ABSTRACT
The taste of food is an essential characteristic for cats and has been shown to affect food selection. However, understanding of food selection by cats using taste characteristics is far from complete. Therefore, the aim of the present review was to summarize the current knowledge on food preference and the role of taste on this selection in domestic cats. Appetite regulation is one of the determinants of palatability in cats and involves a highly complex interplay between hypothalamus, adipose tissue, and digestive tract. However, knowledge on this interplay is scarce in cats. When evaluating different foods for cats, behavioural responses such as facial expressions involving the movements and positions of ears, tongue, and head can provide increased insight into the effectiveness of formulating a more palatable diet. This paper also reviews food additives currently used in industry for enhancing the palatability of cat foods. In summary, a better understanding of the factors that affect the food preference in cats is essential to produce high-quality foods because cats will not eat a food with a flavour they dislike even though it is complete and nutritionally balanced.
1. Introduction
There are two hypotheses about the origin of the domestic cats. The first one presumes that domestic cats originates from Felis silvestris lybica (the African wildcat) and the second hypothesis claims that they should be considered to be the subspecies of Felis silvestris catus which is assumed to have originated from wild cats living in the Middle East (Clutton-Brock Citation1999; Randi and Ragni Citation1991). Recent evidence shows that feline domestication approximately occurred 9000 years ago (Vigne et al. Citation2004). Domestic cat is one of the most popular companion animals throughout the world (Mameno et al. Citation2017). According to a recent report, nearly 35% of the US households own at least a cat (Pallotto et al. Citation2018). Approximately 50% of all pet market is comprised of pet food. The global pet food market size was nearly 75 billion dollars and the United States was the largest market which was valued at around 25 billion dollars in 2016 (Phillips-Donaldson Citation2016).
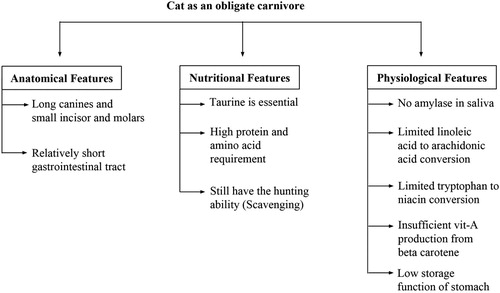
Although there has been an ongoing debate on whether dogs are omnivore or strict carnivore, the cat is considered to be a strict carnivore. Therefore, cats have a relatively higher protein and essential amino acid requirements than those of dogs (Salaun et al. Citation2017). Amylase is not present in cat’s saliva and their gastrointestinal tract is relatively short compared to omnivores so they can digest meat much faster than vegetables (NRC Citation2006). Cats lack the enzyme called ‘β-carotene 15,15 –dioxygenase’ and therefore, they cannot convert beta carotene to vitamin A and need to get vitamin A directly from the food of animal sources (Schweigert et al. Citation2002). Taurine, an amino acid, is essential for cats and they need to get it through dietary animal sources (Knopf et al. Citation1978). Cats lack the ability to convert tryptophan into niacin, a vitamin, and therefore it is required to be taken in through diet (Henderson et al. Citation1949). Arachidonic acid, a fatty acid, is also an essential nutrient for cats since they lack the enzyme necessary to convert linoleic acid to arachidonic acid and therefore their diet should contain sufficient amounts of arachidonic acid (Sinclair et al. Citation1979). The facts mentioned above support the hypothesis that cats are strict carnivores and therefore their diets need to be formulated precisely in order to supply all these essential nutrients ().
Different cats have different preferences towards specific dietary flavours and individual variation in the type of diet given to kittens contributes to differences in diet/flavour selection when they reach adulthood. Therefore, a diet must meet both the nutrients necessary for cats, especially the essential ones, and also flavours that can encourage feeding to be counted as complete and palatable. Without palatability, being complete and balanced diet is not enough for optimal consumption. Thus, diet palatability, which can be increased by using dietary additives such as flavours or natural ingredients, should be high enough to prevent any potential food consumption problems in cats.
2. Eating habits of cats
Despite the domestication process, cats still have the ability to hunt when it is necessary. Because of cats’ innate ability to hunt, they would live solitarily without any human interference in the wild (Bourgeois et al. Citation2006). Long canine teeth and shorter incisors and molars make cats very effective at hunting their prey and stripping the meat from its bones (Van Valkenburgh and Ruff Citation1987). Cats can hunt preys that are smaller in size several times daily in the nature. Fitzgerald and Turner (Citation2000) showed that cats would kill 12 small animals (rodents mostly) on average in order to meet their daily energy and nutrient requirements. This instinctive behaviour must have been inherited from its ancestors and probably helps explain the habit of eating frequent and small meals observed today in domesticated cats (Bradshaw and Thorne Citation1992). In contrast, dogs are prone to eat big meals and mostly use this opportunity as a socialization period like humans. Cats are considered to be a strict carnivore and their diets mostly consist of muscle and organ meat of other animals (Lei et al. Citation2015). Consequently, the percentage of metabolizable energy that is provided from proteins and fats are very high (up to 90–95%) and carbohydrates should not contribute to the metabolizable energy by more than 10%. However, carbohydrate sources are less expensive and most commercial dry foods contain a lot more carbohydrate (around 40%) than cats’ normal requirement (Hilton Citation1987). Cook et al. (Citation1985) reported that a diet with high palatability and low protein content was preferred over a less palatable but complete and balanced diet. Therefore, the palatability of diet plays an important role in optimizing energy and nutrient intake in domestic cats through ensuring enough food consumption. Odour, taste, texture, and particle size are additional factors that play role in food intake preferences by cats (Hullar et al. Citation2001; Small and Prescott Citation2005).
3. Appetite
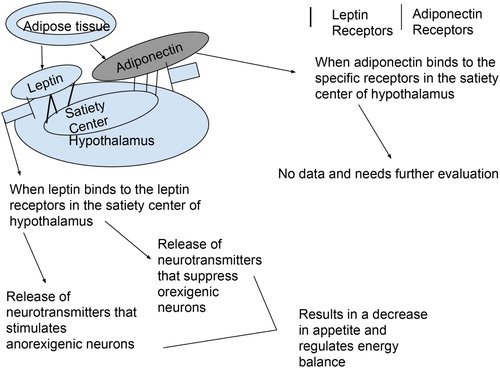
Appetite, by a broad definition, covers a lot of aspects such as palatability, eating frequency, size of eating episodes, energy density of foods eaten, and diversity of food consumed by cats (Arora and Anubhuti Citation2006; ). Appetite regulation is mainly controlled by signals produced both in hypothalamus and peripheral organs such as adipose tissue and digestive tract (Erlanson-Albertsson Citation2005). Hormones or cytokines produced by adipose tissue have become widely known as adipokines and they are involved in the regulation of appetite, energy balance, glucose, and lipid metabolism (Zoran Citation2010). Leptin is a protein hormone secreted from adipose tissue that has been shown to decrease appetite and often referred to as a ‘adipostat’ in cats (Appleton et al. Citation2000; Shibata et al. Citation2003). Since leptin is secreted from adipose tissue, the higher body fat levels are associated with higher plasma leptin concentrations (Appleton et al. Citation2000). Leptin works as a negative feedback mechanism in cats to limit food consumption and helps decrease accumulation of more fat in the body. Therefore, circulating leptin levels has been reported to be positively correlated to body fat levels in cats (Hoenig and Ferguson Citation2002). Although leptin receptors are widely distributed throughout the body, they are mainly located within the satiety centre of hypothalamus (Houseknecht and Portocarrero Citation1998). The binding of leptin with the receptors located within the hypothalamus results in the release of two different types of neurotransmitters; the first type stimulates anorexigenic neurones, and the second type suppresses the orexigenic neurones. Therefore, leptin plays a key role in reducing appetite and thus controlling food intake in cats (Appleton et al. Citation2000; Coppari et al. Citation2005). Adiponectin is another cytokine and acts as a food consumption regulator together with leptin in cats and recently has been shown to be a target molecule for the treatment of obesity and diabetes in cats (Ishioka et al. Citation2009). The same authors found that obese cats had lower plasma adiponectin concentration when compared to non-obese cats. Tvarijonaviciute et al. (Citation2012) reported a significant increase in plasma adiponectin concentration after a weight loss in obese cats. Although there is some evidence to indicate the involvement of adiponectin in fat accumulation in cats, its role on food intake or appetite remains to be further studied, especially in obese cats with increased appetite. A schematic figure summarizing the role of leptin and adiponectin is shown in .
Figure 2. Schematic representation of the main factors involved in appetite and diet palatability in cats.
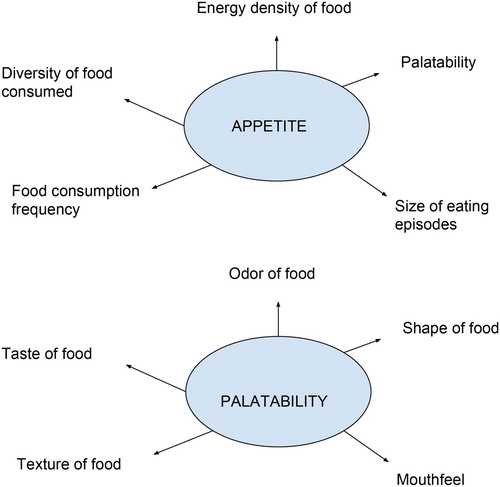
Figure 3. Schematic representation of the role of leptin and adiponectin in appetite control in cats.
Ghrelin (a gut hormone) increases appetite and plays a role in the control of food intake and its plasma concentration has been shown to be inversely correlated with dietary fat concentrations in cats (Backus et al. Citation2007). Another gut hormone named cholecystokinin activates peripheral and central cholecystokinin receptors and causes satiety and acts as a mechanism to limit food intake in cats (Bado et al. Citation1991). Neuroendocrine hormones named glucagon-like peptide-1 and peptide tyrosine are also released from intestinal cells in response to food intake and are involved in the satiety response but there is a lack of research on their role in regulation of appetite and therefore on energy expenditure in cats. The analogues of glucagon-like peptide-1 are used to decrease body weight through a speculated loss of appetite in humans, however whether the same effect occurs in cats remains currently unknown (DeFronzo et al. Citation2008; Hoelmkjaer et al. Citation2016).
An approximately 15% less food intake has been reported in summer compared to winter in cats during a 4-year cohort study that was conducted in the south of France where there is a Mediterranean climate (Serisier et al. Citation2014). These authors indicated that the seasonal consumption differences did not affect the body weights and therefore they speculated that the change in energy requirement was responsible for the food intake difference. Therefore, exact detailed mechanism responsible for these changes is still unclear in cats.
4. Sense of taste in cats
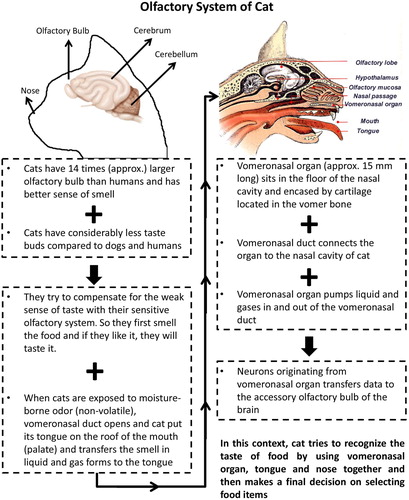
While cats’ sense of taste helps them evaluate the nutrient content of a food, it also protects them from eating toxic, harmful or indigestible ingredients as much as possible. Chemical sensors that respond to a variety of chemicals are called taste buds which are located in tongue, palate, pharynx, and larynx in cats (Shin et al. Citation1995). They have relatively small numbers of taste buds (approximately 470) compared to dogs, cows, and humans which have around 1700, 20,000, and 10,000 taste buds, respectively. Therefore, cats have a weaker taste sensitivity compared to most other animals (Davies et al. Citation1979; Ganchrow and Ganchrow Citation1987; Robinson and Winkles Citation1990). Taste buds can detect five basic tastes which are classified as salty, sour, bitter, sweet, and umami (Li Citation2013). Although cats have a functional sense of taste like most mammals; they lack sweet taste receptors and show no preference for sweet compounds such as sucrose (Li et al. Citation2005). The reason for the lack of sweet taste perception has been largely attributed to the deletion of Tas1r gene of sweet taste receptors found in taste buds (Li et al. Citation2005, Citation2006). Results of a recent study indicate that a mutation is responsible for the deletion of TIR 2 gene very early in the evolution of cats (Adler Citation2014). Therefore, they do not show any preference for glucose, sucrose, and fructose naturally. Cats have been also reported to reject non-nutritive sweeteners like saccharin and cyclamate (Bartoshuk et al. Citation1975). Domestic cats have been reported to have at least seven different functional bitter taste receptors but they tend to reject bitter foods where the opposite is true for dogs and most other mammals (Sandau et al. Citation2015). Electrophysiological records showed that water is not tasteless to cats (Bartoshuk et al. Citation1971). The very few number of taste buds found in cats led them to perceive the taste of food using other complementary senses. Among these alternative senses, odour appears to be the most important one. It is a well-known fact that cat’s sense of smell is 14 times better than humans. The reason for the better sense of smell was largely attributed to having 2 times more receptors in the nasal epithelium of cats (Padodara and Jacob Citation2014). Additionaly, cats have a vomeronasal organ, which is also called Jacobson’s organ located in the roof of their mouth which has a duct that connects it to both nose and mouth. Vomeronasal organ lying along the base of the nasal cavity, with an average length of 15 mm, opens into mouth by vomeronasal duct on the lateral side of incisive teeth through nasal septum laterally (Chung et al. Citation2018; Salazar et al. Citation1995). Vomeronasal organ is almost entirely surrounded by a cartilage. This organ compensate for low taste detection ability of cats since they have very few numbers of taste buds on their tongue and cats use his nose, mouth, and vomeronasal organ collectively to arrive at a decision on the taste of food item (Salazar et al. Citation1996). When a cat smells food, they open their mouth, put their chin in lower position, curve the tip of their nose, then communication starts between the vomeronasal organ and nasal cavity through the vomeronasal duct, while cat rubs its tongue against palate and transfers the smell to the tongue where taste buds are located. Thus, cats use vomeronasal organ, nose, and tongue together to describe the taste of a compound since its limited capacity for the utilization of taste by low numbers of taste buds (Papes et al. Citation2010). The vomeronasal organ also functions in determining pheromones and play a major role in determining sexual behaviour in cats (Doving and Trotier Citation1998; Hart and Leedy Citation1987). Parts of cat’s olfactory system and its role in determining the taste of foods are summarized in .
4.1. Taste preference in cats
Cats were reported to prefer fish and commercial food over rat in a laboratory setting (Houpt and Smith Citation1981). Beauchamp et al. (Citation1977) reported that the increased preference for foods with large amounts of protein and fat might be considered proof that cats are obligate carnivores. Cats uses odour information generated from food as an effective tool to identify the source of food to be eaten. If a cat finds an odour of a food more attractive than another, he will keep eating it without tasting the other food (Hullar et al. Citation2001).
4.2. Behavioural responses to different tastes in cats
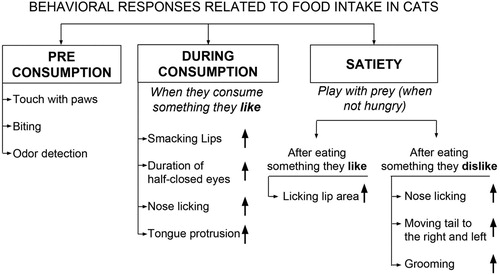
Measuring just preference and consumption patterns when concerning the palatability of a diet may not be complete without assessing behavioural responses of cats. When evaluating a cat’s behavioural responses to a food item, the facial expressions should be the very first one to be evaluated. Facial expressions include the motion of face, tongue, eyes, and nose. Relationship between these expressions and taste of a food can be used in the analysis of palatability and proved to be helpful when combined with preference and consumption data in cats. Behavioural responses related to food consumption may be classified into three: (a) those related to the taste of food; (b) those related to consumption; and (c) those related to satiety (). Touching the food with paws and biting are good behavioural examples related to the taste of any substance for cats. Behavioural responses during feeding including eye and face movements can be indicative of the palatability of the food they consume. Most house cats are likely to play with their prey when they are not hungry and they play to practice their hunting skills. Therefore, playing with food or prey can be given as an example for behavioural responses related to satiety in cats (Levine et al. Citation2016; Leyhausen Citation1979). Hanson et al. (Citation2016) reported an increase in the duration of ‘half-closed eyes’ when they consume a food they prefer. The same authors also noted that when cats eat something they like, they tend to do the behavioural responses such as nose licking, tongue protrusion, smacking lips longer compared to food they do not prefer. Becques et al. (Citation2014) investigated the behavioural responses when given highly palatable or less palatable dry food to cats during 20 h period a day. They concluded that feeding the highly palatable diet resulted in a significant decrease in the length of sniffing the food which corresponds to less hesitation to consume it when compared to less palatable diet on the two first visits to feeding station of first day. After cats eat something they like, they do the licking of the lip region more frequently. On the other hand, after they consume a food they dislike; licking their nose, moving their tail to the right and left, and increased grooming are the behavioural indicators which may be seen at higher frequencies (Savolainen et al. Citation2016). Similarly Van den Bos et al. (Citation2000) reported a significant increase in total duration and frequency of lip-licking after consuming a more palatable diet compared to a less palatable diet. The novelty effect or ‘neophilia’ is mostly occurred with cats that have been fed a single food or diet for a long time. These cats were reported to show a higher preference for a new diet when they were given a chance to select between the diet they used to eat and a new one. This response has been attributed to cats’ evolutionary habit towards consuming more than one food source to prevent any nutritional deficiencies (Bradshaw Citation2006; Stasiak Citation2002). The duration of preferring the novel food depends on its palatability. In contrast, some cats show resistant to a new diet, especially when they were fed one type diet or flavour for years, this form of behaviour is called ‘neophobia’. This type of behaviour has been reported to be a strategy of cats to avoid any toxic or poisonous food item. This type of diet rejection is most commonly reported under physiological, emotional, or environmental stress in cats. Giving a new type of food during a visit to veterinarian, or when a cat has a disease or pain can be good examples of this effect (Bourgeois et al. Citation2006; Bradshaw Citation1991; Bradshaw et al. Citation1996). Thus, it is always a good idea to introduce a new food under positive and usual circumstances to avoid neophobia situation.
5. Palatability
Palatability can be defined as the overall pleasant sensations related to the hedonic or sensory attributes obtained from ingested food that contributes with its acceptability in animals (Hall et al. Citation2018; Stubbs and Whybrow Citation2004; Yeomans et al. Citation1997). Lists of variables affecting palatability have been identified in cats in the literature and are discussed below in this review. However, complex interactions between many factors related to animal and food have been a major issue for the pet food industry.
6. Factors affecting food palatability in cats
6.1. Animal-Related factors affecting food palatability in cats
6.1.1. Preweaning feeding and its influence on feed preference in adult cats
According to Bradshaw (Citation2006), cats observe their mothers’ feeding practices which can affect their food preferences later in the adulthood period. Hamper et al. (Citation2012) fed cats with either raw or canned diet from post weaning 9 weeks to 20 weeks. Then, the same cats were fed with only dry food between 7 and 23 months of age. Cats demonstrated a reduced acceptance of canned (moist diet) food after 23 months of age. It was concluded that feeding neither raw nor canned food earlier affected the transition from dry food to moist food during adulthood. Stasiak (Citation2001) fed one group of cats with tuna only while the other group was fed with beef only during a 3-week-old to 6-month-old period. After feeding only one type of food, cats were retrained using the alternated food. Stasiak (Citation2001) demonstrated that both the beef and tuna cats preferred tuna in stages with alternated food. However, when cats were nondeprived of a food taste during 6-month period, no difference in attractiveness of food tastes observed. Therefore, they concluded that the deprivation of different food tastes could reveal an inborn food choice. Feeding cats through a stomach tube during the first 75 days of their lives had a detrimental role on sensory system that activates the reward mechanism, although they could able to learn to perceive the food reward as attractive (Stasiak and Zernicki Citation2000). Bradshaw et al. (Citation2000). showed that the house cats had an aversion towards raw beef while the farm cats consumed a little of the hard-dry food which might have been difficult to ingest. Therefore, they speculated that the way of life and prior dietary experiences play a role in food preferences in adult cats.
6.1.2. Hunger level
Physiological state of hunger has been shown to affect feeding behaviour in cats (Peachey and Harper Citation2002). Van den Bos et al. (Citation2000) reported that cats prefer to eat more palatable food regardless of their hunger level. However, they also showed that cats consume the less palatable diet depending on their hunger level.
6.1.3. Age of cat
Aging results in a significant decrease in olfactory receptors and fibres, thereby reducing the sense of smell. In addition, aging also has been associated with a concomitant loss of taste in cats (Boyce and Shone Citation2006). Despite this, voluntary eating behaviour of cats has been found to be stable in response to aging (Taylor et al. Citation1995). Feeding a more palatable diet, moistening dry food by adding warm water, and feeding fresh food more frequently have proven to be effective ways of encouraging old cats with appetite problem to consume a satisfactory amount of food to maintain a constant nutrient balance (Laflamme Citation2005).
6.2. Dietary variables that affect the palatability of diets for cats
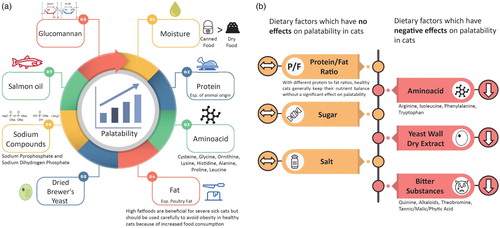
Dietary factors contributing to diet palatability are summarized in (a,b).
Figure 6. (a). Dietary factors which have positive effects on palatability in cats. (b). Dietary factors which have no or negative effects on palatability in cats.
6.2.1. Moisture
Although cats can consume dry or semi-moist foods without a problem, they mostly prefer wet or canned food over dry foods since the moisture level of canned food is very close to that of meat (70–85%) (Zaghini and Biagi Citation2005).
6.2.2. Protein source and content
Kittens were reported to show an impressive regulation of protein intake and also have an upper limit for carbohydrate intake which constrains them to deficits in protein and fat intake on carbohydrate-rich foods (Hewson-Hughes et al. Citation2011). There is a strong positive correlation between the protein level of the food and its palatability, especially when protein sources of animals are used (Zaghini and Biagi Citation2005). Diets formulated for cats are known to vary greatly in protein sources and they can be classified into either vegetable or animal origin. Soybean and soybean-derived products are the main vegetable-based protein sources in cat diets, especially in vegetarian diets; however, they also have low palatability that limits inclusion in diets for cats (Redmon et al. Citation2016). One common industry practice is to use other foods or additives such as porcine liver or polyphosphates to increase the overall palatability of the cat diets (Zentek and Schulz Citation2004). On the other hand, collagen tissue has been used in relatively low-priced cat foods as a source of animal protein. However, it also has a very low palatability compared to muscle meat sources and again the addition of another high palatability ingredient is necessary (Paßlack et al. Citation2017).
6.2.3. Protein/fat ratio
It has been shown that cats are able to regulate and balance their protein and fat intake regardless of its flavour (fish, rabbit or orange) even with different protein to fat ratios (from 10:90 to 70:30) that contribute to the energy density of the diet (Hewson-Hughes et al. Citation2016). Therefore, the authors concluded that macronutrient composition and organoleptic features of diet mostly play independent roles in diet selection by cats but these factors might interact in some cases.
6.2.4. Amino acids
The taste buds in cats are innervated by four different cranial nerves in the mouth (Oliveira et al. Citation2016). The receptors in facial nerve mainly react to tastants such as amino acids, nucleotides, sugar, etc. These reactions may result in either positive or negative response in the central nervous system of cats. Cats have been shown to respond positively to amino acids such as proline, cysteine, ornithine, lysine, histidine, and alanine which results in sweet taste perception in humans (Bradshaw et al. Citation1996). On the other hand, it was confirmed that ‘bitter’ amino acids such as arginine, isoleucine, phenylalanine, and tryptophan were widely rejected by cats due to negatively affected receptors in the facial nerve (Oliveira et al. Citation2016; Zaghini and Biagi Citation2005). Another report also showed that cats rejected L-tryptophan, although they showed a high preference for L-lysine when given as a pure solution (White and Boudreau Citation1975). In contrast, Leucine which has a bitter taste in humans is a positive flavour in cats (Beauchamp et al. Citation1977).
6.2.5. Fat
It is a well-known fact that palatability of food increases proportionally as the fat content is increased. Therefore, increasing the fat content of a diet is a common practice in cats with anorexia. High-fat diet can help meet cats’ energy requirement with higher palatability even their food consumption is lower than expected during an anorexic period, except for cats with pancreatitis (Delaney Citation2006). On the other hand, Kane et al. (Citation1987) did not observe any clear palatability pattern for the low- or high-fat diet in two diet-choice trials. Dietary salmon oil also leads to higher palatability and promote food intake in cats (Filburn and Griffin Citation2005).
6.2.6. Sugar
In a study using electrophysiological recordings, Bartoshuk et al. (Citation1971) showed that water was not tasteless to cats. Some authors also reported that although cats were indifferent to sucrose, with the addition of small amounts of sodium chloride to suppress the taste of water, cats were able to consume sucrose. Also, cats were reported to consume sucrose or lactose when they were offered in diluted milk (Beauchamp et al. Citation1977).
6.2.7. Salt and minerals
Cats were reported to be insensitive to salt similar to sugar morphologically and physiologically as opposed to ruminants and most other herbivores (Bradshaw Citation2006; Li et al. Citation2006). Therefore, they do not have the appetite for salty food that most mammals have.
Alegría-Morán et al. (Citation2019) reported that mineral components including ash and calcium had negative effects on food preferences in cats by analysing data from a 10-year database of two-feeder food preference tests between 2007 and 2017.
6.2.8. Cellulose
Cats tend to show less preference to foods with kaolin or cellulose (Hirsch et al. Citation1978). Moreover, Prola et al. (Citation2006) reported that cats fed on a diet with 6% added cellulose could eat the same amount of food compared to control diet without added cellulose; and therefore a significant decrease in energy intake reported for cellulose fed cats. In this regard, this phenomenon can be used to limit energy intake in especially obese cats. Alegría-Morán et al. (Citation2019) indicated that dietary crude fibre could negatively affect their food preferences as a result of a significant linear regression analysis between dietary fibre and diet preference in a 10-year database food preference study.
6.2.9. Dilution of food with liquid
Cats usually do not have a high preference for dilution of their food with a non-caloric liquid (Hirsch et al. Citation1978; Kanarek Citation1975).
6.2.10. Warmth and shape of food
Cats prefer to eat dry food at room temperature and they also likely to have more tendency to prefer ‘easy to grasp’ foods in shape (NRC Citation2006).
6.2.11. Ph of food
Cats usually show higher preference for acidic (pH = 4.5–5.5) substances (Thombre Citation2004).
7. Flavour and palatability enhancers
Substances that increase the overall palatability of a food for cats are called ‘flavour enhancers’ or ‘palatability enhancers’ and this area of research is of considerable interest by pet food manufacturers.
The palatability of any food item for cats is strongly related to its high quality attributes such as taste, odour, shape, texture, and sensation of mouthfeel (Small and Prescott Citation2005). The odour perception is very important for cats and plays a key role in choosing whether to eat a food item or not. Cats will use odour to define what foods are appropriate for their need and also will help them perceive toxic substances to their body. In this regard, cats compensate for their relatively low ability to taste foods because of having low numbers of taste buds by using their much more developed olfactory system. It has previously shown that cats prefer salmon alone over cat foods mixed with fish, liver, chicken, or beef flavour (Adamec Citation1976). Cats can be easily attracted to a food by its odour initially, especially under the conditions when they can smell properly.
Flavour enhancers for cats usually affect food palatability in cats in two different ways. The first one, called flavours that affect ‘first choice’ which is the first food item tasted by cats in preference tests and these flavours mostly affect olfactory perception of cats and improve attractiveness of the food. The second one, and the most important flavour enhancers are classified as having a ‘continuous choice effect’ in cats when they are given the same food with the same flavour again and it reflects actual acceptance of a food item by cats in the long run (Tobie et al. Citation2015). In the continuous choice affect, taste, mouthfeel, texture, etc. have bigger contribution to the palatability of the specific food item with flavour than just the odour of the specific food.
Flavour enhancers in pet food are classified as natural or synthetic. Examples of natural flavours given in the US Code of Federal Regulations are the essential oil, oleoresin, essence or extractive, protein hydrolysate, distillate or any product of roasting, heating or enzymolysis. The same regulation also describes the origin of the natural flavours and they could be obtained from plant materials such as spices, fruits, leafs but also may be obtained from animal products such as meat, poultry, and seafoods. However, to be classified as flavour, their major role must be just for flavouring rather than nutritional (Thombre Citation2004; Yerger Citation2003). Flavours do not meet these criteria above are classified as synthetic flavours under the same regulation. The efficacy of these flavours depends on multiple factors including dietary and individual differences. For example, a flavour that enhancing the palatability in dry foods might not be as effective in semi-moist or canned foods. Animal proteins, amino acids, and fat are the most efficacious flavours for cats compared to the flavours of plant origin.
8. Food additives
A vast number of compounds may be incorporated into cat foods for nutritional, functional, and also for palatability purposes. Apart from palatability enhancing, food additives can also be used for purposes such as dental cleaning (eg. phosphates), giving colour to food or freshening breath of cats or even masking unpleasing odour to humans in cat diets (eg. vanilla scent). Most commonly used food additives for palatability purposes are discussed below.
8.1. Hydrolysed proteins
Hydrolysis of proteins (mostly meat) by different methods and using them dietary to improve animal performance via different mechanisms (chemical, enzymatic, or microbial) is being studied by different research groups. Disruption of protein molecules results in the production of a vast amount of freely available bioactive peptides which exert a wide range of activities affecting digestion, immune, and central nervous systems (Korhonen and Pihlanto Citation2006). Soy sauce is one of the earliest and good examples of protein hydrolysates to improve the palatability of foods for humans (Pasupuleti and Demain Citation2010). Protein hydrolysates are also called ‘digest’ in animal nutrition and can be in dry or liquid forms with a common dietary application rate of 1–3% as a coating (Nagodawithana et al. Citation2010). Protein hydrolysates are among the most popular palatability enhancers in commercial cat diets because of its high short peptide concentration and free amino acid content (Folador et al. Citation2006; Martı´nez-Alvarez et al. Citation2015). The proteins are broken apart so that the disrupted whole protein structure cannot be recognized by cats immune system and this help reduce the allergies related to these proteins in cats and therefore it is a vital part of any hypoallergenic cat food in the market currently (Cave Citation2006; Neklyudov et al. Citation2000).
8.2. Spray-dried plasma
Spray-dried animal plasma is routinely added especially in canned pet food products due to its high water holding capacity, and therefore promoting better foaming, gelling, and emulsifying properties (Rodríguez et al. Citation2016). Moreover, Polo et al. (Citation2005) reported higher palatability of diets containing spray-dried plasma than diet containing wheat gluten in cat foods.
8.3. Sodium pyrophosphate
Pyrophosphates are chemical compounds that are used as a raising agent or to improve texture, and flavour of foods (Terenteva et al. Citation2017). Oliveira et al. (Citation2016) reported that the coating cat food with 0.5% sodium pyrophosphate resulted in a significant increase in food consumption.
8.4. Yeast extract products
Both dried yeast, mostly comes as a byproduct from ethanol industry, and the brewer’s yeast, are used as a palatability enhancer in pet food industry worldwide. The palatability increase with the addition of yeast has been attributed to its high glutamic acid concentration which gives the umami or meaty aroma (Nagodawithana Citation1992). Although Swanson and Fahey (Citation2004) reported an increase in the palatability of diet for cats with the addition of 1% yeast, the same authors also reported a decrease in palatability with the 2% yeast inclusion. In another study, cats given 0.4% yeast wall dry extract had significantly lower diet palatability compared to cats given a diet without yeast extract (Aquino et al. Citation2010). Thus, the optimal inclusion rate of yeast should be achieved by testing it under different conditions for cat diets.
8.5. Choline
Salt-like taste attributes have been reported for choline chloride in rats and humans. Therefore, it was recommended as a strategy to replace salt with choline to limit the level of salt consumption in humans (Locke and Fielding Citation1994). Although, cats are known to do not possess appetite for salt, according to Lin et al. (Citation1997) addition of 0.3% choline chloride, which reported to have a salt-like taste in other animals, was helpful in increasing the palatability and overall food consumption of a dry cat food. However, physiological mechanism behind this relationship is not clear.
8.6. Salt
Studies also demonstrated that kittens lack the ‘salt appetite’ that most omnivorous animals have (Yu et al. Citation1997). Even sodium-depleted cats reported to show no preference for sodium solution or salted water over plain water. Therefore, cats normally do not show attraction to salty foods and this attribute makes it unsuitable as a palatability enhancer in cat diets. Thus, substantial attention needs to be given to protect cats from any sodium deficiency since they cannot select foods based on their sodium or salt content (Yu and Morris Citation1999).
8.7. Prebiotics
A significantly higher palatability was found in cats when given 0.6% dietary mannanoligosaccharide as a prebiotic in dry food by Aquino et al. (Citation2010). Inulin is another type of prebiotic and can be used in cat diets (Roberfroid and Delzeene Citation1998). Decreased plasma ghrelin levels were achieved through dietary use of inulin type fructans in human subjects (Harrold et al. Citation2013). The same study also revealed that this decrease in plasma ghrelin levels resulted in a significant increase in secretion of glucagon-like peptide-1 which lead to a decrease in hunger and reduced eating highly palatable foods. However, it remains unknown whether and how dietary inulin would affect hunger and therefore food intake and palatability of foods in cats.
9. Conclusion
This review summarized the current knowledge and developments on the understanding of taste preferences, palatability, and factors affecting cats’ reactions when selecting and consuming diets. Overall, studies on leptin, adiponectin, ghrelin, and cholecystokinin and how they regulate appetite are necessary for understanding the feline food intake and this might be important in everyday regulation of food palatability in cats. The use of behavioural responses of cats showing during tasting, consuming, and after consuming in combination with preference and food consumption data may be of benefit as to provide a more comprehensive and robust data on determining the palatability of foods for cats. Increasing moisture, protein, certain amino acids, and fat content of cat foods are effective and proven methods to improve palatability. However, cats are insensitive to dietary salt and sugar addition and therefore these ingredients should not be used as a way to increase the palatability of a food or diet for cats. Currently spray-dried plasma, yeast products, choline chloride, and hydrolysed proteins are the commonly used palatability enhancing food additives in cat diets by the pet food industry. Although protein hydrolysates are among the most popular palatability enhancers in the cat food industry, there is no specific bioactive peptide defined as a pure palatability enhancer in cat diets. Therefore, suggested future research directions on food palatability in cats can include developing easy and economical ways to produce specific functional molecules (esp. specific bioactive peptides for different ingredients) which will improve the acceptability of certain ingredients that are not desirable by cats. Taken together, the direction of future research should be towards the promising palatability enhancers with a clear pattern in cats.
Disclosure statement
No potential conflict of interest was reported by the author(s).
References
- Adamec RE. 1976. The interaction of hunger and preying in the domestic cat (Felis catus): an adaptive hierarchy. Behav Biol. 18:263–272. DOI:10.1016/S0091-6773(76)92166-0.
- Adler EM. 2014. Of BK regulation, repurposed taste receptors, and arrestin recruitment. J Gen Physiol. 144:273–274. DOI:10.1085/jgp.201411286.
- Alegría-Morán RA, Guzmán-Pino SA, Egaña JI, Sotomayor V, Figueroa J. 2019. Food preferences in cats: effect of dietary composition and intrinsic variables on diet selection. Animals (Basel). 9:372.
- Appleton DJ, Rand JS, Sunvold GD. 2000. Plasma leptin concentrations in cats: reference range, effect of weight gain and relationship with adiposity as measured by dual energy X-ray absorptiometry. J Feline Med Surg. 2:191–199. DOI:10.1053/jfms.2000.0103.
- Aquino AA, Saad FMOB, Santos JPF, Alves MP, Ferrazza RA, Miranda MCMG. 2010. Effects of spray-dried yeast cell wall on digestibility, score of faeces, and palatability of diets for cats. Arquivo Brasileiro de Medicina Veterinária e Zootecnia. 62:622–630. DOI:10.1590/S0102-09352010000300018.
- Arora S, Anubhuti. 2006. Role of neuropeptides in appetite regulation and obesity – a review. Neuropeptides. 40:375–401. DOI:10.1016/j.npep.2006.07.001.
- Backus RC, Cave NJ, Keisler DH. 2007. Gonadectomy and high dietary fat but not high dietary carbohydrate induce gains in body weight and fat of domestic cats. Br J Nutr. 98:641–650. DOI:10.1017/S0007114507750869.
- Bado A, Durieux C, Moizo L, Roques BP, Lewin MJ. 1991. Cholecystokinin-a receptor mediation of food intake in cats. Am J Physiol. 260:R693–R697. DOI:10.1152/ajpregu.1991.260.4.R693.
- Bartoshuk LH, Harned MA, Parks LH. 1971. Taste of water in the cat: effects on sucrose preference. Science. 171:699–701. DOI:10.1126/science.171.3972.699.
- Bartoshuk LM, Jacobs HL, Nichols TL, Hoff LA, Ryckman JJ. 1975. Taste rejection of nonnutritive sweeteners in cats. J Comp Physiol Psychol. 89:971–975. DOI:10.1037/h0077172.
- Beauchamp GK, Maller O, Rogers JG. 1977. Flavor preferences in cats (Felis catus and Panthera sp. J Comp Physiol Psychol. 91:1118–1127. DOI:10.1037/h0077380.
- Becques A, Larose C, Baron C, Niceron C, Féron C, Gouat P. 2014. Behaviour in order to evaluate the palatability of pet food in domestic cats. Appl Anim Behav Sci. 159:55–61. DOI:10.1016/j.applanim.2014.07.003.
- Bourgeois H, Elliott D, Marniquet P, Soulard Y. 2006. Dietary behavior of dogs and cats. Bull de L’Acad Veterinaire de France. 4:301–308. www.academie-veterinaire-france.fr.
- Boyce J, Shone G. 2006. Effects of ageing on smell and taste. Postgrad Med J. 82:239–241. DOI:10.1136/pgmj.2005.039453.
- Bradshaw JW. 2006. The evolutionary basis for the feeding behavior of domestic dogs (Canis familiaris) and cats (Felis catus). J Nutr. 136:1927S–1931S. DOI:10.1093/jn/136.7.1927S.
- Bradshaw JWS. 1991. Sensory and experiential factors in the design of foods for domestic dogs and cats. Proc Nutr Soc. 50:99–106. DOI:10.1079/PNS19910015.
- Bradshaw JWS, Goodwin D, Legrand-Defretin V, Nott HMR. 1996. Food selection by the domestic cat, an obligate carnivore. Comparative Biochemistry and Physiology Part A: Physiology. 114:205–209. DOI:10.1016/0300-9629(95)02133-7.
- Bradshaw JWS, Healey LM, Thorne CJ, Macdonald DW, Arden-Clark C. 2000. Differences in food preferences between individuals and populations of domestic cats Felis silvestris catus. Appl Anim Behav Sci. 68:257–268. DOI:10.1016/S0168-1591(00)00102-7.
- Bradshaw JWS, Thorne CJ. 1992. Feeding behaviour. In: Thorne C, editor. The Waltham book of dog and cat behaviour. Oxford: Pergamon; p. 115–129. http://agris.fao.org/agris-search/search.do?recordID=GB9406773.
- Cave NJ. 2006. Hydrolyzed protein diets for dogs and cats. Vet Clinics North Am: Small Anim Pract. 36:1251–1268. DOI:10.1016/j.cvsm.2006.08.008.
- Chung BS, Chung MS, Lee SB, Youn C, Park JS. 2018. Sectioned images of a cat head to contribute to learning of its sectional anatomy. Int J Morphol. 36:537–543. http://www.intjmorphol.com/wp-content/uploads/2018/06/art_28_362.pdf.
- Clutton-Brock JA. 1999. Natural history of domesticated mammals. Cambridge: Cambridge Univ. Press.
- Cook NE, Kane E, Rogers QR, Morris JG. 1985. Self-selection of dietary casein and soy-protein by the cat. Physiol Behav. 34:583–594. DOI:10.1016/0031-9384(85)90053-8.
- Coppari R, Ichinose M, Lee CE, Pullen AE, Kenny CD, McGovern RA, Tang V, Liu SM, Ludwig T, Chua SC, et al. 2005. The hypothalamic arcuate nucleus: a key site for mediating leptin’s effects on glucose homeostasis and locomotor activity. Cell Metab. 1:63–72. DOI:10.1016/j.cmet.2004.12.004.
- Davies RO, Kare MR, Cagan RH. 1979. Distribution of taste buds on fungiform and circumvallate papillae of bovine tongue. Anat Rec. 195:443–446. DOI:10.1002/ar.1091950304.
- DeFronzo RA, Okerson T, Viswanathan P, Guan X, Holcombe JH, MacConell L. 2008. Effects of exenatide versus sitagliptin on postprandial glucose, insulin and glucagon secretion, gastric emptying, and caloric intake: a randomized, cross-over study. Curr Med Res Opin. 24:2943–2952. DOI:10.1185/03007990802418851.
- Delaney SJ. 2006. Management of anorexia in dogs and cats. Vet Clinics North Am: Small Anim Pract 36:1243–1249. DOI:10.1016/j.cvsm.2006.08.001.
- Doving KB, Trotier D. 1998. Structure and function of the vomeronasal organ. J Exp Biol. 201:2913–2925. http://jeb.biologists.org/content/201/21/2913.
- Erlanson-Albertsson C. 2005. How palatable food disrupts appetite regulation. Basic Clinical Pharmacol Toxicol. 97:61–73. DOI:10.1111/j.1742-7843.2005.pto_179.x.
- Filburn C, Griffin D. 2005. Effects of supplementation with a docosahexaenoic acid- enriched salmon oil on total plasma and plasma phospholipid fatty acid composition in the cat. Int J Appl Res Vet Med. 3:116–123. DOI:10.1159/000129651.
- Fitzgerald M, Turner DC. 2000. Hunting behavior of domestic cats and their impact on prey populations. In: Turner D.C., Bateson P, editor. The domestic cat, 2nd ed. Cambridge: Cambridge University Press; p. 151–176. DOI:10.1017/CBO9781139177177.
- Folador JF, Karr-Lilienthal LK, Parsons CM, Bauer LL, Utterback PL, Schasteen CS, Bechtel PJ, Fahey GC. 2006. Fish meals, fish components, and fish protein hydrolysates as potential ingredients in pet foods. J Anim Sci. 84:2752–2765. DOI:10.2527/jas.2005-560.
- Ganchrow JR, Ganchrow D. 1987. Taste bud development in chickens (Gallus gallus domesticus). Anat Rec. 218:88–93. DOI:10.1002/ar.1092180113.
- Hall JA, Vondran JC, Vanchina MA, Jewell DE. 2018. When fed foods with similar palatability, healthy adult dogs and cats choose different macronutrient compositions. J Exp Biol. DOI:10.1242/jeb.173450.
- Hamper BA, Rohrbach B, Kirk CA, Lusby A, Bartges J. 2012. Effects of early experience on food acceptance in a colony of adult research cats: a preliminary study. J Vet Behav. 7:27–32. DOI:10.1016/j.jveb.2011.02.008.
- Hanson M, Jojola SM, Rawson NE, Crowe M, Laska M. 2016. Facial expressions and other behavioral responses to pleasant and unpleasant tastes in cats (Felis silvestris catus). Appl Anim Behav Sci. 181:129–136. DOI:10.1016/j.applanim.2016.05.031.
- Harrold JA, Hughes GM, O’Shiel K, Quinn E, Boyland EJ, Williams NJ, Halford JC. 2013. Acute effects of a herb extract formulation and inulin fibre on appetite, energy intake and food choice. Appetite. 62:84–90. DOI:10.1016/j.appet.2012.11.018.
- Hart BL, Leedy MG. 1987. Stimulus and hormonal determinants of flehmen behavior in cats. Horm Behav. 21:44–52. DOI:10.1016/0018-506X(87)90029-8.
- Henderson LM, Ramasarma GB, Johnson BC. 1949. Quinolinic acid metabolism. IV. urinary excretion by man and other mammals as affected by the ingestion of tryptophan. J Biol Chem. 181:731–738. http://www.jbc.org/content/181/2/731.full.pdf.
- Hewson-Hughes AK, Colyer A, Simpson SJ, Raubenheimer D. 2016. Balancing macronutrient intake in a mammalian carnivore: disentangling the influences of flavor and nutrition. R Soc Open Sci. 3. DOI:10.1098/rsos.160081.
- Hewson-Hughes AK, Miller AT, Hall SR, Simpson SJ, Raubenheimer D. 2011. Geometric analysis of macronutrient selection in the adult domestic cat, Felis catus. J Exp Biol. 214:1039–1051. DOI:10.1242/jeb.049429.
- Hilton JW. 1987. Carbohydrates in cat diets: digestion and utilization. Can Vet J. 28:129–129. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1680355/.
- Hirsch E, Dubos C, Jacobs HL. 1978. Dietary control of food intake in cats. Physiol Behav. 20:287–295. DOI:10.1016/0031-9384(78)90222-6.
- Hoelmkjaer KM, Wewer, Albrechtsen NJ, Holst JJ, Cronin AM, Nielsen DH, Mandrup-Poulsen T, Bjornvad CR. 2016. A placebo-controlled study on the effects of the glucagon-like peptide-1 mimetic, exenatide, on insulin secretion, body composition and adipokines in obese, client-owned cats. Gonzalez-Bulnes A PLoS One. 11:e0154727. DOI:10.1371/journal.pone.0154727.
- Hoenig M, Ferguson DC. 2002. Effects of neutering on hormonal concentrations and energy requirements in male and female cats. Am J Vet Res. 63:634–639. DOI:10.2460/ajvr.2002.63.634.
- Houpt KA, Smith SL. 1981. Taste preferences and their relation to obesity in dogs and cats. Can Vet J. 22:77–85. https://www.ncbi.nlm.nih.gov/pubmed/7248879.
- Houseknecht LK, Portocarrero CP. 1998. Leptin and its receptors: regulators of whole body energy homeostasis. Domest Anim Endocrinol. 15:457–475. DOI:10.1016/S0739-7240(98)00035-6.
- Hullar I, Fekete S, Andrasofszky E, Sz˜ocs Z, Berkenyi T. 2001. Factors influencing the food preference of cats. J Anim Physiol Anim Nutr. 85:205–211. DOI:10.1046/j.1439-0396.2001.00333.x.
- Ishioka K, Omachi A, Sasaki N, Kimura K, Saito M. 2009. Feline adiponectin: molecular structures and plasma concentrations in obese cats. J Vet Med Sci. 71:189–194. DOI:10.1292/jvms.71.189.
- Kanarek RB. 1975. Availability and caloric density of the diet as determinants of meal patterns in cats. Physiol Behav. 15:611–618. DOI:10.1016/S0031-9384(75)80037-0.
- Kane E, Leung PM, Rogers QR, Morris JG. 1987. Diurnal feeding and drinking patterns of adult cats as affected by changes in the level of fat in the diet. Appetite. 9:89–98. DOI:10.1016/0195-6663(87)90038-9.
- Knopf K, Sturman JA, Armstrong M, Hayes KC. 1978. Taurine: an essential nutrient for the cat. J Nutr. 108:773–778. DOI:10.1093/jn/108.5.773.
- Korhonen H, Pihlanto A. 2006. Bioactive peptides: production and functionality. Int Dairy J. 16:945–960. DOI:10.1016/j.idairyj.2005.10.012.
- Laflamme DP. 2005. Nutrition for aging cats and dogs and the importance of body condition. Vet Clinics North Am: Small Anim Pract. 35:713–742. DOI:10.1016/j.cvsm.2004.12.011.
- Lei W, Ravoninjohary A, Li X, Margolskee RF, Reed DR, Beauchamp GK, Jiang P. 2015. Functional analyses of bitter taste receptors in domestic cats (Felis catus). PLoS One. 10:e0139670. DOI:10.1371/journal.pone.0139670.
- Levine ED, Erb HN, Schoenherr B, Houpt KA. 2016. Owner’s perception of changes in behaviors associated with dieting in fat cats. J Vet Behav. 11:37–41. DOI:10.1016/j.jveb.2015.11.004.
- Leyhausen P. 1979. Cat behavior. New York, NY: Garland STPM Press. p. 118–134.
- Li F. 2013. Taste perception: from the tongue to the testis. Mol Hum Reprod. 19:349–360. DOI:10.1093/molehr/gat009.
- Li X, Li W, Wang H, Bayley DL, Cao J, Reed DR, Bachmanov AA, Huang L, Legrand-Defretin V, Beauchamp GK, Brand JG. 2006. Cats lack a sweet taste receptor. J Nutr. 136(7 Suppl):1932S–1934S. DOI:10.1093/jn/136.7.1932S.
- Li X, Li W, Wang H, Cao J, Maehashi K, Huang L, Bachmanov AA, Reed DR, Legrand-Defretin V, Beauchamp GK. 2005. Pseudogenization of a sweet-receptor gene accounts for cats’ indifference toward sugar. PLoS Genet. 1:e27–e35. DOI:10.1371/journal.pgen.0010003.
- Lin CF, Lin JK, Jewell DE, Toll PW, Stout NP, Prewitt LR, Inventors. 1997. Pet food composition of improved palatability and a method of enhancing the palatability of a food composition. United States Patent, US, 5, 690, 988. https://patents.google.com/patent/US5690988A/en.
- Locke KW, Fielding S. 1994. Enhancement of salt intake by choline chloride. Physiol Behav. 55:1039–1046. DOI:10.1016/0031-9384(94)90385-9.
- Mameno K, Kubo T, Suzuki M. 2017. Social challenges of spatial planning for outdoor cat management in amami oshima island, Japan. Global Ecol Conserv. 10:184–193. DOI:10.1016/j.gecco.2017.03.007.
- Martı´nez-Alvarez O, Chamorro S, Brenes A. 2015. Protein hydrolysates from animal processing by-products as a source of bioactive molecules with interest in animal feeding: a review. Food Res Int. 73:204–212. DOI:10.1016/j.foodres.2015.04.005.
- Nagodawithana T. 1992. Yeast-derived flavors and flavor enhancers and their probable mode of action. Food Technol. 11:138–144. https://ci.nii.ac.jp/naid/20000580365/.
- Nagodawithana TW, Nelles L, Trivedi NB. 2010. Protein hydrolysates as hypoallergenic, flavors and palatants for companion animals. In: Pasupuleti VK, Demain AL, editors. Protein hydrolysates in biotechnology. Dordrecht: Springer; DOI:10.3390/agriculture3010112.
- National research council (NRC). 2006. Nutrient requirements of dogs and cats. Washington, DC: National Academies. https://www.nap.edu/read/10668/chapter/1#iv.
- Neklyudov AD, Ivankin AN, Berdutina AV. 2000. Properties and uses of protein hydrolysates (review). Appl Biochem Microbiol. 36:452–459. https://link.springer.com/article/10.1007/BF02731888.
- Oliveira R, HaeseI D, Kill JL, Lima A, Malini PV, Thompson GR. 2016. Palatability of cat food with sodium pyrophosphate and yeast extract. Ciência Rural, Santa Maria. 46:2202–2205. DOI:10.1590/0103-8478cr20151651.
- Padodara RJ, Jacob N. 2014. Olfactory sense in different animals. Indian J Vet Sci. 2:1–14.
- Pallotto MR, de Godoy MRC, Holsher HD, Buff PR, Swanson KS. 2018. Effects of weight loss with a moderate-protein, high-fiber diet on body composition, voluntary physical activity, and fecal microbiota of obese cats. Am J Vet Res. 79:181–190. DOI:10.2460/ajvr.79.2.181.
- Papes F, Logan DW, Stowers L. 2010. The vomeronasal organ mediates interspecies defensive behaviors through detection of protein pheromone homologs. Cell. 141:692–703. DOI:10.1016/j.cell.2010.03.037.
- Paßlack N, Kohn B, Doherr MG, Zentek J. 2017. Impact of dietary protein concentration and quality on immune function of cats. PLoS ONE. 12:e0169822. DOI:10.1371/journal.pone.0169822.
- Pasupuleti VK, Demain A. 2010. Protein hydrolysates in biotechnology. Dordrech: Springer. https://www.springer.com/gp/book/9781402066733.
- Peachey SE, Harper EJ. 2002. Aging does not influence feeding behaviour in cats. J Nutr. 132:1735S–1739S. DOI:10.1093/jn/132.6.1735S.
- Phillips-Donaldson D. 2016. Global pet food sales update: ending 2016 on a high note, pet food industry 2016. http://www.petfoodindustry.com/blogs/7-adventures -in-pet-food/post/6207-global-pet-food-sales-updateending-2016-on-a-high-note.
- Polo J, Rodriguez C, Saborido N, Rodenas J. 2005. Functional properties of spray-dried animal plasma in canned pet food. Anim Feed Sci Technol. 122:331–343. DOI:10.1016/j.anifeedsci.2005.03.002.
- Prola L, Dobenecker B, Kienzle E. 2006. Interaction between dietary cellulose content and food intake in cats. J Nutr. 136(7 Suppl):1988S–1990S.
- Randi E, Ragni B. 1991. Genetic variability and biochemical systematics of domestic and wild cat populations (Felis silvestris: Felidae). J Mammal. 72:79–88. DOI:10.2307/1381981.
- Redmon JM, Shrestha B, Cerundolo R, Court MH. 2016. Soy isoflavone metabolism in cats compared with other species: urinary metabolite concentrations and glucuronidation by liver microsomes. Xenobiotica. 46:406–415. DOI:10.3109/00498254.2015.1086038.
- Roberfroid MB, Delzeene NM. 1998. Dietary fructans. Annu Rev Nutr. 18:117–143. doi: 10.1146/annurev.nutr.18.1.117
- Robinson PP, Winkles PA. 1990. Quantitative study of fungiform papillae and taste buds on the cat’s tongue. Anat Rec. 226:108–111. DOI:10.1002/ar.1092260112.
- Rodríguez C, Saborido N, Rodenas J, Polo J. 2016. Effect of spray-dried plasma on food intake and apparent nutrient digestibility by cats when added to a wet pet food recipe. Anim Feed Sci Technol. 216:243–250. DOI:10.1016/j.anifeedsci.2016.03.026.
- Salaun F, Le Paih L, Roberti F, Niceron C, Blanchard G. 2017. Impact of macronutrient composition and palatability in wet diets on food selection in cats. J Anim Physiol Anim Nutr. 101:320–328. DOI:10.1111/jpn.12542.
- Salazar I, Quinteiro PS, Cifuentes JM. 1995. Comparative anatomy of the vomeronasal cartilage in mammals: mink, cat, dog, cow and horse. Ann Anatomy – Anatomischer Anzeiger. 177:475–481. DOI:10.1016/S0940-9602(11)80156-1.
- Salazar I, Quinteiro PS, Cifuentes JM, Caballero TG. 1996. The vomeronasal organ of the cat. J Anat. 188:445–454. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1167581/pdf/janat00127-0181.pdf.
- Sandau MM, Goodman JR, Thomas A, Rucker JB, Rawson NE. 2015. A functional comparison of the domestic cat bitter receptors Tas2r38 and Tas2r43 with their human orthologs. BMC Neurosci. 16:33–44. DOI:10.1186/s12868-015-0170-6.
- Savolainen S, Telkänranta H, Junnila J, Hautala J, Airaksinen S, Juppo A, Raekallio M, Vainio O. 2016. A novel set of behavioural indicators for measuring perception of food by cats. Vet J. 216:53–58. DOI:10.1016/j.tvjl.2016.06.012.
- Schweigert FJ, Raila J, Wichert B, Kienzle E. 2002. Cats absorb beta-carotene, but it is not converted to vitamin A. J Nutr. 132:1610S–1612S. DOI:10.1093/jn/132.6.1610S.
- Serisier S, Feugier A, Delmotte S, Biourge V, German AJ. 2014. Seasonal variation in the voluntary food intake of domesticated cats (Felis Catus). PLoS ONE. 9(4):e96071. DOI:10.1371/journal.pone.0096071.
- Shibata H, Sasaki N, Honjoh T, Ohishi I, Takiguchi M, Ishioka K, Ahmed M, Soliman M, Kimura K, Saito M. 2003. Feline leptin: immunogenic and biological activities of the recombinant protein, and its measurement by ELISA. J Vet Med Sci. 65:1207–1211. DOI:10.1292/jvms.65.1207.
- Shin T, Nahm I, Maeyama T, Miyazaki J, Matsuo H, Yu Y. 1995. Morphological study of the laryngeal taste buds in the cat. Laryngoscope. 105:1315–1321. DOI:10.1288/00005537-199512000-00010.
- Sinclair AJ, McLean JG, Monger EA. 1979. Metabolism of linoleic acid in the cat. Lipids. 14:932–936. DOI:10.1007/BF02533508.
- Small DM, Prescott J. 2005. Odor/taste integration and the perception of flavor. Exp Brain Res. 166:345–357. DOI:10.1007/s00221-005-2376-9.
- Stasiak M. 2001. The effect of early specific feeding on food conditioning in cats. Dev Psychobiol. 39:207–215. DOI:10.1002/dev.1046.
- Stasiak M. 2002. The development of food preferences in cats: the new direction. Nutr Neurosci. 5:221–228. DOI:10.1080/1028415021000001799.
- Stasiak M, Zernicki B. 2000. Food conditioning is impaired in cats deprived of the taste of food in early life. Neurosci Lett. 279:190–192. DOI:10.1016/S03043940(99)00961-1.
- Stubbs RJ, Whybrow S. 2004. Energy density, diet composition and palatability: influences on overall food energy intake in humans. Physiol Behav. 81:755–764. DOI:10.1016/j.physbeh.2004.04.027.
- Swanson KS, Fahey GC. 2004. The role of yeasts in companion animal nutrition. In: nutritional biotechnology in the feed and food industries. In: Lyons TP, Jacques KA, editors. Proceedings of Alltech’s 20 annual symposium: re-imagining the feed industry. Lexington, Kentucky: Nottingham University Press; p. 475–484. http://www.hilyses.com/wp-content/uploads/2016/11/heinrichs-and-Kehoe-2004-pp-194-203.pdf#page=449.
- Taylor EJ, Adams C, Neville R. 1995. Some nutritional aspects of ageing in dogs and cats. Proc Nutr Soc. 54:645–656. DOI:10.1079/PNS19950064.
- Terenteva EA, Arkhipova VV, Apyari VV, Volkov PA, Dmitrienko SG. 2017. Simple and rapid method for screening of pyrophosphate using 6,6-ionene-stabilized gold and silver nanoparticles. Sens Actuators, B. 241:390–397. DOI:10.1016/j.snb.2016.10.093.
- Thombre AG. 2004. Oral delivery of medications to companion animals: palatability considerations. Adv Drug Delivery Rev. 56:1399–1413. DOI:10.1016/j.addr.2004.02.012.
- Tobie C, Péron F, Larose C. 2015. Assessing food preferences in dogs and cats: a review of the current methods. Animals (Basel). 5:126–137. DOI:10.3390/ani5010126.
- Tvarijonaviciute A, Ceron JJ, Holden SL, Morris PJ, Biourge V, German AJ. 2012. Effects of weight loss in obese cats on biochemical analytes related to inflammation and glucose homeostasis. Domest Anim Endocrinol. 42:129–141. DOI:10.1016/j.domaniend.2011.10.003.
- Van den Bos R, Meijer MK, Spruijt BM. 2000. Taste reactivity patterns in domestic cats (Felis silvestris catus). Appl Anim Behav Sci. 69:149–168. DOI:10.1016/S0168-1591(00)00124-6.
- Van Valkenburgh B, Ruff CB. 1987. Canine tooth strength and killing behaviour in large carnivores. J Zool. 212:379–397. DOI:10.1111/j.1469-7998.1987.tb02910.x.
- Vigne JD, Guilaine J, Debue K, Haye L, Gérard P. 2004. Early taming of the cat in Cyprus. Science. 304:259. DOI:10.1126/science.1095335.
- White TD, Boudreau JC. 1975. Taste preferences of the cat for neurophysiologically active compounds. Physiol Psychol. 3:405–410. DOI:10.3758/BF03326850.
- Yeomans MR, Gray RW, Mitchell CJ, True S. 1997. Independent effects of palatability and within-meal pauses on intake and appetite ratings in human volunteers. Appetite. 29:61–76. DOI:10.1006/appe.1997.0092.
- Yerger J. 2003. What is the difference between artificial and natural flavors? Sci Am. 288:111. https://www.scientificamerican.com/article/what-is-the-difference-be-2002-07-29/.
- Yu S, Morris JG. 1999. Sodium requirement of adult cats for maintenance based on plasma aldosterone concentration. J Nutr. 129:419–423. DOI:10.1093/jn/129.2.419.
- Yu S, Rogers QR, Morris JG. 1997. Absence of salt (NaCl) preference or appetite in sodium-replete or depleted kittens. Appetite. 29:1–10. DOI:10.1006/appe.1996.0088.
- Zaghini G, Biagi G. 2005. Nutritional peculiarities and diet palatability in the cat. Vet Res Commun. 29(Suppl. 2):39–44. DOI:10.1007/s11259-005-0009-1.
- Zentek J, Schulz A. 2004. Urinary composition of cats is affected by the source of dietary protein. J Nutr. 134:2162S–2165S. DOI:10.1093/jn/134.8.2162S.
- Zoran DL. 2010. Obesity in dogs and cats: a metabolic and endocrine disorder. Vet Clinics North Am: Small Anim Pract. 40:221–239. DOI:10.1016/j.cvsm.2009.10.009.