder health, and metabolic status of Holstein cows with various levels of milk somatic cell counts. Int J Dairy Sci, 97(12), 7487–7497. doi:10.3168/jds.2014-7989
- Hatate, K., Shinya, K., Matsuo-Sato, A., Sasaki, S., Devkota, B., Takahashi, M., Hirata, T., & Yamagishi, N. (2016). Changes in the plasma levels of several bone markers in newborn calves during the first two days of life. Journal of Veterinary Medical Science, 78(2), 337–340. doi:10.1292/jvms.15-0358
- Heinrichs, A. J., & Lesmeister, K. E. (2004). Rumen development in the dairy calf. Calf and heifer rearing: Principles of rearing the modern dairy heifer from calf to calving. 60th University of Nottingham Easter School in Agricultural Science, Nottingham, UK (pp. 53–65); March 23–24 2004.
- Hernández-Reyes, J. C., Lara-Bueno, A., Miranda-Romero, L. A., Mendoza-Martínez, G. D., & Martínez-Gómez, D. (2017). Evaluación de productos herbales como aditivos en raciones de finalización de ovinos. Congreso Bienal AMENA. México.
- Hu, W., Liu, J., Wu, Y., Guo, Y., & Ye, J. (2006). Effects of tea saponins on in vitro ruminal fermentation and growth performance in growing Boer goat. Archives of Animal Nutrition, 60, 89–97. doi:10.1080/17450390500353119
- Jouany, J. P., & Morgavi, D. P. (2007). Use of “natural” products as alternatives to antibiotic feed additives in ruminant production. Animals, 1(10), 1443–1466. doi:10.1017/S1751731107000742
- Kampen, A. H., Olsen, I., Tollersrud, T., Storset, A. K., & Lund, A. (2006). Lymphocyte subpopulations and neutrophil function in calves during the first 6 months of life. Veterinary Immunology and Immunopathology, 113(1-2), 53–63. doi:10.1016/j.vetimm.2006.04.001
- Kazemi-Bonchenari, M., Falahati, R., Poorhamdollah, M., Heidari, S. R., & Pezeshki, A. (2018). Essential oils improved weight gain, growth and feed efficiency of young dairy calves fed 18 or 20% crude protein starter diets. Journal of Animal Physiology and Animal Nutrition, 102, 652–661. doi:10.1111/jpn.12867
- Khosla, P., Bhanwra, S., Singh, J., Seth, S., & Srivastava, R. K. (2000). A study of hypoglycaemic effects of Azadirachta indica (neem) in normal and alloxan diabetic rabbits. Indian Journal of Physiology and Pharmacology, 44(1), 69–74. https://scholar.google.com/scholar_lookup?hl=en&volume=44&publication_year=2000&pages=69-74&issue=1&author=P.+Khosla&author=S.+Bhanwra&author=J.+Singh&author=S.+Seth&author=R.+K.+Srivastava&title=A+study+of+hypoglycaemic+effects+of+Azadirachta+indica+%28neem%29+in+normal+and+alloxan+diabetic+rabbits
- Lv, R., Elsabagh, M., Obitsu, T., Sugino, T., & Kurokawa, Y. (2021). Effect of phytol in forage on phytanic acid content in cow’s milk. Anim Biosci, 34(10), 1616–1622. doi:10.5713/ab.21.0086
- Maciel, P. R., Miranda, N. N. J., Restle, J., Oliveira, B. U., Chaves, M. R. F., João, F. A., Soares, F. C. M., & de Oliveira, A. R. (2016). Performance, rumen development, and carcass traits of male calves fed starter concentrate with crude glycerine. R Bras Zootec, 45, 309–318. doi:10.1590/S1806-92902016000600005
- McGuirk, S. M. (2008). Disease management of dairy calves and heifers. The Veterinary Clinics of North America. Food Animal Practice, 24(1), 139–153. doi:10.1016/j.cvfa.2007.10.003
- Mendel, M., Chłopecka, M., Dziekan, N., & Karlik, W. (2017). Phytogenic feed additives as potential gut contractility modifiers—A review. Animal Feed Science and Technology, 230, 30–46. doi:10.1016/j.anifeedsci.2017.05.008
- Mendoza, G. D., Oviedo, M. F., Pinos, J. M., Lee-Rangel, H. A., Vázquez, A., Flores, R., & Pérez, F. (2019). Milk production in dairy cows supplemented with herbal choline and methionine. Rev Fac Cien Agrarias, 1, 1–12. https://scholar.google.com/scholar_lookup?hl=en&volume=52&publication_year=2019&pages=332-343&issue=1&author=G.+D.+Mendoza&author=M.+F.+Oviedo&author=J.+M.+Pinos&author=H.+A.+Lee-Rangel&author=A.+V%C3%A1zquez&author=R.+Flores&author=F.+P%C3%A9rez&author=A.+Roque&author=O.+Cifuentes&title=Milk+production+in+dairy+cows+supplemented+with+herbal+choline+and+methionine
- Mentschel, J., Leiser, R., Műlling, C., Pfarrer, C., & Claus, R. (2001). Butyric acid stimulates rumen mucosa development in the calf mainly by a reduction of apoptosis. Archiv für Tierernaehrung, 55(2), 85–102. doi:10.1080/17450390109386185
- Mirman, D. (2014). Growth curve analysis and visualizationusing R. Chapman & Hall/CRC. https://scholar.google.com/scholar_lookup?hl=en&publication_year=2014&author=D.+Mirman&title=Growth+curve+analysis+and+visualizationusing+R
- Mohamed, M. E., & Frye, R. F. (2011). Inhibitory effects of commonly used herbal extracts on UDP-glucuronosyltransferase 1A4, 1A6, and 1A9 enzyme activities. Drug Metabolism and Disposition, 39(9), 1522–1528. doi:10.1124/dmd.111.039602
- Nakamura, S., Haga, S., Kimura, K., & Matsuyama, S. (2018). Propionate and butyrate induce gene expression of monocarboxylate transporter 4 and cluster of differentiation 147 in cultured rumen epithelial cells derived from preweaning dairy calves. Animal Science Journal, 96(11), 4902–4911. doi:10.1093/jas/sky334
- Nehme, R., Andrés, S., Pereira, R. B., Ben Jemaa, M., Bouhallab, S., Ceciliani, F., López, S., Rahali, F. Z., Ksouri, R., Pereira, D. M., & Abdennebi-Najar, L. (2021). Essential Oils in Livestock: From Health to Food Quality. Antioxidants, 10(330). doi:10.3390/antiox10020330
- NRC. (2001). Nutrient requirements of dairy cattle (7th rev ed. National Research Council. United States of America. doi:10.17226/9825
- Orzuna-Orzuna, J. F., Lara-Bueno, A., Dorantes-Iturbide, G., Mendoza-Martinez, G. D., MirandaRomero, L. A., Hernández-García, P. A., & López-Ordaz, R. (2019). Efecto de la mezcla herbal Animunin en el comportamiento productivo de corderos en finalización. Rev Acad Ciênc Anim, 17(1), 180–182.
- Panousis, N., Siachos, N., Kitkas, G., Kalaitzakis, E., Kritsepi-Konstantinou, M., & Valergakis, G. E. (2018). Hematology reference intervals for neonatal Holstein calves. Research in Veterinary Science, 118, 1–10. doi:10.1016/j.rvsc.2018.01.002
- Pardon, B., Hostens, M., Duchateau, L., Dewulf, J., De Bleecker, K., & Deprez, P. (2013). Impact of respiratory disease, diarrhea, otitis and arthritis on mortality and carcass traits in white veal calves. BMC Veterinary Research, 9(79). doi:10.1186/1746-6148-9-79
- Parish, A. J., Bourg, M. B., Marks, L. M., Simmons, B. N., & Smith, T. (2012). Evaluation of different methods of cattle hip height data collection. App Anim Sci, 28, 292–299. doi:10.15232/S1080-7446(15)30358-2
- Quigley, J. (2011). Direct fed microbials (probiotics) in calf diets. Bov. Alliance. Manag. Nutr, 1(4). https://scholar.google.com/scholar_lookup?hl=en&publication_year=2011&author=J.+Quigley&title=Direct+fed+microbials+%28probiotics%29+in+calf+diets
- Riaz, A., Khan, R. A., Mirza, T., Mustansir, T., & Ahmed, M. (2014). In vitro/in vivo effect of Citrus limon (L. Burm. f.) juice on blood parameters, coagulation, and anticoagulation factors in rabbits. Pak J Pharm Sci, 27. https://pubmed.ncbi.nlm.nih.gov/25015459/
- Roland, L., Drillich, M., & Iwersen, M. (2014). Hematology as a diagnostic tool in bovine medicine. Journal of Veterinary Diagnostic Investigation, 26(5), 592–598.
- Roque-Jiménez, J. A., Mendoza-Martínez, G. D., Vázquez-Valladolid, A., MdlL, G.-G., Flores-Ramírez, R., Pinos-Rodriguez, J. M., Loor, J. J., Relling, A. E., & Lee-Rangel, H. A. (2020). Supplemental Herbal Choline Increases 5-hmC DNA on Whole Blood from Pregnant Ewes and Offspring. Animals, 10(8), 1277. doi:10.3390/ani10081277
- Saleh, A. A., Ebeid, T. A., & Abudabos, A. M. (2018). Effect of dietary phytogenics (herbal mixture) supplementation on growth performance, nutrient utilization, antioxidative properties, and immune response in broilers. Environ Sci Pollut Res, 25(15), 14606–14613. doi:10.1007/s11356-018-1685-z
- Sánchez, N., Lee Rangel, H. A., Martínez-Cortés, I., Mendoza, G. D., Hernández, P. A., Espinoza, E., Vazquez-Valladolid, A., Flores-Ramírez, R., Roque-Jiménez, A., Campillo-Navarro, M., & Relling, A. E. (2021). A polyherbal phytogenic additive improved growth performance, health, and immune response in dairy calves. Food Agr Immunol, 32(1), 482–498. doi:10.1080/09540105.2021.1967296
- Seifzadeh, S., Aghjehgheshlagh, F. M., Hossein, A., Seifdavati, J., & Navidshad, B. (2017). The effects of a medical plant mix and probiotic on performance and health status of suckling Holstein calves. Ital J Anim Sci, 16(1), 44–51. doi:10.1080/1828051X.2016.1249421
- Vakili, A. R., Khorrami, B., Mesgaran, M. D., & Parand, E. (2013). The effects of thyme and cinnamon essential oils on performance, rumen fermentation and blood metabolites in holstein calves consuming high concentrate diet. Asian-Australasian Journal of Animal Sciences, 26(7), 935–944. doi:10.5713/ajas.2012.12636
- Wall, E. H., Perry, H. D., Doane, S., Donkin, S., & Bravo, D. (2014). The effects of supplementation with a blend of cinnamaldehyde and eugenol on feed intake and milk production of dairy cows. Journal of Dairy Science, 97(9), 5709–5717. doi:10.3168/jds.2014-7896
- Zhang, G. G., Yang, Z. B., Wang, Y., & Yang, W. R. (2013). Effects of Astragalus membranaceus root processed to different particle sizes on growth performance, antioxidant status, and serum metabolites of broiler chickens. Poultry Science, 92, 178–183. doi:10.3382/ps.2012-02432