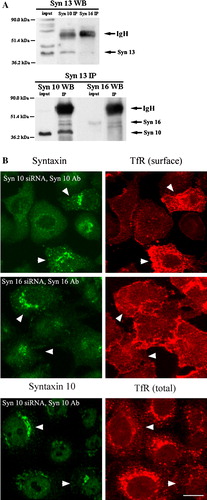
The authors of the paper entitled “Trans-Golgi network syntaxin 10 functions distinctly from syntaxins 6 and 16” (by Wang et al.,) in Molecular Membrane Biology, 22(4), 313–325, apologise for the absence of arrowheads in . A corrected version of the Figure is shown below.
Figure 8. Syntaxin 10 interacts with syntaxin 12/13 and its depletion alters the steady state localization of transferrin receptor. (a). HeLa cell lysates were subjected to immunoprecipitation with antibodies against syntaxin 12/13, 10 or 16. Immunoprecipitates were eluted, resolved on 8% SDS-PAGE gels, and subjected to Western immunoblot analyses with antibodies against Vps45. Input is 1/7.5 of the lysate used for the immunoprecipitation. Representative blots are shown. (b). Cells were transiently transfected with the respective siRNA oligomers for 48 h. Cell were then incubated with culture supernatants (diluted 1:10 with basal RPMI medium) of the transferrin receptor (TfR) monoclonal antibody producing Okt9 hybridoma for 20 min on ice. For the assessment of cell surface TfR levels (surface), cells were then washed extensively, fixed with 4% paraformaldehyde and incubated with primary antibodies against syntaxins 10 or 16, followed by Texas-red or FITC-labeled secondary antibodies. For the assessment of internal TfR levels (internal), cells were washed extensively, immersed in complete RPMI medium, transferred to 37°C, and incubated for 30 min. Cell were then washed, fixed with 4% paraformaldehyde and incubated with primary antibodies against syntaxin 10, followed by Texas-red or FITC-labeled secondary antibodies. Arrowheads indicate cells where syntaxin 10 or 16 knockdown is incomplete or minimal. Bar = 10 µm. This figure is reproduced in colour in Molecular Membrane Biology online.