 ?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.ABSTRACT
Concern about the use of antimicrobials in food producing animals is increasing. The study objective was to quantify antimicrobial drug usage in calves using antimicrobial treatment records from Irish suckler beef and dairy farms. Antimicrobial treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed. Calves were followed from birth (day 0) until 6 months of age. According to standard farm protocol, calves exhibiting clinical signs of any disease were identified and antimicrobial treatment was administered. Farmers recorded the following information for each treatment administered: calf identification, age at treatment, disease event, drug name, number of treatment days, and amount of drug administered. In total, 3,204 suckler beef calves and 5,358 dairy calves, representing 540,953 and 579,997 calf-days at risk, respectively, were included in the study. A total of 1,770 antimicrobial treatments were administered to suckler beef (n = 841) and dairy calves (n = 929) between birth and 6 months of age. There was large variation in TIDDDvet and TIDCDvet by farm. This study provides new insights into the time periods and indications for which specific antimicrobial substances are used in Irish dairy and beef suckler calves.
1. Introduction
Antimicrobial resistance, the ability of (pathogenic) bacteria to withstand the action of antimicrobials, is a growing global public and animal health concern. An increasing number of studies have shown that antimicrobial usage in humans (Charbonneau et al. Citation2006; Costelloe et al. Citation2010; Sun et al. Citation2012) and animals (Burow et al. Citation2013; Hammerum et al. Citation2014; Simoneit et al. Citation2015) is a key driver for the development of antimicrobial resistance, although an environmental role in the antimicrobial resistance development has been proposed as a new driver (Chamosa et al. Citation2017).
In 2009, the European Surveillance of Veterinary Antimicrobial Consumption (EMA ESVAC Citation2015, Citation2016) project was launched, following a request form the European Commission (EC). The EC (DG ENTR and DG SANCO) requested the European Medicines Agency (EMA), among other activities, to develop a harmonized approach for the collection and reporting of data based on national sales figures, combined with estimations of usage in at least major groups of species (poultry, pigs, veal calves, other ruminants, pets and fish). Estimates of antimicrobial usage, based on sales data, vary across Europe, in terms of the total amounts sold (per kilogram of animal biomass) and in the different antimicrobial classes used and their routes of administration (EMA ESVAC Citation2015; Carmo, Schüpbach, et al. Citation2017; Carmo, Nielsen, et al. Citation2017, Citation2018; Carmo, Bouzalas, et al. Citation2018). Lists of critically important antimicrobials are available and used widely for the treatment and prevention of infectious diseases in humans (WHO Citation2011, Citation2012, Citation2016) and animals (OIE Citation2015). In 2017, guidelines were published on technical units and appropriate indicators for quantification of antimicrobial usage (Collineau et al. Citation2017). The defined daily dose for animals (DDDvet) is the assumed average dose per kg animal per species per day. The defined course dose for animals (DCDvet) was defined as the assumed average dose per kg animal per species per treatment course. Priority has been given to assignment of DDDvet and DCDvet for broilers, cattle and pigs. DDDvet and DCDvet are technical units of measurement solely intended for the reporting of antimicrobial consumption data.
Farm-level antimicrobial usage data are now collected in several European countries, including, Denmark (Wielinga et al. Citation2014; DANMAP Citation2013), the Netherlands (Bos et al. Citation2013; Speksnijder et al. Citation2015; Autoriteit Diergeneesmiddelen Citation2016) and France (Jarrige et al. Citation2017). In Switzerland, the use of an antibiotic usage database will become mandatory for treatments of groups of animals from January 2019. From October 2019, the database will become mandatory for the recording of all antimicrobial treatments (Personal communication Gertraud Schüpbach-Regula). In Europe, various monitoring programmes have outlined antimicrobial consumption for animals through annual antimicrobial sales data (DANMAP Citation2013; ANMV Citation2014; Veldman et al. Citation2017). These programmes are structured to observe trends at the national level and for comparison of data between years and countries (Grave et al. Citation2010; ECDC/EFSA/EMA Citation2015; EMA Citation2015). In Ireland, calves for beef production come from two sources, the beef suckler herd and the dairy herd, which comprise ca. 0.9 and 1.4 million cows, respectively. These herds are mainly spring-calving, to coincide with the seasonal grass growth profile, and thus, ruminant production systems in Ireland are predominantly grass-based. In suckler beef production systems, spring-born calves have continuous access to their dams at pasture until the end of the (first) grazing season in autumn, when they are weaned and housed indoors (Drennan and McGee Citation2009). Gradual weaning of the beef calf in autumn typically combines a number of physical and psychological stressors (e.g. Lynch et al. Citation2010; O’Loughlin et al. Citation2012). In dairy beef production systems calves are artificially-reared, usually indoors, on milk replacer and concentrates (plus forage) and, after gradual weaning, are turned out to pasture for their (first) grazing season (Keane and Moloney Citation2010; Keane et al. Citation2011). The period between birth and 180 days of age represents a time of high risk for suckler beef and artificially reared dairy calves; national survey results indicate that 4.0% and 5.7% of live-born beef and dairy calves die during this period (AIM Citation2016).
The main objective of the present study was to quantify antimicrobial drug usage in calves using antimicrobial treatment records from Irish suckler beef and dairy farms. A secondary objective was to compare usage in dairy and beef suckler calves, and examine the variation in antimicrobial consumption over time from birth to 6 months of age.
2. Materials and methods
2.1. Farmer recruitment and participation
Data were obtained from a longitudinal study on herd-level factors associated with the health and survival of calves on Irish farms (hereafter referred to as the herd-level study). The herd-level study was conducted between 1 July 2014 and 31 December 2015 Farmers, enrolled in the herd-level study, recorded birth, disease and antimicrobial treatment, and death information on their calves using standardized recording sheets. Case definitions () were provided to the farmers to assist with the classification of disease. Farmers were responsible for detecting, diagnosing, and administering treatment to any calf exhibiting clinical signs of disease, and encouraged to consult with their veterinarian when making antimicrobial treatment decisions. Farmers enrolled in the herd-level study completed and submitted the project recording sheets on a monthly basis. Health data were available for calves on 79 suckler beef and 44 dairy farms from the herd-level study. The number of calves involved in the study, days at risk and standard weights are presented in .
Table 1. Case definitionsa provided to the farmers to assist with classification of disease events.
Table 2. Number of farms, calves, days at risk and standard calf body weights during the study.
Crude and cause-specific morbidity were determined using the health data collected from the project recording sheets. All antimicrobial treatment data were reviewed and the following criteria applied to differentiate between disease events: long-acting antibiotics administered more than 7 days apart, or other medications administered more than 3 days apart were classified as separate disease events (Windeyer et al. Citation2014). Crude morbidity was defined as calves being treated for at least one disease event, attributed to any cause, excluding injury. Calves treated for illnesses other than diarrhoea, BRD, navel infection, or joint infection/lameness were categorized as receiving treatment for other disease events.
Cumulative incidence and incidence rate of crude and cause-specific morbidity were calculated for the following treatment at risk periods: birth (day 0) to 30 days of age, 31–90 days of age, 91–180 days of age, and birth (day 0) to 180 days of age. Both cumulative incidence and incidence rate were calculated as measures of disease frequency throughout the study period. Cumulative incidence was calculated as the number of calves treated for disease within each age category, relative to the number of calves at risk of disease (Dohoo et al. Citation2009). Incidence rate was calculated as the number of disease events (all occurrences, not just the first event) treated within each age category, relative to the total animal-time at risk (Dohoo et al. Citation2009). Animal-time at risk was calculated for each individual calf as the number of days from birth until it was either sold off the home farm, died, or the observation period ended. Birth, movement, and death dates for the animal-time at risk calculations were retrieved from the project recording sheets, as well as the Animal Identification and Movement (AIM Citation2015; Department of Agriculture, Food and the Marine, Co. Dublin, Ireland) and Irish Cattle Breeding Federation (Bandon, Co. Cork, Ireland) databases. Total animal-time at risk for all calves included within each age category was determined.
2.4. Antimicrobial usage
A list of all antimicrobials prescribed by veterinarians and administered by the farmer was drawn up in one Excel file in order to facilitate further consultation and calculations. The antibiotics were categorized according to ESVAC recommendations, tetracyclines, amphenicols, penicilins, 1st and 2nd generation cephalosporins, 3rd and 4rd generation cephalosporins, sulfonamides, trimethoprim, macrolides, lincosamides, fluoroquinolones, other quinolones, aminoglycosides, polymyxins, pleuromutilins and others.
The reporting recommendations of Collineau et al. (Citation2017) were followed in the current study and the indicator selected to report the drug consumption was the Treatment Incidence (TI). The used daily dose (UDDvet) was calculated as the amount of an antimicrobial drug administered during a given period (days) divided by the number of calves at risk and their average live weight at the beginning of the treatment. Defined daily dose for animals (DDDvet) and defined course dose for animals (DCDvet) were the technical units used to measure antimicrobial consumption. The treatment incidence was defined as the number of animals per 1000 receiving a UDDvet, a DDDvet or a UCDvet, also expressed as the percentage of animal life expectancy treated with one UDDvet, one DDDvet or one UDDvet (Timmerman et al. Citation2006; Collineau et al. Citation2017).
The DDDvet represents the assumed average dose per kg animal per species per day. DCDvet represents the assumed average dose per kg animal per species per treatment course.
The Treatment Incidence (TI) provides a standardized technical unit of measurement that quantifies how many animals out of a theoretical group of 1000 animals receive daily an antimicrobial treatment. Or, if one animal would live for a theoretical period of 1000 days, how many of these days it would have been treated with an antimicrobial. The calculations applied were:
The total amount of active substance administered was determined from the antimicrobial records. DDDvet and DCDvet for cattle, as assigned by the European Medicines Agency, were used in the calculations. Body weights (BW) were calculated for each treatment period for suckler beef and dairy calves, respectively. Total number of calf-days at risk was determined using movement data from the Animal Identification and Movement central database. Antimicrobials were grouped by class according to ESVAC recommendations.
2.5. Data handling and statistical analyses
All data were analyzed using SAS 9.4 (SAS Institute Inc., Cary, North Carolina, USA). Data were evaluated by descriptive statistics. Normality of data was assessed using the Shapiro–Wilks normality test. Generalized linear mixed models were constructed to evaluate associations between calf type (suckler beef versus dairy) and morbidity within each age category. Initially, each morbidity response was treated as a dichotomous outcome variable (disease occurred or did not) and these data were modelled with a binomial distribution, a logit link function, and a random effect to account for within-farm correlation (GLIMMIX Procedure, SAS 9.4, SAS Institute Inc., Cary, North Carolina, USA). Subsequently, each morbidity response was treated as a count outcome variable (number of disease events) and these data were modelled with a Poisson distribution, a log link function, offset as the natural logarithm of animal-time at risk, and a random effect to account for within-farm correlation (GLIMMIX Procedure, SAS 9.4, SAS Institute Inc., Cary, North Carolina, USA). The logistic regression was used to estimate the odds ratio (OR) and the poisson regression was used to estimate the incidence rate ratio (IRR).
Calf type was included as a dichotomous explanatory variable in each of the morbidity models. UDDvet, TIUDDVET, TIDDDVET and TIDCDVET displayed a non-normal distribution. A Mann–Whitney test was performed in order to detect differences between beef and dairy farms. The UDDvet, TIUDDVET, TIDDDVET and TIDCDVET are presented as mean with minimum and maximum values.
3. Results
3.1. Antimicrobial usage
A total of 1,770 antimicrobial treatments were administered to suckler beef (n = 841) and dairy calves (n = 929) between birth and 180 days of age. The most frequent indication (%) for antimicrobial group treatment, and number of drugs used, were calf diarrhoea (53.8%; 23 drugs), respiratory disease (31.0%; 19 drugs), navel ill (6.1%; 10), joint ill (2.4%; 9 drugs) and injuries (0.98%; 6 drugs). The number of calves included from birth to 180 days of age was 3,204 and 5,358 in beef and dairy farms respectively, resulting in 540,953 and 579,997 days at risk, respectively. No statistical differences (P > 0.05) were observed between beef and dairy farms for UDDvet, TIUDDvet, TIDDDvet and TIDCDvet. The population corrected unit (PCU) was calculated as number of animals by standard body weight in each period and in addition mg/PCU was calculated as a sum of all antibiotics used expressed in mg divided by the corresponding PCU. The number of calves, days at risk, body weights and mg/PCU for the three studied periods (birth to 30 days of age; 31–90 days of age, 91–180 days of age and overall from birth to 180 days of age) are summarized in .
3.2. UDDvet
A relatively large variation between farms was observed regarding antimicrobial use. From birth (day 0) to 180 days, the sulfonamide class of antimicrobials had the greatest UDDvet with beef and dairy farms having values of 12.1 (0–133), 18.4 (0–462), respectively (; ). First and second generation cephalosporins and lincosamines were not used in any of the farms.
Figure 1. UDDVet by antibiotic group, period and farm type. Antimicrobial treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
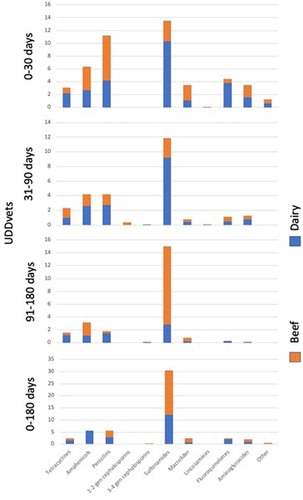
Table 3. UDDVet by antibiotic group, period and farm type. Health treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
From birth to 30 days of age, sulfonamides (10.4; 0–250) and penicillins (6.9; 0–67.5) had the greatest UDDvet in beef and dairy farms, respectively. Cephalosporins were not used in any of the farms while lincosamines were not used in any of the dairy farms (; ).
From 31 to 90 days of age, sulfonamides displayed the greatest UDDvet (9.2; 0–158 and 2.6, 0–82, for beef and dairy farms, respectively) and first and second generation cephalosporins were not used in beef farms, while third and fourth generation cephalosporins and lincosamines were not used in dairy farms. Spectinomycin was not used in beef or in dairy farms (; ).
From 91 to 180 days, sulfonamides showed the greatest UDDvet, 2.9 (0–100) and 12.1 (0–359) for beef and dairy farms, respectively. Cephalosporins, lincosamines or spectinomycin were not used in any studied farm (; ).
3.3. TIUDDvet
From Birth to 180 days of age, the greatest TIUDDvet was for fluoroquinolones,in beef 13.1(0–307) and dairy farms 26.5(0–740) (; ).
Figure 2. TIUDDVet by antibiotic group, period and farm type. Antimicrobial treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
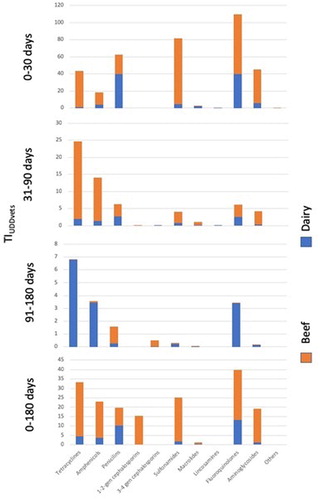
Table 4. TIUDD Vet by antibiotic group, period and farm type. Health treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
From birth to 30 days of age, the greatest TIUDDvet in beef farms were 40.0 (0–640) and 40.0 (0–1014) for penicillins and fluoroquinolones respectively, with sulfonamides being numerically greater in dairy farms (77.0; 0–1490) than in beef farms (4.9; 0–67) (; ).
From 31 to 90 days of age, the antimicrobial groups with the most used treatments based on TIUDDvet were penicillins (2.7; 0–168) and fluoroquinolons (2.6; 0–118) in beef farms, and tetracyclines in dairy farms (23.0; 0–709) (; ).
From 91 to 180 days of age, tetracyclines and penicillins were the most used treatments in beef (6.8; 0–526) and dairy (1.3; 0–35) farms (; ).
From birth to 180 days of age, fluoroquinolones and penicillins were the most used treatments on beef farms (10.2 (0–254); 0.65 (0–4.8)) and corresponding treatment values for dairy farms were 26.5 (0–740) and 29.0 (0–740) (; ).
3.4. TIDDDvet
From birth to 30 days of age, the greatest TIDDDvet was observed with penicillins (10.1; 0–143) in beef farms, and for fluoroquinolones (8.5; 0–223) in dairy farms (; ).
Figure 3. TIDDDVet by antibiotic group, period and farm type. Antimicrobial treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
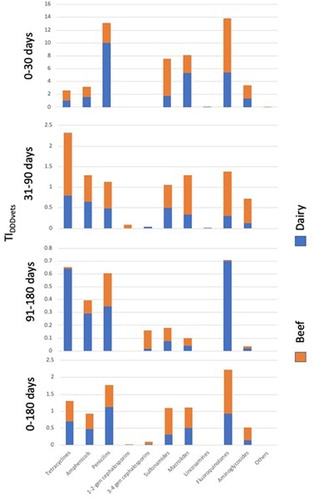
Table 5. TIDDD Vet by antibiotic group, period and farm type. Health treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
From 31 to 90 days of age, tetracyclines had the greatest TIDDDvet (0.8; 0–29 and 1.5; 0–42) in beef and dairy farms, respectively. From 91 to 180 days of age, fluoroquinolones (beef, 0.70 (0–53); dairy, 0.01 (0–0.26) and penicillins (beef, 0.4 (0–17); dairy, 0.3 (0–5.4)) had the greatest TIDDDvet in beef and dairy farms, respectively (; ).
3.5. TIDCDvet
The TIDCDvet values are presented in . From birth to 180 days of age, the most used antibiotics were penicillins and fluoroquinolones (0.3 (0–3.5) and 0.4 (0–9.3)) in beef and dairy farms, respectively (; ). From birth to 30 days of age, the greatest TIDDDvet was observed for penicillins (beef, 2.8 (0–41); dairy, 0.8 (0–7)) in beef calves and for fluoroquinolones (beef, 1.9 (0–69); dairy, 2.4 (0–59)) in dairy calves (; )
Figure 4. TIDCDVet by antibiotic group, period and farm type. Antimicrobial treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
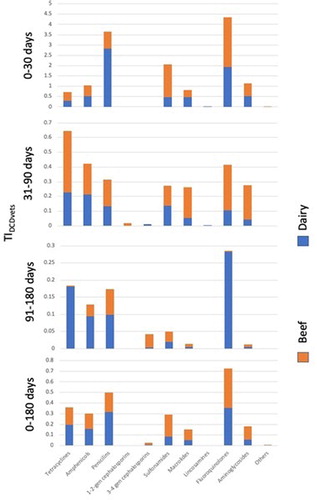
Table 6. TIDCD Vet by antibiotic group, period and farm type. Health treatment records for calves born between 1 July 2014 and 30 June 2015 on 79 suckler beef and 44 dairy farms were analyzed.
3.6. Morbidity and mortality
Morbidity results are presented in . In the first 30 days of age, suckler beef calves had greater cumulative incidence and incidence rates of navel (P < 0.05) and joint infection/lameness (P < 0.05) compared to dairy calves. From 31 to 90 days of age, beef calves had greater cumulative incidence and incidence rate of BRD (P < 0.05) than dairy calves. Conversely, the incidence rate of diarrhoea among dairy calves in the first 180 days of life was greater than that of suckler beef calves (P < 0.05). From birth to 80 days of age the cumulative index of BRD, navel infection, and joint infection/lameness (P < 0.05) were greater in beef than in dairy calves, while the differences were only observed (beef greater than dairy) for incidence rate of navel infection/lameness (P < 0.05).
Median age at first treatment for crude morbidity in suckler beef and dairy calves was 14 (min. = 0, Q1 = 8, Q3 = 43, max. = 155) and 13 (min. = 0, Q1 = 7, Q3 = 20, max. = 145) days, respectively. Median age at first treatment for diarrhoea in suckler beef and dairy calves was 13 (min. = 0, Q1 = 8, Q3 = 23, max. = 83) and 12 (min. = 0, Q1 = 7, Q3 = 19, max. = 117) days, respectively. Median age at first treatment for BRD in suckler beef and dairy calves was 48 (min. = 0, Q1 = 31, Q3 = 96, max. = 155) and 20 (min. = 0, Q1 = 11, Q3 = 30, max. = 145) days, respectively. Median age at first treatment for navel infection in suckler beef and dairy calves was 7 (min. = 2, Q1 = 5, Q3 = 12, max. = 30) and 18 (min. = 0, Q1 = 6, Q3 = 27, max. = 35) days, respectively. Median age at first treatment for joint infection/lameness in suckler beef and dairy calves was 24 (min. = 5, Q1 = 11, Q3 = 52, max. = 174) and 37 (min. = 7, Q1 = 20, Q3 = 91, max. = 93) days, respectively. Median age at first treatment for other disease events in suckler beef and dairy calves was 49 (min. = 1, Q1 = 16, Q3 = 73, max. = 153) and 27 (min. = 0, Q1 = 12, Q3 = 88, max. = 139) days, respectively.
4. Discussion
4.1. Calf disease
The calves included in the present study represent the two systems of calf rearing in Ireland; (1) dairy calf-to-beef and (2) Suckler calf-to-beef. Farms recruited to this study were representative of commercial practice in Ireland. In this study, antimicrobial usage refers to the exposure of a given animal or group of animals over a period of time to the active substance in each antimicrobial that was administered. We have provided the first detailed information on antimicrobial usage in suckler beef and artificially reared dairy calves from birth to 180 days of age. This study not only determined the diseases that were targeted for antimicrobial treatment, but also the specific antimicrobial classes that were used. Only animals showing signs of disease were treated with antimicrobials. No mass administration of antimicrobials was practiced. This is in contrast to Pardon, Catry, et al. (Citation2012) and Bos et al. (Citation2013) where healthy animals received medication. In the present study, overall, 20.4% of suckler beef calves and 14.8% of dairy calves exhibited clinical signs of disease and were treated for at least one disease event by 180 days of age. The leading cause of morbidity from birth to 180 days of age in the present study was diarrhoea, accounting for 44 and 77% of the disease events in suckler beef and dairy calves, respectively. The second and third most frequent causes of morbidity in calves during the first 180 days of life were BRD and navel infection, respectively. These results are not unexpected. Diarrhoea and BRD are widely reported as the two main causes of calfhood morbidity and mortality for beef and dairy calves in other European countries (Svensson et al. Citation2003; Gulliksen et al. Citation2009; Slavík et al. Citation2009; Pardon, De Bleeker, et al. Citation2012) and North America (Wittum and Perino Citation1995; Dewell et al. Citation2006; Waldner and Rosengren Citation2009; Henderson et al. Citation2011; Windeyer et al. Citation2014).
The highest risk period for disease in the present study was between birth and 30 days of age, with approximately two-thirds of all disease events occurring during this time period. The first 30 days of life, often referred to as the neonatal period in calves, is known to be associated with high levels of morbidity and mortality (Wittum and Perino Citation1995; Sivula et al. Citation1996; Slavík et al. Citation2009; Windeyer et al. Citation2014). More than 90% of diarrhoea treatments were administered during the first 30 days of life, which would be considered the high risk period for diarrhoea (Foster and Smith Citation2009; Smith Citation2012). The veterinarians’ experience of using a particular drug to treat a specific condition is also an important consideration when prescribing an antimicrobial. Consequently, the prescribing behaviour of veterinarians in cattle practice is of importance, however, collection of data relating to this was not an objective of the present study.
4.2. Antimicrobial usage
In Ireland, antimicrobials may only be supplied for use in animals under veterinary prescription in accordance with European Communities (Animal Remedies) (No. 2) Regulations 2007. Many papers on the usage of antimicrobials in farm animals (pigs and veal calves mostly) have been published, more especially dealing with group treatments (Pardon, Catry, et al. Citation2012; Bos et al. Citation2013; Jarrige et al. Citation2017). Pardon, Catry, et al. (Citation2012) calculated the TI, based on the DDDA (TIDDDA) and the UDDA (TIUDDA) and the live weight of the animals, as 414 and 379 calves treated with one daily dose of antimicrobial agents per 1000 animals, respectively.
For this reason, different lists of DDD values (also called ADD before the consensus) have been drawn up, depending on the study. These values have been taken from the summary of products characteristics (SPCs) of drugs marketed in the country of interest. Further investigations and comparisons on the drug usage were carried out by some authors (Jarrige et al. Citation2017) by means of units of measurements like the PDD (Prescribed Daily Dose) and the UDD (Used Daily Dose). The principles for assignment of DDDvet (and DCDvet) are harmonized with the principles for assignment of DDDs in human medicine to the greatest extent possible. In human medicine only DDDs have been assigned. Assignment of DDDvet and DCDvet is generally based on SPC information on dosing collected from nine volunteer European Union Member States (Czech Republic, Denmark, Finland, France, Germany, the Netherlands, Spain, Sweden and United Kingdom).
4.3. Quantifying antimicrobial usage
A number of different approaches exist for measuring antimicrobial usage, the most commonly reported ones are; defined daily dose (DDDvet) or defined course dose (DCDvet) and mg of antimicrobials used per kg of livestock (mg/PCU [population corrected unit]). The DDDvet and the DCDvet methods are an estimation of doses or courses of an antibiotic per animal (European Medicines Agency Citation2015, Citation2016). The methods are not without their limitations; for example, mg/PCU may have the potential to encourage incomplete courses of antibiotics, or selection of antimicrobials simply based on a low mg/kg dose rate. DDDvet and DCDvet overcome some issues by assigning a standard dose/course rate across products and analyzing how many doses or courses are prescribed per animal. However, DDDvet and DCDvet take no clear account of the actual amount (mg) of active agent used.
The values of mg/PCU in the present study are lower than those reported by Van Boeckel et al. (Citation2015) with 45 mg/PCU being indicated as a global estimation for cattle. Van Boeckel et al. used PCU values to estimate global consumption of antimicrobials per kg of animal produced at 45 mg/PCU which provides an appreciation for the overall use of antimicrobials within livestock species but does not indicate usage within the various stages of the production system.
In agreement with our finding, Sherwin and Down (Citation2018) reported that the main diseases of calves in the first month of life in calves were diarrhoea, septicaemia and bovine respiratory disease with sulphonamides being the class of drug that was most frequently administered.
Another interesting finding with antimicrobial treatment in the present study was the large number of different antimicrobials used per disease condition, including the use of critically important cephalosporins, fluoroquinolones, penicillins, and tetracyclines. However, the antimicrobial treatments that were used in Ireland were similar to other European countries with the exception of colistin, which was not used.
5. Conclusion
This study provides the first detailed information into on-farm usage of antimicrobials in suckler beef and artificially reared dairy calves from birth to 180 days of age, in Ireland. It was concluded that beef and dairy calves in the study population were treated with antimicrobial substances for diseases at a relatively low frequency (mainly individual treatments).
Acknowledgements
This research was supported under the Department of Agriculture, Food and the Marine Research Stimulus Fund (11/S/131) with Dr. B. Earley as the Principal Investigator. Research partners included University College Dublin, Irish Cattle Breeding Federation, and Animal Health Ireland (Technical Working Group on Calf Health, CalfCare®). The authors acknowledge: the participating farmers and their Teagasc KT advisors for their contribution to the research; Post-Doctoral Researcher (Todd, C), Technician (Butler, O.), administrative staff (Doggert, D., Gilsenan, A., Murphy, M., Weldon, M.) and under-graduate students (Aili, M., Browne, E., Calvin, E., Ceriani, F., Conway, A., Doyle, P., Drought, Y., Hoch, A., Kane, S., Kervick, M., Lefevre, H., LeRet, S., Loayan, Mas, V., Mulligan, J., Palmer, R., Scacchi, A., van Giersbergen, M.) with data collection at Teagasc Grange.
Disclosure statement
No potential conflict of interest was reported by the authors.
Additional information
Funding
References
- AIM . 2015. Department of agriculture, food and the marine, 2015. Animal identification and movement bovine statistics report 2015.
- AIM . 2016. Department of agriculture, food and the marine, 2016. AIM Bovine statistics report 2016. Backweston Campus, Celbridge, Co. Kildare.
- ANMV . 2014. Sales survey of veterinary medicinal products containing antimicrobials in France − 2013. Annual report, France. French Agency for Veterinary Medicinal Products [accessed 2016 June 26]. https://www.anses.fr/en/system/files/ANMV-Ra-Antibiotiques2013EN.pdf .
- Autoriteit Diergeneesmiddelen (Netherlands Veterinary Medicines Authority, sDa) . 2016. Trends, benchmarking of livestock farms and veterinarians, and a revision of the benchmarking method. [Accessed 2016 Dec 23]. http://www.autoriteitdiergeneesmiddelen.nl .
- Bos MEH , Taverne FJ , van Geijlswijk IM , Mouton JW , Mevius DJ , Heederik DJJ. 2013. Consumption of antimicrobials in pigs, veal calves, and broilers in The Netherlands: quantitative results of nationwide collection of data in 2011. PLoS One. 8. doi:10.1371/journal.pone.0077525.
- Burow E , Simoneit C , Tenhagen BA , Keasbohrer A. 2013. Oral antimicrobials increase antimicrobial resistance in porcine E. coli – a systematic review. Prev Vet Med. 113:364–375. doi:10.1016/j.prevetmed.2013.12.007.
- Carmo LP , Bouzalas I , Nielsen LR , Alban L , Martins da Costa P , Müntener C , Schüpbach G , Abreu Y , Magouras I. 2018. Expert opinion on livestock antimicrobial usage indications and patterns in Denmark, Portugal and Switzerland. Vet Rec Open. 5(1):e000288. doi:10.1136/vetreco-2018-000288. eCollection 2018.
- Carmo LP , Nielsen LR , Alban L , da Costa PM , Schüpbach-Regula G , Magouras I. 2018. Veterinary expert opinion on potential drivers and opportunities for changing antimicrobial usage practices in livestock in Denmark, Portugal, and Switzerland. Front Vet Sci. 5(1):29. doi:10.3389/fvets.2018.00029. eCollection 2018.
- Carmo LP , Nielsen LR , Alban L , Müntener CR , Schüpbach-Regula G , Magouras I. 2017. Comparison of antimicrobial consumption patterns in the Swiss and Danish cattle and swine production (2007–2013). Front Vet Sci. 2(4):26. doi:10.3389/fvets.2017.00026. eCollection 2017.
- Carmo LP , Schüpbach-Regula G , Müntener C , Chevance A , Moulin G , Magouras I. 2017. Approaches for quantifying antimicrobial consumption per animal species based on national sales data: a Swiss example, 2006 to 2013. Euro Surveill. 22(6). doi:10.2807/1560-7917.ES.2017.22.6.30458.
- Chamosa LS , Álvarez VE , Nardelli M , Quiroga M-P , Cassini MH , Centrón D. 2017. Lateral antimicrobial resistance genetic transfer is active in the open environment. Sci Rep. 7:513. doi:10.1038/s41598-017-00600-2.
- Charbonneau P , Parienti JJ , Thibon P , Ramakers M , Daubin C , du Cheyron D , Lebouvier G , Coutour XL , Leclercq R. 2006. Fluoroquinolone use and methicillin-resistant Staphylococcus aureus isolation rates in hospitalized patients: a quasi experimental study. Clin Infect Dis. 42:778–784. doi: 10.1086/500319
- Collineau L , Belloc C , Stark KDC , Hemonic A , Postma M , Dewulf J , Chauvin C. 2017. Guidance on the selection of appropriate indicators for quantification of antimicrobial usage in humans and animals. Zoonoses Public Health. 64:165–184. doi:10.1111/zph.12298.
- Costelloe C , Metcalfe C , Lovering A , Mant D , Hay AD. 2010. Effect of antibiotic prescribing in primary care on antimicrobial resistance in individual patients: systematic review and meta-analysis. BMJ. 340:c2096. doi:10.1136/bmj.c2096.
- DANMAP . 2013. Use of antimicrobial agents and occurence of antimicrobial resistance in bacteria from food animals, food and humans in Denmark. Ministry of Health, Ministry of Science, Innovation and Higher Education, Ministry of Food, Agriculture and Fisheries, report, Denmark.
- Dewell RD , Hungerford LL , Keen JE , Laegreid WW , Griffin DD , Rupp GP , Grotelueschen DM. 2006. Association of neonatal serum immunoglobulin G1 concentration with health and performance in beef calves. J Am Vet Med Assoc. 228:914–921. doi:10.2460/javma.228.6.914.
- Dohoo I , Martin W , Stryhn H. 2009. Veterinary epidemiologic research. 2nd ed. Charlottetown : VER Inc.
- Drennan MJ , McGee M. 2009. Performance of spring-calving beef suckler cows and their progeny to slaughter on intensive and extensive grassland management systems. Livest Sci. 120:1–12. doi:10.1016/j.livsci.2008.04.013.
- ECDC/EFSA/EMA . 2015. ECDC/EFSA/EMA first joint report on the integrated analysis of the consumption of antimicrobial agents and occurrence of antimicrobial resistance in bacteria from humans and food-producing animals. EFSA J. 13:4006. (Stockholm/Parma/London). doi: 10.2903/j.efsa.2015.4006
- European Medicine Agency [EMA] . 2015. Sales of veterinary antimicrobial agents in 25 EU/EEA countries in 2015. In: Fifth ESVAC report. European Medicines Agency [accessed 2016 June 26]. http://www.ema.europa.eu/docs/enGB/documentlibrary/Report/2015/10/WC500195687.pdf .
- European Medicines Agency [EMA] . 2016. Defined daily doses for animals (DDDvet) and defined course doses for animals (DCDvet). [accessed 2016 Sep 22]. http://www.ema.europa.eu/ema/pages/includes/document/open_document.jsp?webContentId=WC500205408 .
- European Medicines Agency, European Surveillance of Veterinary Antimicrobial Consumption [ESVAC] . 2016. Sales of veterinary antimicrobial agents in 29 European countries in 2014: trends across 2011 to 2014 – Sixth ESVAC report, 2016. http://www.ema.europa.eu/docs/en_GB/document_library/Report/2016/10/WC500214217.pdf .
- Foster DM , Smith GW. 2009. Pathophysiology of diarrhea in calves. Vet Clin North Am Food Anim Pract. 25:13–36. doi:10.1016/j.cvfa.2008.10.013.
- Grave K , Torren-Edo J , Mackay D. 2010. Comparison of the sales of veterinaryantibacterial agents between 10 European countries. J Antimicrob Chemoth. 65:2037–2040. doi:10.1093/jac/dkq247.
- Gulliksen SM , Lie KI , Østerås O. 2009. Calf health monitoring in Norwegian dairy herds. J Dairy Sci. 92:1660–1669. doi:10.3168/jds.2008-1518.
- Hammerum AM , Larsen J , Andersen VD , Lester CH , Skovgaard Skytte TS , Hansen F , Olsen SS , Mordhorst H , Skov RL , Aarestrup FM , Agerso Y. 2014. Characterization of extended-spectrum β-lactamase (ESBL)-producing Escherichia coli obtained from Danish pigs, pig farmers and their families from farms with high or no consumption of third- or fourth-generation cephalosporins. J Antimicrob Chemother. 69:2650–2657. doi:10.1093/jac/dku180.
- Henderson L , Miglior F , Sewalem A , Kelton D , Robinson A , Leslie KE. 2011. Estimation of genetic parameters for measures of calf survival in a population of Holstein heifer calves from a heifer-raising facility in New York State. J Dairy Sci. 94:461–470. doi:10.3168/jds.2010-3243.
- Jarrige N , Cazeau G , Morignat E , Chanteperdrix M , Gay E. 2017. Quantitative and qualitative analysis of antimicrobial usage in white veal calves in France. Prev Vet Med. 144:158–166. doi:10.1016/j.prevetmed.2017.05.018.
- Keane MG , Dunne PG , Kenny DA , Berry DP. 2011. Effects of genetic merit for carcass weight, breed type and slaughter weight on performance and carcass traits of beef × dairy steers. Animal. 5:182–194. doi: 10.1017/S1751731110001758
- Keane MG , Moloney AP. 2010. Comparison of pasture and concentrate finishing of Holstein-Friesian, Aberdeen Angus × Holstein-Friesian and Belgian Blue × Holstein-Friesian steers. Ir J Agric Food Res. 49:11–26.
- Lynch EM , Earley B , McGee M , Doyle S. 2010. Effect of abrupt weaning at housing on leukocyte distribution, functional activity of neutrophils, and acute phase protein response of beef calves. BMC Vet Res. 6:39. doi:10.1186/1746-6148-6-39.
- OIE . 2015. http://www.oie.int/fileadmin/Home/eng/Our_scientific_expertise/docs/pdf/Eng_OIE_List_antimicrobials_May2015.pdf .
- O’Loughlin A , Lynn DJ , McGee M , Doyle S , McCabe M , Earley B. 2012. Transcriptomic analysis of the stress response to weaning at housing in bovine leukocytes using RNA-seq technology. BMC Gen. 13:250. doi:10.1186/1471-2164-13-250.
- Pardon B , Catry B , Dewulf J , Persoons D , Hostens M , De bleecker K , Deprez P. 2012. Prospective study on quantitative and qualitative antimicrobial and anti-inflammatory drug use in white veal calves. J Antimicrob Chemother. 67:1027–1038. doi:10.1093/jac/dkr570.
- Pardon B , De Bleecker K , Hostens M , Callens J , Dewulf J , Deprez P. 2012. Longitudinal study on morbidity and mortality in white veal calves in Belgium. BMC Vet Res. 8:26. doi:10.1186/1746-6148-8-26.
- Sherwin G , Down P. 2018. Calf immunology and the role of vaccinations in dairy calves. In Pract. 40:102–114. doi: 10.1136/inp.k4889
- Simoneit C , Burow E , Tenhagen B-A , Kasbohrer A. 2015. Oral administration of antimicrobials increase antimicrobial resistance in E. coli from chicken – a systematic review. Prev Vet Med. 118:1–7. doi:10.1016/j.prevetmed.2014.11.010.
- Sivula N , Ames T , Marsh W , Werdin R. 1996. Descriptive epidemiology of morbidity and mortality in Minnesota dairy heifer calves. Prev Vet Med. 27:155–171. doi:10.1016/0167-5877(95)01000-9.
- Slavík P , Illek J , Brix M , Musilová L , Rajmon R , Klabanová P , Jílek F. 2009. Health status of beef cows and their calves in the Czech Republic. Acta Vet Brno. 78:47–56. doi:10.2754/avb200978010047.
- Smith DR. 2012. Field disease diagnostic investigation of neonatal calf diarrhea. Vet Clin North Am – Food Anim Pract. 28(3):465–481. doi:10.1016/j.cvfa.2012.07.010.
- Speksnijder DC , Mevius DJ , Bruschke CJM , Wagenaar JA. 2015. Reduction of veterinary antimicrobial use in the Netherlands. The Dutch success model. Zoonoses Public Health. 62:79–87. doi:10.1111/zph.12167.
- Sun L , Klein EY , Laxminarayan R. 2012. Seasonality and temporal correlation between community antibiotic use and resistance in the United States. Clin Infect Dis. 55:687–694. doi:10.1093/cid/cis509.
- Svensson C , Lundborg K , Emanuelson U , Olsson S-O. 2003. Morbidity in Swedish dairy calves from birth to 90 days of age and individual calf-level risk factors for infectious diseases. Prev Vet Med. 58:179–197. doi:10.1016/S0167-5877(03)00046-1.
- Timmerman T , Dewulf J , Catry B , Feyen B , Opsomer G , de Kruif A , Maes D. 2006. Quantification and evaluation of antimicrobial drug use in group treatments for fattening pigs in Belgium. Prev Vet Med. 74:251–263. doi:10.1016/j.prevetmed.2005.10.003.
- Van Boeckel TP , Brower C , Gilbert M , Grenfell BT , Levin SA , Robinson TP , Teillant A , Laxminarayan R. 2015. Global trends in antimicrobial use in food animals. Proc Nat Acad Sci. 112:5649–5654. doi:10.1073/pnas.1503141112.
- Veldman KT , Wit B , Van Pelt W , Heederik DJJ , Mevius DJ. 2017. MARAN 2017: monitoring of antimicrobial resistance and antibiotic usage in animals in the Netherlands in 2016. Combined with NETHMAP-2017: consumption of antimicrobial agents and antimicrobial resistance among medically important bacteria in the Netherlands. Technical report 30 June 2017, Affiliation: http://www.wur.nl Affiliation: WUR/RIVM.
- Waldner CL , Rosengren LB. 2009. Factors associated with serum immunoglobulin levels in beef calves from Alberta and Saskatchewan and association between passive transfer and health outcomes. Can Vet J. 50(3):275–281.
- Wielinga PR , Jensen VF , Aarestrup FM , Schlundt J. 2014. Evidence-based policy for controlling antimicrobial resistance in the food chain in Denmark. Food Contr. 40:185–192. doi:10.1016/j.foodcont.2013.11.047.
- Windeyer MC , Leslie KE , Godden SM , Hodgins DC , Lissemore KD , LeBlanc SJ. 2014. Factors associated with morbidity, mortality, and growth of dairy heifer calves up to 3 months of age. Prev Vet Med. 113:231–240. doi:10.1016/j.prevetmed.2013.10.019.
- Wittum TE , Perino LJ. 1995. Passive immune status at postpartum hour 24 and long-term health and performance of calves. Am J Vet Res. 56:1149–1154.
- World Health Organization [WHO] . 2011. http://apps.who.int/iris/bitstream/handle/10665/77376/9789241504485_eng.pdf;jsessionid=E4D933BF6E38D34779845DEC4C6CFB9E?sequence=1 .
- World Health Organization [WHO] . 2012. Advisory group on integrated surveillance of antimicrobial resistance (AGISAR). 2012. Critically important antimicrobials for human medicine. 3rd review 2011. World Health Organisation.
- World Health Organization [WHO] . 2016. Critically important antimicrobials for human medicine. 4th revision. 2016. p. 1–31.
