Article Title: Efficacy and safety comparison of combination of 0.04% tretinoin microspheres plus 1% clindamycin versus their monotherapy in patients with acne vulgaris: a phase 3, randomized, double-blind study
Authors: S. Dogra, T. K. Sumathy, C. Nayak, G. Ravichandran, P.P Vaidya, S. Mehta, R. Mittal, A. Mane, and S.N. Charugulla
Journal: Journal of Dermatological Treatment
DOI: 10.1080/09546634.2020.1720579
When the above article was published online, the authors’ names are not in initials. The authors’ names should be as follows:
S. Dogra, T. K. Sumathy, C. Nayak, G. Ravichandran, P.P Vaidya, S. Mehta, R. Mittal, A. Mane, and S.N. Charugulla
Also, Figure 2 has been published incorrectly. Below is the correct figure: 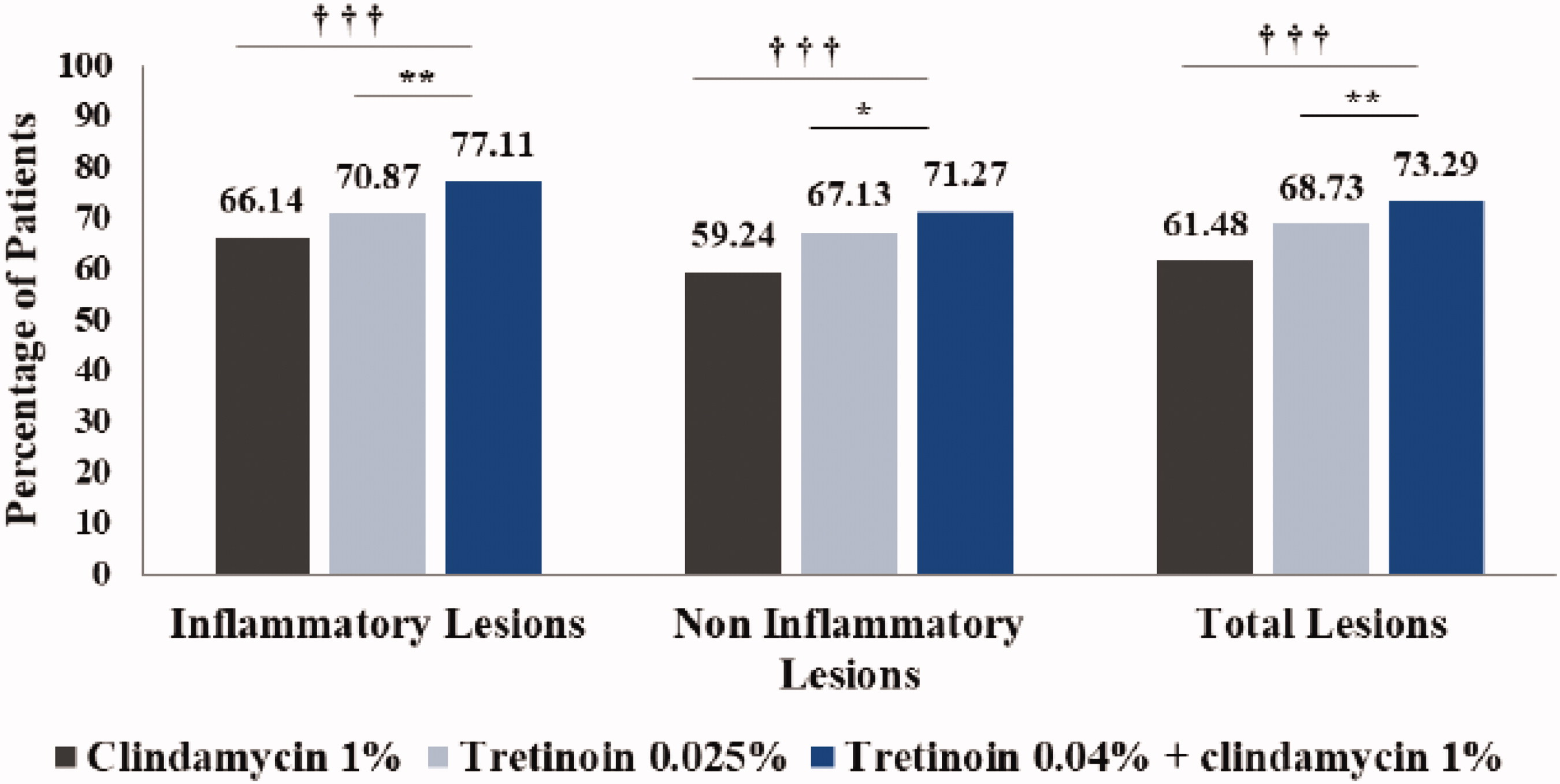
The Acknowledgement section has also been published with missing information. Below is the correct acknowledgement:
Akshada Deshpande, PhD and Jyothi Ramanathan, PhD (both from SIRO Clinpharm Pvt. Ltd., India) provided writing assistance for this manuscript. The authors thank the study participants without whom this study would not have been accomplished, and the investigators:- Dr. A. Gugle, Dr. B. J. Shah, Dr. C. B. Mhaske, Dr. D. V. S. Pratap, Dr. G. Kiran, Dr. G. Prasad, Dr. H. R. Yogeesh, Dr. H. S. Chopade, Dr. H. V. Nataraja, Dr. J. Betkerur, Dr. J. Martis, Dr. M. Kuruvila, Dr. M. Parekh, Dr. M. Philip, Dr. N. Khanna, Dr. N. Sarma, Dr. P. Padmaja, Dr. P. Shukla, Dr. R. Agarwal, Dr. R. Dhurat, Dr. R. P. Singh, Dr. R. Rathod, Dr. R. Shah, Dr. S. C. Bharija, Dr. S. Sengupta, Dr. Y. S. Marfatia, for their contribution to this study.
The last paragraph of the manuscript should be a conclusion.
Conclusion:
Taken together, these findings suggest that the novel microsphere technology allows high concentration of tretinoin to be delivered at the acne site in a controlled manner for a longer duration and thus reducing irritation. The discontinuation rates can be a critical factor for patients’ adherence to treatment amongst available tretinoin and clindamycin combinations in Indian scenario, thus this controlled-release, once-daily, gel-based formulation of tretinoin and clindamycin could be considered as a potential option to improve patients’ compliance and overall therapeutic outcomes.
